Efficacy and Safety of DA-8010, a Novel M3 Antagonist, in Patients With Overactive Bladder: A Randomized, Double-Blind Phase 2 Study
Article information
Abstract
Purpose
DA-8010 is a novel muscarinic M3 receptor antagonist with significant selectivity for bladder over salivary gland in preclinical studies. We evaluated the clinical efficacy and safety of DA-8010 in overactive bladder (OAB) patients.
Methods
This phase 2, randomized, double-blind, parallel-group, active reference- and placebo-controlled trial was conducted at 12 centers in South Korea (NCT03566134). Patients aged ≥19 years with OAB symptoms for ≥3 months were enrolled. Three hundred six patients (30.07% male) were randomized to 12 weeks of treatment among 4 groups; 2 experimental groups (DA-8010 2.5 or 5 mg), an active reference group (solifenacin 5 mg), and a placebo group. The change from the baseline of (=∆) 24-hour frequency at 12 weeks (primary endpoint), episodes of urgency, overall/urgency urinary incontinence, average/ maximum voided volume, nocturia, and patients’ subjective responses were analyzed.
Results
In the full analysis set, the mean (standard deviation) [median] values for ∆ 24-hour frequency at 12 weeks were -1.01 (2.44) [-1.33] for placebo, -1.22 (2.05) [-1.33] for DA-8010 2.5 mg, and -1.67 (2.25) [-1.67] for DA-8010 5 mg; DA-8010 5 mg showed a significant decrease compared with placebo (P=0.0413). At 4 and 8 weeks, both DA-8010 2.5 mg (P=0.0391 at 4 weeks, P=0.0335 at 8 weeks) and DA-8010 5 mg (P=0.0001 at 4 weeks, P=0.0210 at 8 weeks) showed significant decrease in ∆ 24-hour frequency compared with placebo. DA-8010 5 mg achieved a significant decrease in ∆ number of urgency episodes, compared with placebo at 4 (P=0.0278) and 8 (P=0.0092) weeks. Adverse drug reactions (ADRs) were observed in 3.95% of placebo, 6.67% of DA-8010 2.5 mg, 18.42% of DA-8010 5 mg, and 17.33% of solifenacin 5 mg groups. No serious ADRs were observed in any patient.
Conclusions
Both DA-8010 2.5 mg and 5 mg showed therapeutic efficacy for OAB without serious ADRs. Therefore, both dosages of DA-8010 can advance to a subsequent large-scale phase 3 trial.
INTRODUCTION
Overactive bladder (OAB) is a symptom complex including urgency, frequency, and nocturia, with or without urgency urinary incontinence, and is mainly associated with unnecessary bladder contraction or hypersensitivity in the storage phase [1,2]. Bladder contraction is primarily caused by activation of muscarinic receptor in the bladder [1,3]. Hyperactivity of bladder afferent nerves is also believed to be associated with the muscarinic receptor [3]. Therefore, antimuscarinic agents are regarded as the mainstay treatment for OAB [4]. Five subtypes of muscarinic receptors have been identified thus far—M1 to M5—and all of their mRNAs have been identified in the human bladder [1,5-7]. The major muscarinic receptors distributed in the detrusor smooth muscle are the M2 and M3 subtypes, existing in a 3:1 ratio [3]. Although smaller in number, M3 receptors are believed to play a major role in mediating bladder contraction [1,3,8], which may be a target of OAB treatment. Meanwhile, M3 receptors are also distributed in the salivary gland, gastrointestinal tract, eye, and brain [3,8,9], causing a broad range of bothersome side effects and resulting in poor persistence with antimuscarinics [10]. Therefore, the development of receptor-selective and organ-selective antimuscarinic agents is a crucial goal in the medical treatment of OAB.
DA-8010 ((R)-(1-methylpyrrolidin-3-yl) methyl (3’-chloro-4’-fluoro[1,1’-biphenyl]−2-yl) carbamate) is a novel compound developed by Dong-A ST Co., Ltd. (Korea) for the treatment of OAB. Preclinical in vitro studies have shown that DA-8010 has higher binding affinity to the human M3 receptor, and higher antagonistic activity against carbachol-induced intracellular Ca2+ release in cells expressing the human muscarinic M3 receptor than other antimuscarinics, such as tolterodine, oxybutynin, darifenacin, and solifenacin [11]. DA-8010 demonstrated greater functional selectivity for bladder cells over salivary gland cells than tolterodine, oxybutynin, darifenacin, and solifenacin in mice (in vitro), and higher selectivity for the bladder over the salivary gland than oxybutynin, darifenacin, and solifenacin in rats (in vivo), suggesting that DA-8010 is less likely to induce dry mouth and more likely to act faster and longer in the bladder [11]. Another in vivo study showed that DA-8010 had higher selectivity for the bladder over the brain than solifenacin in mice, suggesting a lower probability of side effects in the central nervous system [12]. The potential of DA-8010 as a therapeutic agent for OAB has been confirmed in another in vivo study in rats, showing a significant increase in the contraction interval and a significant decrease in contraction pressure in detrusor overactivity induced by bladder outlet obstruction [13].
DA-8010 has undergone a phase 1 clinical trial for 109 healthy volunteers in England (NCT02821312) to assess its safety and pharmacokinetics. Regarding safety, tolerability was excellent overall up to 10 mg, and poor tolerability was observed after 7-day repeated administration of 20 mg. The primary purpose of this phase 2 clinical trial was to evaluate the efficacy and safety of DA-8010 compared with placebo after administering DA-8010 for 12 weeks in OAB patients, and to select the optimal dose of DA-8010. Based on the results of the phase 1 trial, 2.5 and 5 mg of DA-8010 were used in this trial.
MATERIALS AND METHODS
Study Design and Patient Population
This Phase 2, multicenter, randomized, double-blind, parallelgroup, placebo-controlled, and active reference-controlled trial enrolled male and female patients aged ≥19 years with OAB symptoms (frequency, urgency, and/or urgency urinary incontinence) for ≥3 months. After a single-blind placebo run-in period of 2 weeks, patients with 24-hour frequency ≥8 and 72-hour urgency episode ≥6 (with or without incontinence), based on a 72-hour frequency-volume chart, were randomized to 12 weeks of treatment in one of 4 groups: 2 experimental groups (DA-8010 2.5 or 5 mg), a placebo, or an active reference (solifenacin succinate 5 mg) group in a 1:1:1:1 ratio. Using a double-blind, four-arm double-dummy technique, all the patients were administered 2 tablets daily during treatment; DA8010 (2.5 or 5 mg) or placebo, and solifenacin succinate 5 mg or placebo. The study protocol was registered at ClinicalTrials. gov (NCT03566134) and approved by an institutional review board at each of the 12 institutions. All the participants provided written informed consent.
Efficacy Assessment
The primary endpoint was ‘change from the baseline of (=∆)’ mean 24-hour frequency at the end of treatment (12 weeks). The secondary endpoints included the ∆ mean 24-hour frequency at 4 and 8 weeks; ∆ number of overall incontinence episode/24 hours, ∆ number of urgency urinary incontinence episodes/24 hours, ∆ number of urgency episodes/24 hours, ∆ number of nocturia episode/24 hours, ∆ average voided volume, and ∆ maximum voided volume at 4, 8, and 12 weeks; ∆ ‘patient perception of bladder condition’ (PPBC) scores at 4, 8, and 12 weeks; ∆ Overactive Bladder Questionnaire (OAB-q) scores at 4 and 12 weeks; and ‘benefit, satisfaction, and willingness to continue’ (BSW) at 12 weeks. For all the variables included in the primary and secondary endpoints, additional exploratory analyses were performed comparing the DA-8010 (2.5 and 5 mg) and solifenacin 5 mg groups.
Safety Assessment
Drug safety evaluations were performed using laboratory assessments, vital sign measurements, electrocardiography, postvoid residual urine volume, incidence of treatment-emergent adverse events (TEAEs; adverse events not present prior to study drug treatment or events that had already existed and aggravated in intensity or frequency after exposure to the study drug) and adverse drug reactions (ADRs; adverse events reasonably suspected to be induced by the study drug, which may occur in part due to the pharmacological action of the study drug).
Statistical Analysis
Assuming that the effect size for a mean 24-hour frequency in the DA-8010 group compared with placebo is 0.5, 63 patients in each group provided a power of 80% to detect a significant difference between the DA-8010 and placebo groups at a significance level (α) of 0.05 (2-sided). Considering the postulated dropout rate of 15%, 75 patients in each group (a total of 300 patients) were assigned for 1:1:1:1 randomization. Central allocation was performed using the stratified block randomization method, depending on the gender and presence or absence of urgency urinary incontinence. Treatment efficacy evaluations were performed for the full analysis set (FAS; comprising all patients who received at least one dose of the test drug and had primary efficacy data at baseline and at least one post-baseline assessment). To evaluate episodes of incontinence, FAS patients with at least one incontinence episode in the baseline diary (FAS-I [incontinence]) were included.
The statistical analyses of the major efficacy variables (comparing the DA-8010 2.5-mg and 5-mg treatment groups with the placebo group) and exploratory comparisons of the DA-8010 2.5 mg and 5 mg groups with the solifenacin 5 mg group were performed using the analysis of covariance (ANCOVA) model or rank ANCOVA model adjusting for the baseline values and gender as covariates. BSW (benefit, satisfaction, and willingness to continue) were analyzed using the Cochrane-Mantel-Haenszel test. To assess the efficacy variables, missing values were treated using the last observation carried forward method. All statistical analyses were performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA). Unless otherwise specified, statistical tests were performed using a 2-sided test with a significance level of 5%. Continuous variables were presented as numbers, means, standard deviations, and medians, and categorical variables were presented as frequencies and percentages (%).
RESULTS
Demographic and Baseline Characteristics
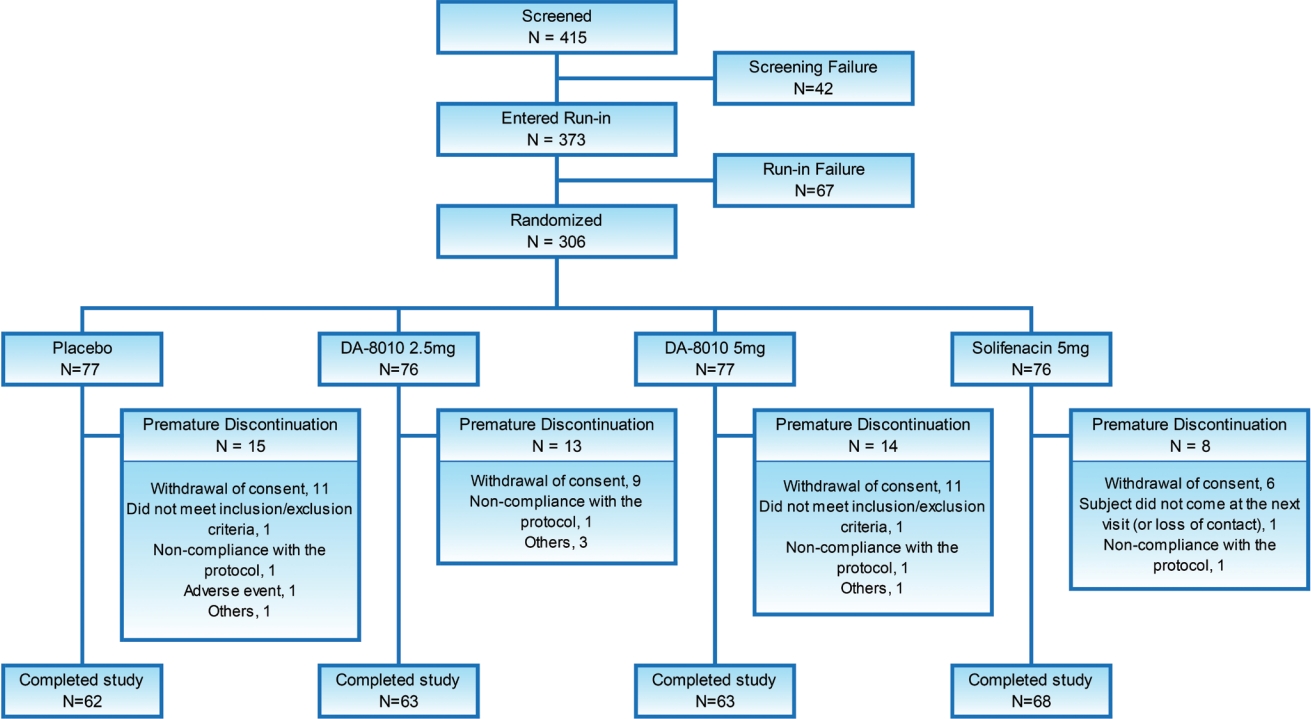
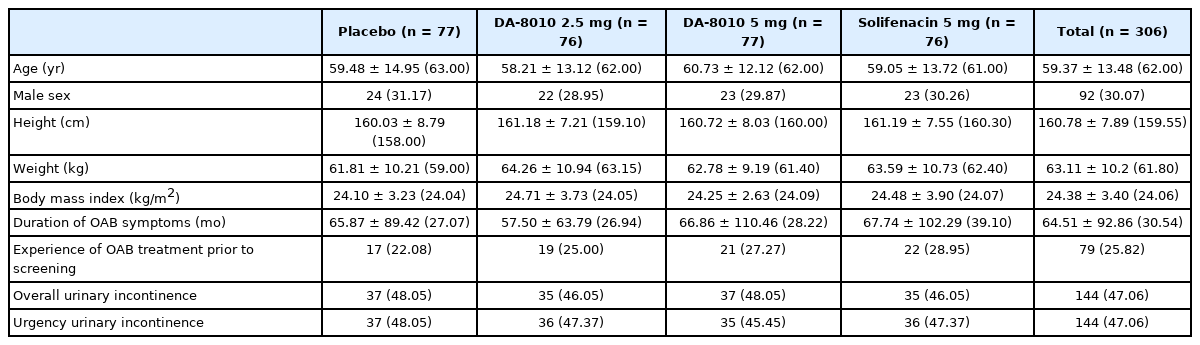
Patient screening was started in July 2018, and the trial ended in December 2019. Of 415 subjects screened, 373 proceeded to the single-blind placebo run-in phase, 306 were randomized (intention-to-treat [ITT] set), 302 received test drugs, 285 had at least one efficacy evaluation (FAS), 16.34% (50 of 306) dropped out after randomization, and 256 completed the trial. The mean±standard deviation (SD) age of 306 subjects included in the ITT set was 59.37±13.48 years, and 30.07 % were male. The mean ±SD OAB symptom duration period was 64.51 ± 92.86 months, and 25.82% had OAB treatment before screening. On a baseline diary basis, 47.06% of patients had episodes of urinary incontinence. No statistically significant difference was found among the placebo, DA-8010 2.5 mg, DA-8010 5 mg, and solifenacin 5 mg groups regarding the demographic and baseline characteristics (Fig. 1, Table 1).
Efficacy results
Primary endpoint: ∆ 24-hour frequency at 12 weeks
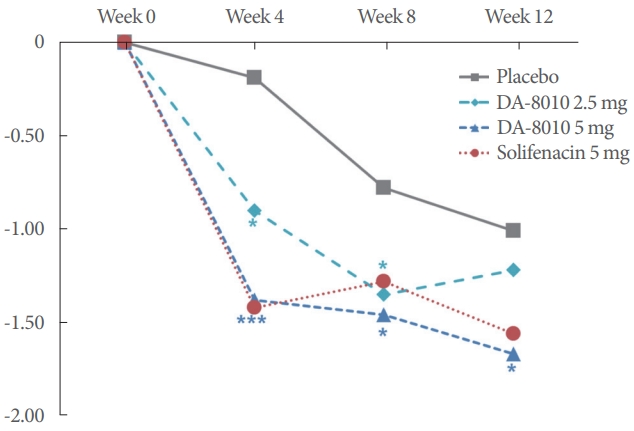
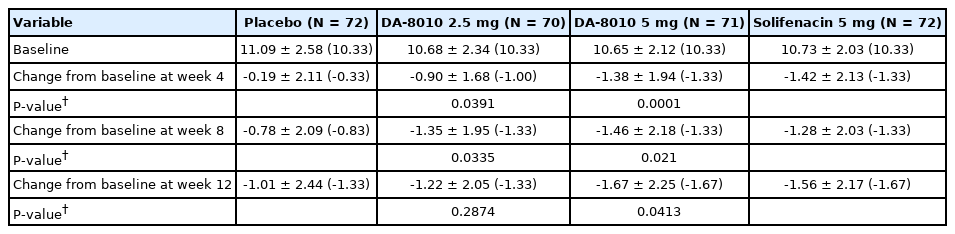
The mean (SD) [median] ∆ 24-hour frequency at the end of treatment (12 weeks) was -1.01 (2.44) [-1.33] in the placebo group, -1.22 (2.05) [-1.33] in the DA-8010 2.5 mg group, and -1.67 (2.25) [-1.67] in the DA-8010 5 mg group. Compared with placebo, DA-8010 2.5 mg showed no significant difference (P=0.2874), but DA-8010 5 mg showed a significant difference (P=0.0413) (Fig. 2, Table 2).
Secondary endpoints: DA-8010 vs. Placebo
Compared with that in the placebo group, the ∆ 24 hour mean frequency at 4 and 8 weeks were significantly lower in both the DA-8010 2.5 mg (4 weeks: P=0.0391; 8 weeks: P=0.0335) and DA-8010 5 mg (4 weeks: P=0.0001; 8 weeks: P=0.0210) groups. The ∆ episode of urgency/24 hours was significantly lower in the DA-8010 5 mg group at 4 weeks (P=0.0278) and 8 weeks (P=0.0092), while DA-8010 2.5 mg showed a trend toward significance at 8 weeks (P=0.0684). In addition, there were frequency volume chat variables that showed numerical effects but did not reach the conventional significance; nocturia episodes decreased in both the DA-8010 2.5 mg (P=0.0952) and 5 mg (P=0.0779) groups at 8 weeks. The ∆ average voided volume increased in the DA-8010 5mg group at 4 weeks, almost approaching statistical significance (P=0.0524) (Fig. 2; Tables 2, 3).

Secondary efficacy variables-frequency volume chart parameters (full analysis set; full analysis set-incontinence)
In terms of the patients’ subjective response, the proportion of patients who were ‘willing to continue treatment’ were significantly higher in both the DA-8010 2.5 mg (P=0.0238) and 5 mg (P=0.0030) groups than in the placebo group at the end of treatment (12 weeks). Regarding the other parameters of secondary efficacy variables, no statistically significant differences were found in both the DA-8010 2.5 mg and DA-8010 5 mg groups compared with those in the placebo group (Supplementary Tables 1, 2).
Exploratory analysis: DA-8010 versus solifenacin succinate 5 mg
Both DA-8010 2.5 mg and DA-8010 5 mg showed no significant differences compared with solifenacin 5 mg regarding the efficacy variables included in the primary and secondary endpoints, except for one parameter; The ∆ voided volume per micturition at 12 weeks was mean 14.34 mL for DA-8010 2.5 mg, 8.71 mL for DA-8010 5 mg, and 29.16 mL for solifenacin 5 mg. No significant difference was found between DA-8010 2.5 mg and solifenacin 5 mg (P=0.1991). However, a paradoxically significant difference was observed between DA-8010 5 mg and solifenacin 5 mg (P=0.0185). Regarding the urgency episode, numerical efficacy was observed in both the DA-8010 2.5 mg and 5 mg groups compared with the solifenacin 5 mg group, but the difference was not statistically significant. In the patient subjective assessment parameters regarding OAB symptoms, quality of life, and treatment satisfaction (∆ PPBC, ∆ OAB-q, BSW), no significant differences were observed between the 2 dosage groups of DA-8010 and solifenacin 5 mg (Fig. 2; Tables 2, 3; Supplementary Tables 1, 2).
Safety results
Of the 302 patients in the safety set (who received the allocated drug at least once and had at least one safety assessment), TEAEs were observed in 81 patients (26.82%, 124 cases); 14 patients (18.42%, 23 cases) in the placebo group, 19 patients (25.33%, 28 cases) in the DA-8010 2.5 mg group, 23 patients (30.26%, 35 cases) in the DA-8010 5 mg group, and 25 patients (33.33%, 38 cases) in the solifenacin 5 mg group (Table 4). ADRs were observed in 35 patients (11.59%, 42 cases); 3 patients in the placebo group (3.95%, 4 cases), 5 patients in the DA-8010 2.5 mg group (6.67%, 5 cases), 14 patients in the DA-8010 5 mg group (18.42%, 17 cases), and 13 patients in the solifenacin 5 mg group (17.33%, 16 cases). Among the 42 cases of ADRs, 36 cases (85.71%) were mild. Moderate ADRs were reported in 4 cases (9.52%); 2 patients in the DA-8010 5 mg group (headache and dysuria) and 2 patients in the solifenacin 5 mg group (constipation). Severe ADRs were reported in 2 cases (4.76%) in the placebo group (vomiting and nausea). No serious ADRs were observed in any of the study drug groups.
The most frequent ADRs were dry mouth, observed in 22 patients (7.28%), but the severity was mild in all groups. This was followed by constipation in 5 patients (1.66%) (Supplementary Table 3). Regarding organ-based classification, ADRs in the gastrointestinal tract were most frequent, reported in 26 patients (8.61%, 30 cases), followed by the nervous system and urinary tract; each in 5 patients (1.66%, each in 5 cases), skin/ subcutaneous tissue disorder and general condition change (fatigue); each in 1 patient (0.33%, each in 1 case). No ADRs resulted in the permanent discontinuation of treatment or death during this trial.
No significant difference was found in ∆ postvoid residual urine volume among the treatment groups at 4, 8, and 12 weeks (P=0.3254, P=0.8061, and P=0.8024, respectively). No clinically significant changes were observed in blood pressure, pulse rate, body temperature, or electrocardiogram findings in any of the patients. Additionally, no drug-related clinically significant abnormal findings were noted in the laboratory values (blood test and urinalysis) for all participants throughout the study period.
DISCUSSION
In this phase II trial, the novel M3 antagonist DA-8010 2.5 mg and 5 mg showed a dose-dependent effect in improving the OAB symptoms compared with placebo. Regarding the primary endpoint (∆ 24-hour frequency at 12 weeks), statistically significant efficacy was observed only in the DA-8010 5 mg group. However, at 4 and 8 weeks, significant efficacy in ∆ 24-hour frequency was observed in both the DA-8010 2.5 mg and 5 mg groups. Concerning the ∆ urgency episode, a significant therapeutic effect was observed in the DA-8010 5 mg group at 4 and 8 weeks compared with that in the placebo group. For most of the other efficacy variables, numerically noticeable improvements in OAB symptoms were observed in both the DA-8010 2.5 mg and 5 mg groups compared with the placebo group, although the difference did not reach statistical significance.
Similar to other clinical trials for OAB, we observed nonnegligible therapeutic effects in the placebo group. Considering the pathophysiology of OAB involving psychological aspects, treatment of patients with only placebo may have some effect [14]. Writing the frequency-volume chart, visiting outpatient clinics periodically, and probable encouragement from health care providers might also affect the efficacy in the placebo group [15]. Nevertheless, significantly more patients in both the DA-8010 2.5 mg and DA-8010 5 mg groups were ‘willing to continue treatment’ compared to the placebo group.
As an active reference drug, we used solifenacin 5 mg, another selective M3 antagonist that also has a bladder affinity over the salivary gland as DA-8010, and shows higher levels of drug persistence and compliance than other antimuscarinic agents to date [10,16,17]. No statistically significant difference was found in the therapeutic efficacy of DA-8010 2.5 mg and DA-8010 5 mg compared with solifenacin 5 mg, except for one parameter; the ∆ voided volume per micturition was increased in all treatment groups (DA-8010 2.5 mg, DA-8010 5 mg, and solifenacin 5 mg). Paradoxically, at 12 weeks, the increment was smaller in the DA-8010 5 mg group than in the DA-8010 2.5 mg group, resulting in a statistically significant difference between the DA-8010 5 mg and solifenacin 5 mg groups. The cause of this clinical result opposite to the dose administered is uncertain. A low statistical power might be one of the probable reasons.
Regarding drug safety, both DA-8010 2.5 and 5 mg treatment did not result in serious ADRs or treatment discontinuation; all the ADRs were transient and well-tolerated as commonly observed for other anticholinergics, suggesting a balanced clinical efficacy versus side-effect profile. The postvoid residual urine volume with both 2.5 mg and 5 mg of DA-8010 did not increase significantly compared with the baseline throughout the study period. Therefore, both doses of DA-8010 are considered to have an effect in inhibiting unnecessary involuntary detrusor contraction during the storage phase, not inhibiting the detrusor contraction mediated by acetylcholine in the voiding phase. Thus, the 2.5-mg and 5-mg doses showed potential as effective and safe for OAB and are considered appropriate for subsequent phase 3 trials.
On the other hand, since this study is the first to evaluate the therapeutic efficacy of DA-8010 in OAB patients, the number of subjects was determined by extrapolating the results of several previous studies on other anticholinergics, focusing only on the primary efficacy variable. The reason for the presence of multiple variables showing the numerical efficacy of DA-8010 without statistical significance might be the low statistical power to detect significance. Therefore, the results of the secondary endpoints should be interpreted exploratory with caution. Subsequent large-scale phase 3 trials may provide more accurate information.
In conclusion, DA-8010 showed potential as a novel therapeutic drug for the treatment of OAB. Dose-dependent effectiveness in frequency volume chart parameters and patients’ subjective satisfaction were observed in the DA-8010 2.5 mg and 5 mg groups. No significant difference was found between DA-8010 2.5 mg or 5 mg and solifenacin 5 mg for most of the efficacy endpoints. The incidence of ADRs was higher in the DA-8010 5 mg group than in the DA-8010 2.5 mg group, but they were all mild to moderate, and both doses were deemed to be well tolerated compared with those in the solifenacin 5 mg group. Therefore, both 2.5 and 5 mg DA-8010 are suitable for a subsequent large-scale phase 3 trial.
SUPPLEMENTARY MATERIALS
Supplementary Tables 1-3 can be found via https://doi.org/10.5213/inj.2142382.191.
Supplementary Table 1.
Patients’ subjective bladder condition (full analysis set)
Supplementary Table 2.
‘Benefit-Satisfaction-Willingness to continue’ at the end of treatment (full analysis set)
Supplementary Table 3.
Severity of common adverse drug reactions (safety set)
Notes
Funding Support
This study was funded by Dong-A ST Co., Ltd. (Korea), which developed and manufactured DA-8010.
Research Ethics
This study was performed at 12 centers in South Korea. It was registered at ClinicalTrials.gov (NCT03566134) and was approved by an institutional review board at each of the 12 institutions.
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION STATEMENT
·Conceptualization: HSS, CYO, MC, HGK, JCK, KL, DGS, SYC, SJJ, JTS, HY, HSM, JHK
·Data curation: HSS, CYO, MC, HGK, JCK, KL, DGS, SYC, SJJ, JTS, HY, HSM, JHK
·Formal analysis: HSS, JHK
·Funding acquisition: JHK
·Methodology: HSS, CYO, MC, HGK, JCK, KL, DGS, SYC, SJJ, JTS, HY, HSM, JHK
·Project administration: HSS, CYO, MC, HGK, JCK, KL, DGS, SYC, SJJ, JTS, HY, HSM, JHK
·Visualization: HSS, JHK
·Writing-original draft: HSS
·Writing-review & editing: HSS, JHK