|
 |


- Search
| Int Neurourol J > Volume 27(1); 2023 > Article |
|
ABSTRACT
SUPPLEMENTARY MATERIAL
NOTES
REFERENCES
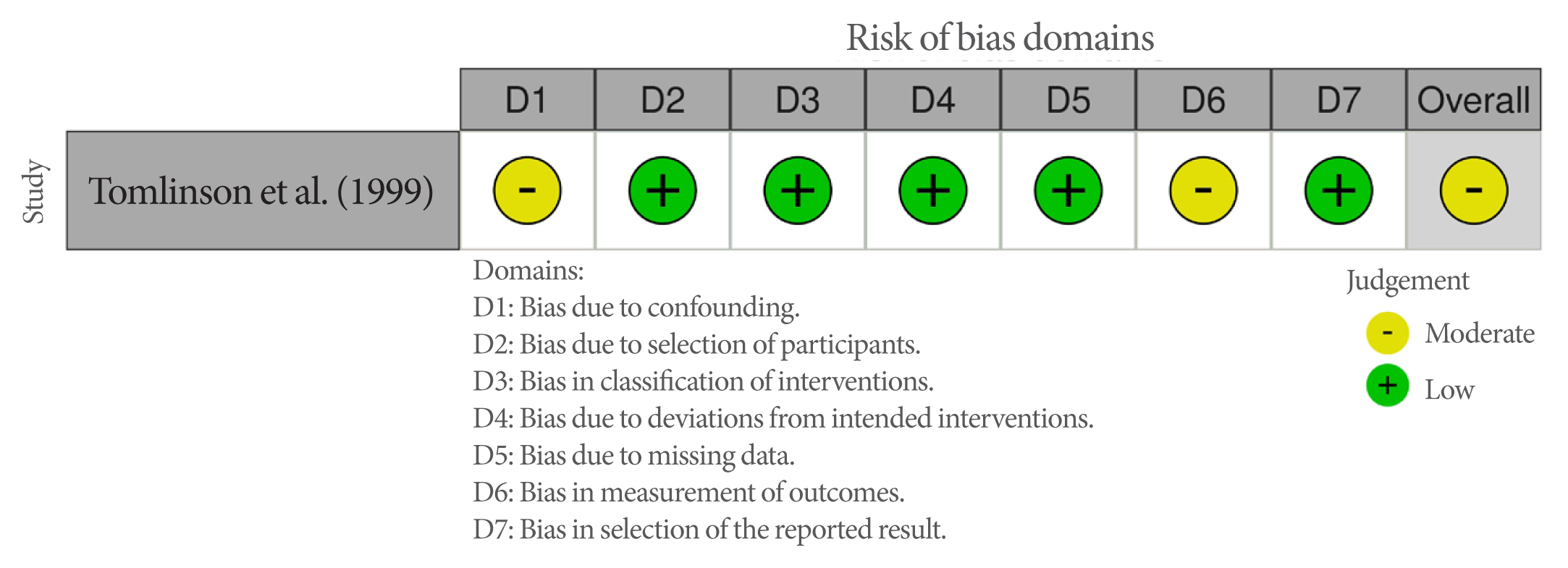
Fig. 1
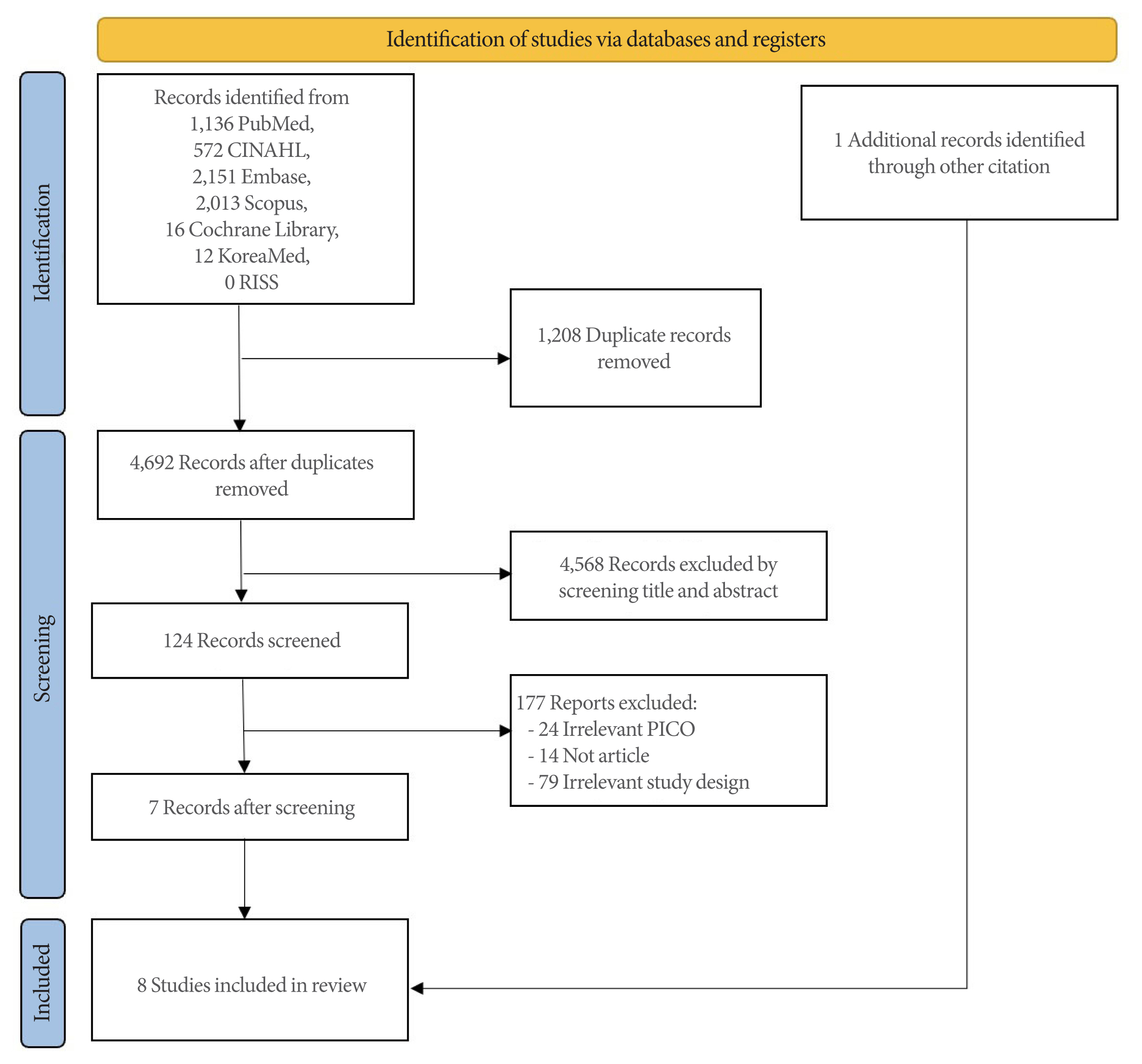
Fig. 2

Table 1
| Study | Country | Design | Inclusion/exclusion criteria | Settings | Age (yr) | No. of participants (N, at baseline→at outcome measurement) | Intervention | ||
|---|---|---|---|---|---|---|---|---|---|
| Experimental | Duration | Control | |||||||
| Bryant et al. (2002) [26] | Australia | RCT | Home |
E: 56±18a) C: 58±16a) |
Total: 95→74 E: 48→36 C: 47→38 |
Caffeine reduction education and bladder training | 4 Weeks | Bladder training only | |
| Dowd et al. (1996) [28] | USA | RCT | Home | 70.25 (52–89)b) |
Total: 58→32 Increase: 20→14 Decrease: 20→10 Maintain: 18→8 |
Increase fluid intake by 500 mL or decrease by 300 mL | 5 Weeks | Maintain current fluid intake | |
| Kincade et al. (2007) [30] | USA | RCT | Inclusion: Exclusion: | Home |
18–39 yr: 12.5% 40–64 yr: 62.5% 65+ yr: 25% |
Total: 224→184 E: 117→90 C: 107→94 |
Individualized counseling about caffeine consumption, fluid intake, simple PFMT | 3 Weeks | Waitlist |
| Kyoda et al. (2021) [31] | Japan | RCT | Inclusion: Exclusion: | Home |
E: 72.9±9.0a) C: 71.9±8.4a) |
Total: 100→78 E: 50→37 C: 50→41 |
CBT and FVC including caffeine restriction and decrease water intake in the evening. | 4 Weeks | FVC only |
| Spigt et al. (2006) [29] | Netherlands | RCT | Inclusion Exclusion | Home | 55–75 |
N=141→138 E: 70→69 C: 71→69 |
Extra intake of daily 1.5-L water | 6 Months | One tablespoon of placebo syrup daily |
| Swithinbank et al. (2005) [32] | UK | RCT | Inclusion: Exclusion: | Home | 54.8 (31–76)b) |
Total: 69 USI: 39 IDO: 30 |
3 Weeks of caffeine restriction. During week 3 and 4, increase decaffeinated fluids to 3L/day or decreased decaffeinated fluids to 750 mL/day |
4 Weeks | None |
| Tomlinson et al. (1999) [33] | USA | QES | Inclusion: Exclusion: | Home |
67.65±8.04a) E: 65.8±7.2a) |
Total: 94 E: 41 C: 53 |
Complete home visiting self-monitoring of the behavioral management for continence intervention and encouraged to decrease their caffeine intake and increase their fluid intake | 2–4 Weeks | Usual care |
| Wells et al. (2014) [27] | UK | RCT | Inclusion Exclusion | Home | 52 (27–79)b) |
Total: 14→11 Caffeinated: 7→5 Decaffeinated: 7→6 |
14-Day caffeinated drink, 14-day washout period, 14-day decaffeinated drink or 14-day decaffeinated drink, 14-day washout period, 14-day caffeinated drink | 6 Weeks | None |
RCT, randomized controlled trial; E, experimental; C, control; UI, urinary incontinence; UTI, urinary tract infection; MMSE, mini-mental state examination; PFMT, pelvic floor muscle training; IPSS, International Prostate Symptom Score; OAB, overactive bladder; OABSS, overactive bladder symptom score; LUTS, lower urinary tract symptom; CBT, cognitive behavior therapy; FVC, frequency volume chart; IDO, idiopathic detrusor overactivity; USI, urodynamic stress incontinence; QES, quasi-experimental study.
Table 2
| Study | Intervention | Time of measuring | Outcome variables (mean, at baseline → at outcome measurement) | Findings | ||||
|---|---|---|---|---|---|---|---|---|
| Urinary urgency | Urinary frequency | UI | Nocturia | QoL | ||||
| Bryant et al. (2002) [26] | Caffeine restriction less than 100 mg a day | Baseline, 4 wk after baseline |
Occasions of urgency per 24 hr E: 4.8→1.6 C: 4.6→3.2 (P=0.002) |
Voids per 24 hr E: 11.4→6.8 C: 11.2→7.9 (P=0.037) |
Occasions of leakage per 24 hr E: 2.8→1.2 C: 3.1→1.4 (P=0.219) |
The experimental group reduced their caffeine intake than the control group (P<0.001). Urgency and frequency outcomes were significantly improved. |
||
| Wells et al. (2014) [27] |
Group A: caffeinated drinks for 2 wk, washout period for 2 wk, decaffeinated drinks for 2 wk, Group B: decaffeinated drinks for 2 wk, washout period for 2 wk, caffeinated drinks for 2 wk. |
Baseline, after caffeinated period, decaffeinated period, washout period |
Urgency episodes/day CP: 9 (7.3–9.5)a) DP: 7 (6.2–8.3)a) |
Frequency episodes/day CP:9 (7.3–10.5) a) DP:7.3 (6.5–10.3)a) |
UI episodes/day CP: 0.3 (0–1.3)a) DP: 0 (0–0.8)a) |
ICIQ-OAB nocturia score CP: 2, DP: 1 P=0.031 |
Overall ICIQ-OABQoL score CP: 69, DP: 50 P=0.065 |
Significant reduction was found in urgency and frequency and total ICIQ-OAB score |
| Dowd et al. (1996) [28] | Increase fluid intake by an extra 500 mL or decrease it by 300 mL | Baseline, 4 times of weekly follow-up after baseline |
UI episodes Increase: 0.6→0.61→0.67→ 0.5→0.55 Decrease: 0.54→0.26→0.17→ 0.14→ 0.07 Maintain: 0.48→0.71→0.81→ 0.57→0.48 |
Quantitative results were not significant. UI episodes decreased the most in the decrease group. |
||||
| Spigt et al. (2006) [29] | Drinking daily extra 1.5-L water | Baseline, 6 mo after baseline |
Irritative symptoms (IPSS items 2,4,7) E: 6.0→5.8 C: 5.7→4.4 (effect size: 1.3; P<0.001) |
IPSS QoL E: 2.6→2.3 C: 2.9→2.5 (effect size: 0.1; P=0.06) |
Water consumption in the intervention group increased by 359ml per day than in the control group. Statistically significant effect was found for bladder pressure and bladder wall stress. Subjective parameters were improved in both groups but no statistically significant differences were found. |
|||
| Kincade et al. (2007) [30] | Individualized counseling about caffeine consumption, and amount and timing of fluid intake | Baseline, 3 wk after baseline |
Amount of urine loss E:27.6→19 C:45.8→47.4 (t=2.53, P=0.012) |
Incontinence impact questionnaire E: 125.4→99.3 C: 119.9→ 112.1 (t=−1.91, P=0.059) |
Self-monitoring on grams of urine loss was significant but that on episodes of urine loss was not. The intervention group lost lesser urine, average of 13.3 g, and had improved 26.1 points in QoL The intervention group reduced their caffeine intake but did not increase their fluid intake compared to the control group. |
|||
| Kyoda et al. (2021) [31] | Behavioral therapy includes avoiding the intake of caffeine from the evening and trying to drink less water from the evening | Baseline, 4 wk after baseline |
Total No. of voids/24 hr E: 9.2→8.9 C: 10.1→9.9 |
IPSS Q7 (night-time frequency) E: 3.6→2.6 (P<0.01) C: 3.7→3.1 (P<0.01) Episodes of nocturia/24 hr E: 2.3→1.9 C: 2.8→2.4 (P=0.039) |
N-QoL E: 25→20.8 (P=0.019) C: 22.3→22.3 (P=1.000) |
There were no significant differences in night-time frequency and QoL between two groups but after removing six patients with achievement of CBT<50%, night-time frequency was significantly lower in the intervention group. | ||
| Swithinbank et al. (2005) [32] | 3 Weeks of caffeine restriction. | Baseline, 3 times of weekly follow-up after baseline |
IDO group Increase: 5.2→ 5.4→ 7.6 Decrease: 5.2→ 5.4→ 4.3 |
Fluid increase USI: 7.2→7→8.3 IDO: 9→8.9→ 10.8 Fluid decrease1USI: 7.2→7→ 6.3 IDO: 9→8.9→ 7.7 |
No. of wetting episodes Fluid increase USI: 1.6→0.8→ 0.7 IDO: 0.9→0.6→1.1 Fluid decrease USI: 1.6→0.8→0.5 IDO: 0.9→0.6→0.5 (P=0.006) |
USI & IDO Decreasing fluid intake showed significant improvement compare with baseline week (P<0.003 each) |
In the IDO group, decreasing fluid intake significantly decreased voiding frequency, urgency, and wetting episodes while improved quality of life. In the USI group, there was significant decrease in wetting episodes when fluid intake was decreased. |
|
| Tomlinson et al. (1999) [33] | Baseline, 4 wk after baseline |
Voiding interval 2.41→2.38 Frequency episodes/day 9.96→10.08 |
Episodes of urine loss 2.6→1.68 (P=0.0744) |
The most frequently recommended intervention was caffeine restriction and increasing fluid intake. | ||||
UI, urinary incontinence; QoL, quality of life; E, experimental; C, control; CP, caffeinated period; DP, decaffeinated period; ICIQ-OAB (QoL), international consultation on incontinence-overactive bladder module (quality of life); IPSS, International Prostate Symptom Score; N-QoL, nocturia quality of life questionnaire; CBT, cognitive behavioral therapy; IDO, idiopathic detrusor overactivity; USI, urodynamic stress incontinence.
- TOOLS
-