INTRODUCTION
The artificial urinary sphincter (AUS) is the gold standard treatment for moderate-to-severe male stress urinary incontinence (SUI) according to the current European Association of Urology (EAU) guidelines [
1]. AUS implantation is associated with excellent continence outcomes and patient satisfaction; however, long-term AUS failure rate is high, with consequent frequent need for revision [
2]. In this regard, a pooled analysis showed an overall revision rate of 26% (range, 14.8%–44.8%) [
3], as well as a long-term study revealed that the 20-year survival rate without revision was 5% [
4].The main causes of AUS failure, with rates that vary considerably in the literature, are mechanical dysfunction, urethral atrophy, urethral erosion, and device infection [
2,
5].
The identification of risk factors for revision could improve patient counseling and selection, resulting in better outcomes of surgery. History of pelvic radiotherapy (RT), diabetes mellitus (DM), prior urethral strictures, anticoagulation, and doublecuff placement, were already found as independent predictors of AUS failure; however, the few studies available reported conflicting results [
6,
7].
The aim of the present study was to evaluate the preoperative and intraoperative risk factors for revision after AUS implantation in male patients with SUI.
MATERIALS AND METHODS
Study Design and Patient Population
We performed a retrospective analysis from a prospectively maintained single-center database in which patients with SUI undergoing AUS implantation were registered from September 2009 to August 2019. The Institutional Review Board approval was obtained. The study was conducted in accordance with the Declaration of Helsinki on ethical principles for medical research involving human subjects and each patient provided written informed consent to participate. Male patients over 18 years old, with moderate-to-severe SUI, undergoing AUS implantation were included in the study. Neurogenic SUI and previous AUS placement were exclusion criteria.
Patient Assessment and Variables Collected
Each patient underwent medical and surgical history, as well as an accurate physical examination. The variables collected at baseline were: age, body mass index, cause of SUI, degree of SUI, comorbidities (hypertension, coronary artery disease, peripheral artery disease, DM, obesity), vesicourethral anastomosis stenosis, previous pelvic RT, and previous incontinence surgery (e.g., fixed sling, urethral injection). SUI was diagnosed according to ICS definition [
8], whereas the degree of incontinence was evaluated with 24-hour pad weight test (mild SUI: <100 g/24 hr; moderate SUI 100-400 g/24 hr; severe SUI: >400 g/24 hr) [
9].
The variables recorded at the time of surgery were: time to AUS implantation (from the event that caused incontinence to surgery), operative time (from the first incision to the dressing), patient position, anesthesia, surgical approach, type and size of urethral cuff, pressure-regulating balloon (PRB) location, simultaneous inflatable penile prosthesis (IPP) implantation, use of drainage, and hospital stay.
All adverse events (AEs) that occurred during the study period were reported according to Clavien-Dindo classification [
10] and classified as intraoperative (from the beginning to the end of anesthesia), preactivation (from the end of surgery to the device activation), and postactivation (after the device activation) events. The revision was defined as any type of surgery performed following complications related to AUS implantation. In patients who underwent revision, the cause of revision, type of surgery (repair or explant), and time to revision were recorded. The revisions were classified as “explants” if all components of the AUS were removed, otherwise, they were categorized as “repairs.”
All subjects had a scheduled follow-up at 6 weeks, 3, 6, 12 months, and then yearly after each surgery (AUS implantation or revision) with the assessment of device operation, patient continence, and patient satisfaction. The patient continence was classified according to the number of pads used in a 24-hour period as complete continence (0 pad), social continence (0–1 pad), or incontinence (≥2 pads) [
11]. The patient satisfaction was evaluated whit the question “How satisfied are you with your current urinary continence?” to which it was possible to respond with “very satisfied,” “quite satisfied,” “neither satisfied nor dissatisfied,” “quite dissatisfied,” “very dissatisfied.”
Surgical Details
All AUS implantations were performed by 9 experienced surgeons ( ≥25 devices implanted [
12]). Each revision was performed by the same surgeon who initially implanted the AUS.
All surgeries were performed in the lithotomy position after the administration of a single prophylactic dose of antibiotic (cefazoline and gentamicin). Spinal or general anesthesia was administrated. All patients underwent placement of AMS 800 (Boston Scientific, Marlborough, MA, USA), consisting of a PRB, a urethral cuff, and a control pump.
Surgical approach, type and size of urethral cuff, PRB location, use of drainage, and dressing were at the discretion of each surgeon. Simultaneous placement of IPP (dual implantation) was performed in patients with erectile dysfunction refractory to medical treatment.
When revision was needed, the surgeons decided whether to perform a “repair” or an “explant” of AUS based on the specific clinical scenario.
Statistics
The categorical variables were described as frequencies and percentages, while for the quantitative variables mean with standard deviation (SD) or median with interquartile range (IQR) were used depending on the type of distribution determined with the normality tests.
Univariate analyzes were performed with chi-square or Fisher exact test. Multivariable analyzes were performed by means of a logistic regression and the effect sizes were reported as odds ratios (ORs) and 95% confidence intervals (CIs).
Competing risk analysis was used to investigate time to event data. The death of patients with working AUS acted as a competing event for AUS survival, therefore traditional Kaplan-Meier and proportional hazards regression techniques were not recommended. Regression model followed the strategy described by Fine-Gray [
13]. In this context, subhazard ratios (SHR) were estimated with their corresponding 95% CI.
Significance level was set at 0.05. Stata 16 (StataCorp., College Station, TX, USA) was the software used for the statistical analysis.
DISCUSSION
Despite the excellent results in terms of urinary continence and patient satisfaction, the AUS failure rate reported in the literature is considerable, resulting in a high percentage of revisions and short revision-free survival of the devices over the long term [
2]. The causes of AUS failure can be classified into mechanical and nonmechanical. The mechanical failure can be due to the malfunction of any part of the device (urethral cuff, PRB, or control pump), while the device infection, urethral erosion, and urethral subcuff atrophy are the main nonmechanical causes [
3]. When a patient complains about the new onset of urinary incontinence or worsening of the degree of continence achieved with AUS implantation, the aforementioned complications should be suspected and, after proper investigations, a revision surgery should be scheduled. The type of revision needed (e.g., repair of components, repositioning, explant) should be based on the cause of the AUS failure, taking into account the specific clinical case [
5].
In our cohort, a revision was performed in 22 patients (31.4%) after a median time of 26.5 months from the AUS implantation. The mechanical dysfunctions were responsible for 50% of all revisions, while the most frequent nonmechanical cause was the urethral erosion (27.3%). The earlier and later complications requiring a new surgery were device infection (12.5 months) and urethral atrophy (47.0 months), respectively. Despite the high percentage of subjects undergoing revision during the study period, the majority of patients at the last follow-up visit had a working device (85.7%), declaring complete or social continence (98.3%) and satisfaction with the state of continence achieved (89.9%).
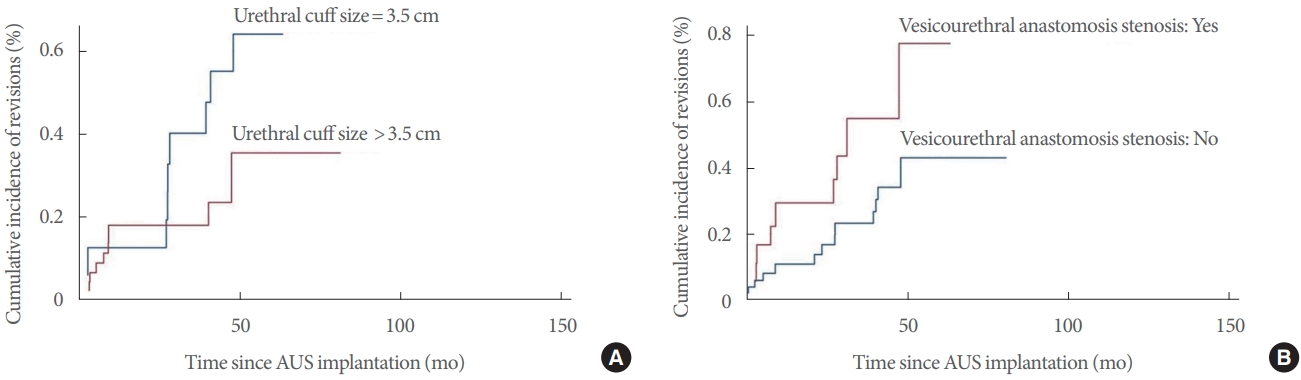
The multivariable analysis showed that vesicourethral anastomosis stenosis, urethral cuff size of 3.5 cm, and dual implantation were independent predictors for revision. However, the competing risk analysis revealed that vesicourethral anastomosis stenosis and urethral cuff size of 3.5 cm, but not dual implantation, predicted a lower survival of the AUS. All other factors investigated, including previous pelvic RT, were not associated with a significant risk of revision. Our results confirm the findings of some published studies; on the other hand, they contradict the results of other previously published research.
Kretschmer et al. [
7] found a significantly lower revisions rate in patients who received a 3.5-cm cuff compared to those receiving a 4.0- to 4.5-cm cuff (P=0.026), however, this parameter was investigated by the authors only with univariate analysis. Brant et al. [
14] demonstrated with a multivariable analysis that men with 3.5-cm cuff had a significantly higher rate of explant. Simhan et al. [
15] showed that patients with a history of RT who underwent implantation of a 3.5-cm cuff experienced a significantly increased risk of cuff erosion (21% vs. 4%, P = 0.01). It is important to underline that even in our cohort there was a high percentage of revision in subjects with previous RT who received a 3.5-cm cuff. In particular, we found 71.4%, 50.0%, 41.7%, and 16.7% of revisions in subgroups with 3.5-cm cuff plus RT, 3.5-cm cuff without RT, >3.5-cm cuff plus RT, and >3.5-cm cuff without RT, respectively. The higher risk of complications associated with a 3.5-cm cuff could be consequent to its use especially in patients with spongiosal atrophy, while the tissue alterations induced by RT could act as a facilitating factor [
15].
Hüsch et al. [
6] found that prior urethral stricture was associated with a higher risk of explant in the univariate analysis and was identified as an independent predictor for urethral erosion. Other studies confirmed the significant association between prior urethral strictures and AUS failure [
6,
7], however, not specifying the etiology of stricture. On the contrary, Linder et al. [
16] did not find in the univariate analysis a higher risk of revision in patients with prior vesicourethral anastomosis stenosis. Fibrosis and impaired vascularization at the stricture site could be the causes of the higher incidence of complications in this group of patients [
17].
Ravier et al. [
18] showed that infection (P=0.018) and explant (P=0.016) rates, but not erosion rate (P=0.13), were significantly higher in patients with prior RT. Other studies found that previous RT was an independent predictor for AUS failure [
6,
7]. Pelvic RT induces the obliteration of microvessels with fibrosis and atrophy of the tissues due to chronic hypoperfusion; this could be the pathophysiological substrate at the basis of the higher risk of complications after AUS placement [
18]. Although we did not find a significant association between previous pelvic RT and risk of revision after AUS implantation, this could derive from the small sample size. However, it is essential to emphasize that other studies showed results comparable to ours regarding RT. Léon et al. [
4] found that prior RT was not significant risk factors for revision in a retrospective study with a median follow-up of 15 years on 57 consecutive patients. Linder et al. [
16] described the results of a retrospective research with a median follow-up of 4.1 years, involving 1,802 subjects. The authors found that pelvic RT was associated with an increased risk of repeat surgery (hazard ratio, 1.34; P=0.02) on univariate but not multivariable analysis.
Boysen et al. [
19] concluded that dual AUS/IPP implantation did not adversely affect perioperative complications or device survival relative to the placement of either device alone. Yafi et al. [
5] in a recent review confirmed that most studies reported comparable complication rates with dual- and single-device implantations. Some papers showed a higher risk of complications with additional surgical procedures during AUS placement without specifying the type of procedure [
6]. Dual implantation may be related to a more distal dissection of the bulbar urethra, which could lead to a higher risk of urethral atrophy and erosion; moreover, the simultaneous presence of the components of the 2 devices could increase the risk of AUS malfunction. Our limited sample size could be the cause of the discrepancy between multivariable and competing risk analyzes regarding dual implantation.
To the best of our knowledge, this is the study specifically evaluating risk factors for revision after AUS implantation with the longest follow-up available in the literature. The involvement of multiple surgeons and the small percentage of patients lost during the follow-up are further significant strengths of our research.
Our results should be read and interpreted considering the limitations of the study. The main limitation is the retrospective design with the consequent bias that may arise from it. Other significant weaknesses are the relatively small sample size, the single-center design, and the presence of some missing data. The latest patients included, although a small minority, had a shorter follow-up than the median time to revision and this may have prevented some events from being recorded. Finally, we did not investigate the association between individual complications and risk factors.
In conclusion, the vesicourethral anastomosis stenosis, urethral cuff size of 3.5 cm, and dual implantation are independent predictors for revision after AUS implantation. However, at competing risk analysis, only the vesicourethral anastomosis stenosis and urethral cuff size of 3.5 cm predict a lower survival of the AUS. Further large prospective studies are needed to confirm our findings.