|
 |


- Search
| Int Neurourol J > Volume 28(2); 2024 > Article |
|
ABSTRACT
Purpose
This study was performed to assess the risk factors for artificial urinary sphincter (AUS) explantation in a large multicenter cohort.
Methods
We retrospectively reviewed the medical records for all 1,233 implantations of the AMS-800 AUS device in male nonneurological patients from 2005 to 2020 across 13 French centers. Patients with neurological conditions were excluded from the study. To identify factors associated with explantation-free survival, survival analysis was performed. Explantation was defined as the complete removal of the device, whereas revision referred to the replacement of the device or its components.
Results
The study included 1,107 patients, of whom 281 underwent AUS explantation. The median survival without explantation was 83 months. The leading causes of explantation were infection and erosion. Univariate analysis revealed several significant risk factors for explantation: age above 75 years (34.6% in the explanted group vs. 25.8% in the nonexplanted group, P=0.007), history of radiotherapy (43.5% vs. 31.3%, P=0.001), and anticoagulant use (15% vs. 8.6%, P<0.001). In logistic regression analysis, the only significant risk factor was previous radiotherapy (odds ratio [OR], 2.05; P<0.05). Cox proportional hazards analysis revealed 2 factors associated with earlier explantation: transcorporal cuff implantation (hazard ratio [HR], 2.67; P=0.01) and the annual caseload of the center (HR, 1.08; P=0.02). When specifically examining explantation due to erosion, radiotherapy was the sole factor significantly associated with the risk of erosion (OR, 2.47; P<0.05) as well as earlier erosion (HR, 1.90; P=0.039).
Conclusions
In this series, conducted in a real-world setting across multiple centers with different volumes and levels of expertise, the median survival without AUS explantation was 83 months. This study confirms that radiotherapy represents the primary independent risk factor for AUS erosion in male nonneurological patients.
Prostate surgery can result in stress urinary incontinence (SUI). The reported incidence of incontinence following prostate surgery varies widely; rates after radical prostatectomy range from 6.3% to 65%, while those following endoscopic prostate surgery are substantially lower, at under 3% [1, 2]. SUI greatly impacts patient quality of life and has considerable economic implications [3].
For the past few decades, the artificial urinary sphincter (AUS) has been the gold-standard surgical treatment for male SUI [4]. The primary strengths of the AUS lie in its efficacy and reliability; however, it is also associated with substantial morbidity, including a relatively high rate of infection, erosion, and the need for explantation or revision due to mechanical and non-mechanical failures. Despite this, the prevalence and risk factors for AUS explantation have rarely been explored using large multicenter datasets. The examination of more data is crucial to improve preoperative counseling, particularly for patients who may be at elevated risk; these include elderly individuals with multiple comorbidities as well as those with a history of radiation, incontinence surgery, or complex urethral surgery [5]. Indeed, multiple studies have explored risk factors for AUS failure [6, 7] to improve the efficacy and safety of this device. However, the results of these studies have been contradictory, possibly due to the heterogeneity of endpoints and small sample sizes [8]. The present study was performed to assess the prevalence of AUS explantation in men and to identify risk factors for explantation within a large multicenter cohort.
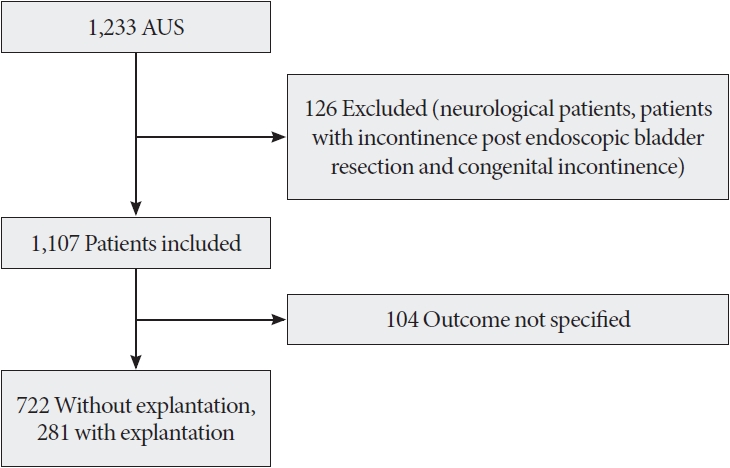
We retrospectively reviewed the charts of all male patients who underwent AUS implantation between 2005 and 2020 across 13 French urology departments. The inclusion criteria were men over 18 years old with urinary incontinence following prostate surgery or pelvic radiotherapy. In all cases, the AMS-800 (American Medical Systems, Inc., Minnetonka, MN, USA) AUS device was used. We excluded neurological patients and those with congenital incontinence.
Each center collected data in a computerized database. These data encompassed preoperative patient characteristics, including antiplatelet and anticoagulant intake, age, American Society of Anesthesiologists (ASA) physical status classification score, the widely used Charlson score, based on age and comorbidities, to represent patients’ general condition, and previous anti-incontinence surgical procedures; incontinence features, namely the severity of incontinence and any coexistent lower urinary tract symptoms; and biological data, comprising serum creatinine levels and preoperative urine culture results. The severity of incontinence was evaluated based on the number of pads used per day and self-administered questionnaires, specifically the International Consultation on Incontinence Questionnaire and the Urinary Symptom Profile.
The following surgical data were also collected: cuff location, surgical approach (perineal vs. penoscrotal), cuff size, length of hospitalization, postoperative complications, and functional outcomes. Postoperative complications were documented and classified according to the Clavien-Dindo system.
Due to the retrospective and multicentric design of the study, preoperative and postoperative workups, as well as surgical techniques, were not standardized. In total, 103 surgeons from 13 institutions participated in the study. Most of the procedures were carried out by 24 experienced surgeons, while the remaining operations were generally conducted under the supervision of an experienced surgeon.
We documented whether each patient underwent device explantation during the follow-up period. Explantation was defined as the complete removal of the device without simultaneous replacement. The primary endpoint of the study was explantation-free survival. We also noted and classified the reasons for explantation, which included infection, erosion, or other causes such as pain, difficulty operating the pump, or patient preference. Erosion was defined as the protrusion of the cuff through the urethral wall, which typically necessitates the complete removal of the sphincter.
Statistical analyses were conducted using R (R Foundation for Statistical Computing, Vienna, Austria, 2008), with the significance level set at 5%. In the descriptive analysis, qualitative parameters were presented as numbers and percentages, while quantitative parameters were summarized using means and standard deviations.
The Fisher exact test was employed for the comparison of nominal variables, the Kruskal-Wallis test was utilized for ordinal variables, and the Student t-test was applied for quantitative continuous variables.
Survival analysis was performed using the nonparametric Kaplan-Meier method. Survival was assessed from the date of implantation surgery to the date of explantation or (for cases that did not require explantation) the last follow-up.
Variables significantly associated with explantation, as identified in univariate analysis based on a P-value of less than 0.25, were included in a multivariate Cox proportional hazards model. Additionally, multivariate logistic regression was performed.
Among 1,233 men who underwent AUS implantation, 1,107 patients were included in this study (Fig. 1). The characteristics of these patients are detailed in Table 1. The mean participant age was 70.5 years, and the mean preoperative number of pads used per day was 3.7. A total of 326 patients (29.4%) had a history of pelvic radiotherapy, while 177 patients (16%) had undergone previous anti-incontinence surgery, predominantly male sling procedures. The perineal approach was the primary surgical technique, employed in 61% of cases. In most patients, incontinence had developed after radical prostatectomy (86.4%).
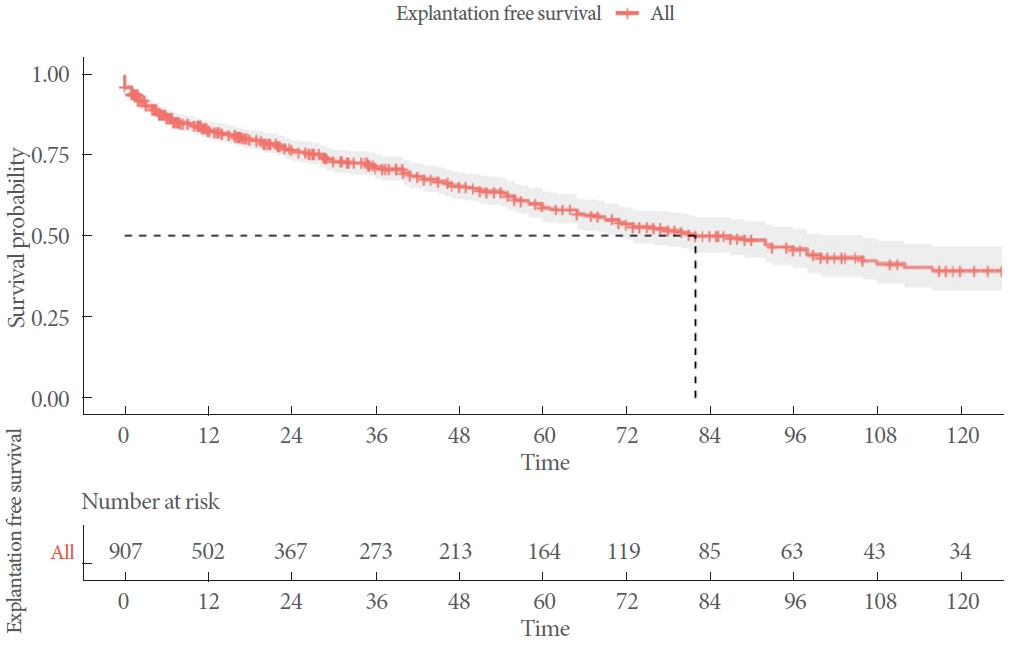
Of the 1,107 participants, 281 underwent AUS explantation (28%) (Table 2). The median duration of explantation-free survival was 83 months (Fig. 2). Infection and erosion were the leading causes of explantation, representing 94% of cases. Other causes were highly heterogeneous and included pain, inability to manipulate the pump, patient request, and radiation cystitis requiring urinary diversion, among others.
In the univariate analysis, several factors were significantly associated with explantation. These included age over 75 years (34.6% in the explanted group vs. 25.8% in the nonexplanted group, P<0.007), high preoperative 24-hour pad weight (P=0.015), low annual number of procedures performed at the center (P=0.047), high ASA physical status classification score (P= 0.019), history of pelvic radiotherapy (43.5% vs. 31.3%, P=0.001), and anticoagulant use (15% vs. 8.6%, P<0.001). The patient’s history of anti-incontinence procedures and cuff size were not significantly associated with the risk of explantation.
In the multivariate logistic regression analysis (Fig. 3), radiotherapy remained significantly associated with an increased risk of explantation (odds ratio [OR], 2.05; P<0.05). Cuff size, surgical approach, and cuff location displayed no significant associations with explantation risk.
In the multivariate Cox analysis (Fig. 3), 2 risk factors were identified as being associated with earlier explantation: transcorporal cuff position (hazard ratio [HR], 2.67; P=0.01) and higher annual caseload at the center (HR, 1.08; P=0.02).
More specifically, we aimed to identify risk factors associated with explantation due to erosion. In the multivariate logistic regression analysis (Fig. 4), radiotherapy was the only factor significantly associated with the risk of erosion (OR, 2.47; P<0.05). Cuff size, surgical approach, and cuff location exhibited no significant associations with erosion-free survival.
In the multivariate Cox analysis (Fig. 4), radiotherapy was the only factor significantly associated with shorter erosion-free survival (HR, 1.90; P=0.039).
For 50 years, the prevalence and risk factors associated with explantation have been topics of debate [9-11]. More robust data would assist in treatment decision-making and support patient counseling. To our knowledge, the present report describes the most extensive series focused on AUS explantation to date. The results suggest that in a real-world setting, device survival among nonneurogenic male patients was approximately 7 years. Our findings also confirmed that radiotherapy is the primary independent risk factor for AUS erosion and explantation.
Radiotherapy exerts both short- and long-term effects on tissues, potentially leading to fibrosis in the more vulnerable tissues, such as the urethra [12].
Our study corroborates the prior finding that radiation therapy to the urethra increases the risk of cuff erosion [13]. Similar results have been reported in other large cohorts [9, 14]. These findings align with those of Fuller et al. [15], who observed poorer survival outcomes in 150 patients with a history of radiation compared to 174 patients without such a history. In 2016, Hüsch et al. [16] identified radiotherapy as a risk factor for erosion in a cohort of 506 participants. Additionally, a single prospective study of 386 patients conducted in 2014 indicated that radiotherapy was a risk factor for explantation, with an OR of 4.872 [17].
Several possibilities may explain the heightened risk of erosion and explantation in patients who have undergone radiation therapy. Radiotherapy is known to cause long-term tissue effects, including fibrosis and microvascular damage. We posit that these changes to the urethral wall, which is in close contact with the AUS cuff, can predispose the tissue to erosion and subsequent explantation. Furthermore, radiation-induced bladder dysfunction—most notably gross hematuria resulting from radiation cystitis—may necessitate endoscopic procedures and catheterization, which can also increase the likelihood of erosion.
Another key accomplishment of the present study was the determination of median device survival without explantation, which was 83 months. This information is crucial for preoperative patient counseling. Previously, data on this important issue were relatively limited, primarily originating from small sample series or large tertiary referral centers [18]. The real-world data presented here are somewhat less favorable than those reported in earlier series. This discrepancy may be attributed to differences in the volume and expertise of the involved centers and surgeons, as well as the high proportion of patients who had undergone radiation [19]. The latter point may reflect a recent trend among survivors of prostate cancer.
Transcorporal cuff implantation is often employed in cases involving a fragile urethra to minimize the risk of urethral erosion and atrophy. Interestingly, this implantation method was associated with a shorter duration of explantation-free survival in the present study. While this result may seem surprising, recent investigations have suggested that transcorporal implantation may not extend the period of explantation-free survival [10, 20]. Our findings could reflect the notion that the transcorporal approach is indicative of a challenging implantation and poor tissue quality. The lack of significant association between surgical approach and erosion risk suggests that transcorporal implantation may also indicate the presence of other frailties that no study has yet clarified.
A 2015 study of 37 patients investigated the risk factors associated with complications arising from transcorporal cuffs [21]. The authors identified both systemic and urethral risk factors. At 35 months, they reported no cases of erosion or explantation in patients with 0 or 1 urethral risk factors. However, among those with 2 or more urethral risk factors, the rates of erosion and explantation were 64% and 52%, respectively.
Most previous studies have not distinguished between erosion and infection when assessing the cause of explantation. However, practices can differ among centers regarding erosion, which does not always necessitate explantation surgery.
To better understand the process, we aimed to identify the specific factors associated with erosion.
Concerning patient-related risk factors, several studies have identified diabetes, cardiovascular, and respiratory comorbidities as potential contributors to the risk of erosion and explantation [22-24]. In our study, the use of antiplatelet therapy, which serves as a proxy for cardiovascular comorbidities, was associated with a higher rate of explantation in univariate analysis. This association has also been observed in smaller cohorts [25]. Other indicators of general cardiovascular comorbidities, such as coronary artery disease, have been implicated as risk factors for AUS erosion and explantation [24]. The underlying pathophysiology remains unclear. Cardiovascular disease may lead to increased rates of explantation due to poor vascularization, resulting in suboptimal healing or urethral atrophy. Additionally, anticoagulants, which are often prescribed for cardiovascular conditions, could contribute to hematoma formation and further compromise the healing process. Alternatively, the higher explantation rate may simply be a consequence of the fragile, elderly, and multimorbid state of the patients. Accordingly, it is unsurprising that our univariate analysis indicated a relationship between age and explantation. Contrary to previous findings [25], we did not identify urethral strictures as a risk factor. Our results also showed no significant difference in explantation rates based on the surgical approach used. While some research suggests that the penoscrotal approach leads to a higher rate of explantation [26], this trend may disappear after adjusting for surgeon experience.
Our study has several limitations that warrant mention. The retrospective design is susceptible to information bias, particularly regarding surgeon experience. Furthermore, while utilizing a multicentric database can increase statistical power, it carries the drawback of added heterogeneity in practices and data collection methods. Additionally, we did not account for certain established risk factors for erosion and explantation, such as the use of indwelling catheters or cystoscopy, which are potential confounding variables. Finally, due to the absence of details regarding each surgeon’s history performing the procedure, we chose not to evaluate the influence of surgeon experience on the risk of explantation and erosion. This decision may represent a further limitation of our study.
In conclusion, in this series, conducted in a real-world setting across multiple centers with different volumes and levels of expertise, the median survival without AUS explantation was 83 months. This study confirms that radiotherapy represents the primary independent risk factor for AUS erosion in male nonneurological patients. These findings can aid in patient counseling and inform treatment decisions.
NOTES
Grant/Fund Support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Research Ethics
This study was approved by Institutional Review Board of Nice University Hospital (CNIL2216559).
Conflict of Interest
Daniel Chevallier: Expert consultant for Boston Scientific; Benoit Peyronnet: Consultant for Boston Scientific; Xavier Biardeau: Speaker for Boston Scientific. Except for that, no potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION STATEMENT
· Conceptualization: AP, PLecoanet, BP, IB
· Formal analysis: AP, PLecoanet, BP, IB
· Methodology: AP, PLecoanet, BP, IB
· Project administration: BP
· Writing - original draft: AP
· Writing - review & editing: PLecoanet, CM, VG, BP, TT, CS, TB, XG, FB, XB, FB, DR, MEA, DC, TC, GC, JNC, HD, HM, NH, JFH, PLeon, BP, IB
REFERENCES
1. Tutolo M, Cornu J, Bauer RM, Ahyai S, Bozzini G, Heesakkers J, et al. Efficacy and safety of artificial urinary sphincter (AUS): results of a large multi‐institutional cohort of patients with mid‐term follow‐up. Neurourol Urodyn 2019;38:710-8. PMID: 30575997



2. MacDonald S, Colaco M, Terlecki R. Waves of change: national trends in surgical management of male stress incontinence. Urology 2017;108:175-9. PMID: 28709851


3. Sandhu JS. Treatment options for male stress urinary incontinence. Nat Rev Urol 2010;7:222-8. PMID: 20383187



4. Scott FB, Bradley WE, Timm GW. Treatment of urinary incontinence by an implantable prosthetic urinary sphincter. 1974. J Urol 2002;167(2 Pt 2):1125-9 discussion 1130. PMID: 11905886

5. Farag F, van der Doelen M, van Breda J, D’Hauwers K, Heesakkers J. Decline in artificial urinary sphincter survival in modern practicedo we treat a different patient? Neurourol Urodyn 2017;36:1350-5. PMID: 27580297



6. Bentellis I, El-Akri M, Cornu JN, Brierre T, Cousin T, Gaillard V, et al. Prevalence and risk factors of artificial urinary sphincter revision in nonneurological male patients. J Urol 2021;206:1248-57. PMID: 34184925


7. Dupuis HGA, Bentellis I, El-Akri M, Brierre T, Cousin T, Gaillard V, et al. Early efficacy and safety outcomes of artificial urinary sphincter for stress urinary incontinence following radical prostatectomy or benign prostatic obstruction surgery: results of a large multicentric study. Eur Urol Focus 2022;8:1053-9. PMID: 34548254


8. Van der Aa F, Drake MJ, Kasyan GR, Petrolekas A, Cornu JN. The artificial urinary sphincter after a quarter of a century: a critical systematic review of its use in male non-neurogenic incontinence. Eur Urol 2013;63:681-9. PMID: 23219375


9. Moser DC, Kaufman MR, Milam DF, Johnsen NV, Cleves MA, Broghammer JA, et al. Impact of radiation and transcorporeal artificial sphincter placement in patients with prior urethral cuff erosion: results from a retrospective multicenter analysis. J Urol 2018;200:1338-43. PMID: 30563652


10. Ortiz NM, Wolfe AR, Baumgarten AS, Ward EE, VanDyke ME, Hudak SJ, et al. Artificial urinary sphincter cuff erosion heat map shows similar anatomical characteristics for transcorporal and standard approach. J Urol 2020;204:1027-32. PMID: 32459559


11. Guralnick ML, Miller E, Toh KL, Webster GD. Transcorporal artificial urinary sphincter cuff placement in cases requiring revision for erosion and urethral atrophy. J Urol 2002;167:2075-8 discussion 2079. PMID: 11956443


12. Hird AE, Radomski SB. Artificial urinary sphincter erosion after radical prostatectomy in patients treated with and without radiation. Can Urol Assoc J 2015;9(5-6):354. PMID: 26225177




13. Mamane J, Sanchez S, Lellouch AG, Gaillard V, Poussot B, Tricard T, et al. Impact of radiation therapy on artificial urinary sphincter implantation in male patients: a multicenter study. Neurourol Urodyn 2022;41:332-9. PMID: 34816473



14. Bates AS, Martin RM, Terry TR. Complications following artificial urinary sphincter placement after radical prostatectomy and radiotherapy: a meta-analysis. BJU Int 2015;116:623-33. PMID: 25601072


15. Fuller TW, Ballon-Landa E, Gallo K, Smith TG, Ajay D, Westney OL, et al. Outcomes and risk factors of revision and replacement artificial urinary sphincter implantation in radiated and nonradiated cases. J Urol 2020;204:110-4. PMID: 31951498


16. Hüsch T, Kretschmer A, Thomsen F, Kronlachner D, Kurosch M, Obaje A, et al. Debates on Male Incontinence (DOMINO)-Project. Risk factors for failure of male slings and artificial urinary sphincters: results from a large middle European Cohort Study. Urol Int 2017;99:14-21. PMID: 27598774



17. Brant WO, Erickson BA, Elliott SP, Powell C, Alsikafi N, McClung C, et al. Risk factors for erosion of artificial urinary sphincters: a multicenter prospective study. Urology 2014;84:934-9. PMID: 25109562



18. Agarwal D, Linder B, Elliott D. Artificial urinary sphincter urethral erosions: temporal patterns, management, and incidence of preventable erosions. Indian J Urol 2017;33:26-9. PMID: 28197026



19. Halpern JA, Sedrakyan A, Hsu W, Mao J, Daskivich TJ, Nguyen PL, et al. Use, complications, and costs of stereotactic body radiotherapy for localized prostate cancer. Cancer 2016;122:2496-504. PMID: 27224858



20. El-Akri M, Bentellis I, Tricard T, Brierre T, Cousin T, Dupuis H, et al. Transcorporal vs. bulbar artificial urinary sphincter implantation in male patients with fragile urethra. World J Urol 2021;39:4449-57. PMID: 34272596



21. Mock S, Dmochowski RR, Brown ET, Reynolds WS, Kaufman MR, Milam DF. The impact of urethral risk factors on transcorporeal artificial urinary sphincter erosion rates and device survival. J Urol 2015;194:1692-6. PMID: 26141851


22. Medendorp AR, Anger JT, Jin C, Amin KA, Hampson LA, Lee UJ, et al. The impact of frailty on artificial urinary sphincter placement and removal procedures. Urology 2019;129:210-6. PMID: 31005655



23. Radomski SB, Ruzhynsky V, Wallis CJD, Herschorn S. Complications and interventions in patients with an artificial urinary sphincter: long-term results. J Urol 2018;200:1093-8. PMID: 29940253


24. Raj GV, Peterson AC, Webster GD. Outcomes following erosions of the artificial urinary sphincter. J Urol 2006;175:2186-90 discussion 2190. PMID: 16697836


25. Kretschmer A, Buchner A, Grabbert M, Stief CG, Pavlicek M, Bauer RM. Risk factors for artificial urinary sphincter failure. World J Urol 2016;34:595-602. PMID: 26253655



26. Kretschmer A, Hüsch T, Thomsen F, Kronlachner D, Obaje A, Anding R, et al. Complications and short-term explantation rate following artificial urinary sphincter implantation: results from a large middle European multi-institutional case series. Urol Int 2016;97:205-11. PMID: 27310718



Fig. 3.
Risk factors for explantation, based on logistic regression analysis (above) and Cox proportional hazards analysis (below). AIC, Akaike information criterion. *P<0.05. **P<0.01.
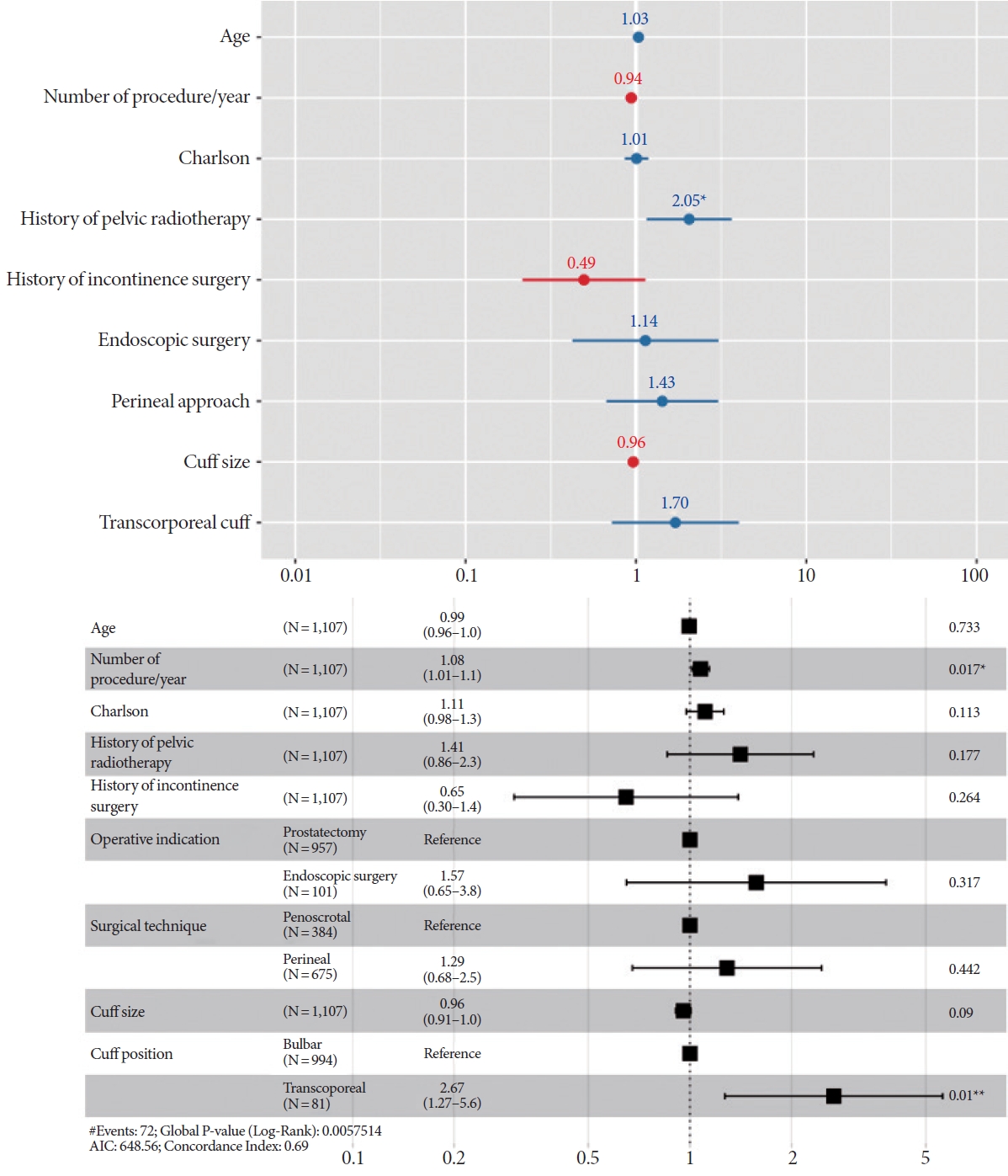
Fig. 4.
Risk factors for explantation due to erosion, based on logistic regression analysis (above) and Cox proportional hazards analysis (below). AIC, Akaike information criterion. *P<0.05. **P<0.01.
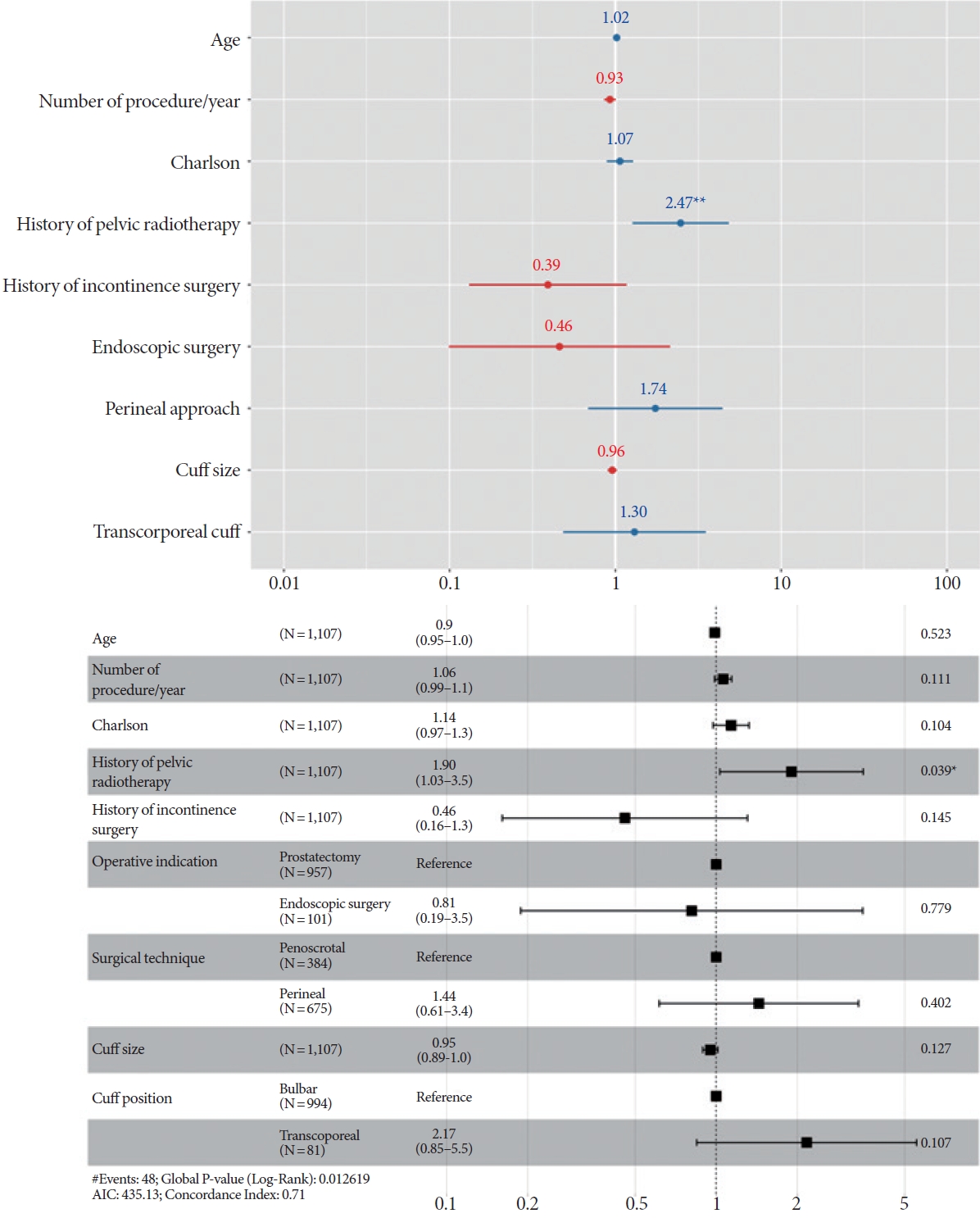
Table 1.
Cohort characteristics (n=1,107)
Table 2.
Risk factors associated with explantation, based on univariate analysis