Treadmill Running Improves Spatial Learning Memory Through Inactivation of Nuclear Factor Kappa B/Mitogen-Activated Protein Kinase Signaling Pathway in Amyloid-β-Induced Alzheimer Disease Rats
Article information
Abstract
Purpose
Exercise is known to reduce proinflammatory cytokines production and apoptosis. We investigated the effect of treadmill running on spatial learning memory in terms of activation of nuclear factor kappa B (NF-κB) and mitogen-activated protein kinase (MAPK) signaling pathway in Alzheimer disease (AD) rats. We also evaluated the effect of treadmill running on proinflammatory cytokine production and apoptosis.
Methods
Using the stereotaxic frame, amyloid-β (Aβ) was injected into the lateral ventricle of the brain. The rats belong to treadmill running groups were forced to run on a motorized treadmill for 30 minutes per a day during 4 weeks, starting 3 days after Aβ injection. Morris water maze task was done for the determination of spatial learning memory. Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay, immunohistochemistry for cleaved caspase-3, and western blot for NF-κB, inhibitory protein of NF-κB (IκB), MAPK signaling pathway, tumor necrosis factor (TNF)-α, interleukin (IL)-1β were done.
Results
Induction of AD increased proinflammatory cytokine secretion by activating the NF-κB/MAPK signaling pathway. These changes induced apoptosis in the hippocampus and reduced spatial learning memory. In contrast, treadmill running inactivated the NF-κB/MAPK signaling pathway and suppressed proinflammatory cytokine production. These changes inhibited apoptosis and improved spatial learning memory.
Conclusions
Current results showed that treadmill running promoted spatial learning memory through suppressing proinflammatory cytokine production and apoptosis via inactivation of NF-κB/MAPK signaling pathway. Treadmill exercise can be considered an effective intervention for symptom relieve of AD.
• HIGHLIGHTS
- Injection of Aβ impaired spatial learning memory and increased proinflammatory cytokines production and apoptosis.
- Treadmill running improved spatial learning memory via inactivation of NF-κB/MAPK signaling pathway.
- Treadmill running inhibited proinflammatory cytokine production and apoptosis.
INTRODUCTION
Alzheimer disease (AD) is a typical brain degenerative disorder that causes neuronal cell death. Although the cause of AD has not been accurately reported, it has been reported to be associated with oxidative stress due to amyloid-beta (Aβ) accumulation in the cerebrum, dysfunction of nerve fiber bundles due to over phosphorylation of tau protein, and increased inflammatory substances [1,2]. In particular, through various researches, it has been known that inflammation of neurons is closely associated with the onset of AD [3,4].
Inflammation is a critical reaction for the immune system to pathogen, injured cells, and irritations [5,6]. Brain inflammation causes neurodegeneration, including necrosis and apoptosis through tumor necrosis factor-α (TNF-α) and interleukin 1 beta (IL-1β) [6,7]. Aβ promotes inflammatory response of the brain, of which brain inflammation contributes to the onset of AD and Parkinson disease [8-10]. Aβ is a major feature of AD, where overproduction of Aβ leads to the formation of plaque [11].
Mitogen-activated protein kinase (MAPK) cascade is implicated in the cell growth, migration, proliferation, differentiation, and apoptosis. Long-term activation of MAPK is suggested to promote cell death [12]. The activity of nuclear factor kappa B (NF-κB) is maintained by the inhibitory protein of NF-κB (IκB). However, when IκB is phosphorylated by an external stimulus, proteasome-mediated degradation occurs. Active NF- κB induces the expression of inflammatory cytokines, which causes neuronal cell death in the brain [13].
Apoptosis is an important mechanism for maintaining normal cell or tissue homeostasis when inflammation increases, but excessive or inadequate apoptosis caused by external changes or stimuli leads to neurological disease [14]. The activation of various caspases is an important step in the apoptosis process, and caspase-3 activation plays as an executor of apoptosis [15]. The B-cell lymphoma 2 (Bcl-2) family is distinguished by function into anti-apoptotic proteins and proapoptotic proteins, which can determine the mitochondrial response to apoptosis stimulation. Among the Bcl-2 family, Bcl-2 suppresses apoptosis stage by blocking cytochrome c release from mitochondria, while the Bcl-2 related X protein (Bax) inhibits apoptosis process [16]. Terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) staining is an experimental method to find DNA fragmentation, a typical feature indicative of apoptosis [16].
Physical exercise has been suggested as one of the noninvasive treatments for the prevention or treatment of various brain diseases, and treadmill running reduces the expression of genes related to apoptosis [17,18]. Regular exercise relieved symptoms in Parkinson disease mice such as decreased coordination, activated MAPK cascade, and proinflammatory cytokine production [19]. In this current research, we studied the effect of treadmill running on spatial learning memory in AD-evoked rats in relation to the NF-κB/MAPK signaling pathway. We also evaluated the effect of treadmill running on proinflammatory cytokine production and apoptosis. For this study, Morris water maze test was done for the detection of spatial learning memory. TUNEL staining, immunohistochemistry for cleaved caspase-3, and western blot for NF-κB, IκB, MAPK, TNF-α, IL-1β were performed.
MATERIALS AND METHODS
Experimental Animals
This experiment was deliberated by the Animal Care and Use Committee and acquired the approval number (KSNU-2020-165). For the experimental procedure, 40-week-old (400±20 g) Sprague-Dawley male rats of were used. The animals were classified into control group, running group, AD-evoked group, and AD-evoked with running group.
AD Induction
The experimental animals were anesthetized by intraperitoneal injection of Zoletil 50 (10 mg/kg; Virbac Laboratories, Carros, Franc), then the animals were put on a stereotaxic instrument. After drilling a hole in the skull with a dental drill, a 26-G needle was inserted into the lateral ventricle on each side of the brain. Aβ (1-42) (Sigma Chemical Co., St. Louis, MO, USA) was diluted with saline to a concentration of 1 μg/μL. As described below [20], Aβ was incubated for 72 hours at 37°C to obtain an aggregated form of Aβ. The rats belong to AD-evoked groups were injected with 0.5 μL of Aβ with a Hamilton syringe, while the rats belong to control group and the rats belong to running group were injected with 0.5 μL of saline.
Exercise Protocol
The rats belong to the running groups were allowed running on an electric treadmill machine (Columbus Instruments, Columbus, OH, USA) for 30 minutes every day for 4 weeks starting from the 3 days after Aβ injection (Fig. 1). The run kept the slope at 0° and ran at a speed of 8 m/min without stopping.
Morris Water Maze Task
Morris water maze task was done to detect the spatial learning memory of the experimental rats, as described below [16,21]. Water mixed with black paint was placed in a round tank (diameter, 140 cm; height, 45 cm). The rats dived from their initial starting point and learned for 1 minute to reach their submerged destination (diameter, 15 cm; height, 35 cm). The round tank was divided into 4 quadrants, and the rats had learning opportunity. Learning took place 21 days after the start of the treadmill exercise, and measurement took place 5 days after the training. After all the lessons were completed, the time and distance traveled to the destination were measured. The measurement equipment was a video equipment (Smart ver. 2.5, Panlab, Barcelona, Spain).
Tissue Preparation
Following Morris water maze task was completed, the rats were intraperitoneally injected with Zoletil 50 (10 mg/kg; Virbac Laboratories). Through the heart, 50mM phosphate-buffered saline was injected and followed by infusion of 50mM phosphate buffer containing 4% paraformaldehyde. After filling the 50-mL tube with 4% paraformaldehyde, the extracted brains were immersed and left for a day for fixation.
TUNEL Assay
In order to find DNA fragmentation, TUNEL staining was done utilizing with the In Situ Cell Death Detection Kit (Roche, Mannheim, Germany), as explained below [13,16]. The sections were treated with ethanol-acetic acid (2:1), proteinase K (100 mg/mL), 3% H2O2, 0.5% Triton X-100, and TUNEL reaction mixture. After treating the sections with Converter-POD using 0.03% diaminobenzidine, Nissl staining was done for counterstaining, then placed on gelatin-coated slides.
Immunohistochemistry
Immunohistochemistry was done to find the cleaved caspase-3 in the hippocampus, as described below [13,16]. Tissues were blocked using horse serum for 1 hour, washed 3 times for 5 minutes, and incubated with rabbit anticleaved casepase-3 antibody (1:1,000; Cell Signaling Technology, Inc., Danvers, MS, USA) overnight. After one day, the tissues were reacted with the secondary antibody (1:200; Vector Laboratories, Burlingame, CA, USA) for 1 hour. The sections were treated with avidin- biotin-peroxidase complex (Vector Laboratories) for 1 hour and then incubated with a solution composing of 0.03% 3,3ʹ- diaminobenzidine and 0.03% hydrogen peroxide for 5 minutes.
Western Blot Analysis
Western blot analysis was done, as explained below [16,22]. The primary antibodies were treated during 24 hours for TNF-α (1:1,000; Santa Cruz Biotechnology, Santa Cruz, CA, USA), IL-1β (1:1,000; Santa Cruz Biotechnology), NF-κB (1:1,000; Abcam, Cambridge, UK), IκBα (1:1,000; Santa Cruz Biotechnology), phosphorylated IκBα (p-IκBα) (1:1,000; Santa Cruz Biotechnology), extracellular signal-regulated kinase (ERK) (1:1,000; Cell Signaling Technology), phosphorylated ERK (1:1,000; Cell Signaling Technology), c-Jun N-terminal kinases (JNK) (1:1,000; Cell Signaling Technology), phosphorylated JNK (1:1,000; Cell Signaling Technology), p38 (1:1,000; Cell Signaling Technology), and phosphorylated p38 (1:1,000; Cell Signaling Technology). The secondary antibodies were applied to the membrane, and then the bands were calculated by enhanced chemiluminescent kit.
Data Analysis
TUNEL-positive and cleaved caspase-3-positive cell number in the dentate gyrus of hippocampus were calculated by the optical microscope (Olympus, Tokyo, Japan). Western blot results were quantified by setting the control group as 1.00. For statistical processing, IBM SPSS Statistics ver. 25.0 (IBM Co., Armonk, NY, USA) was used and the results were compared by 1-way analysis of variance followed by Duncan posttest. All data were presented as the mean±standard error of the mean, and statistically significance was determined at P<0.05.
RESULTS
Spatial Learning Memory in Morris Water Maze Test
Data from the Morris water maze task were exhibited in Fig. 2. Induction of AD reduced spatial learning memory (P<0.05), and treadmill running ameliorated impairments of spatial learning memory in AD-evoked rats (P<0.05).
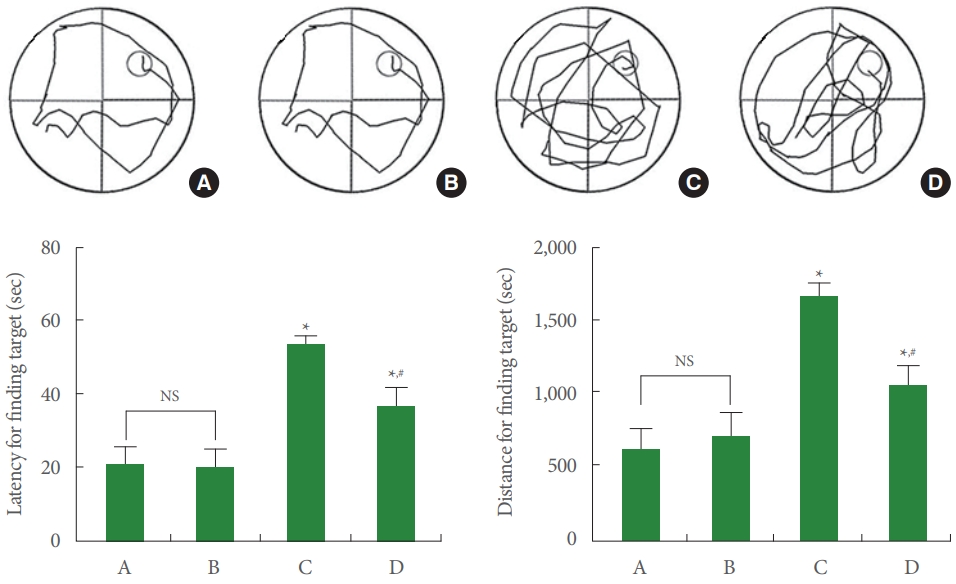
Spatial learning memory. Upper panel: representative swimming path. Lower left panel: latency for finding target. Lower right panel: distance for finding target. A, control group; B, running group; C, Alzheimer disease (AD)-evoked group; D, AD-evoked and running group. *P<0.05 compared to the control group. #P<0.05 compared to the AD-evoked group. NS, nonsignificant.
TNF-α and IL-1β Expression in Hippocampus
Data from the western blotting of TNF-α and IL-1β were exhibited in Fig. 3. Induction of AD increased TNF-α and IL-1β expression (P<0.05), and treadmill running inhibited TNF-α and IL-1β expression in AD-evoked rats (P<0.05).
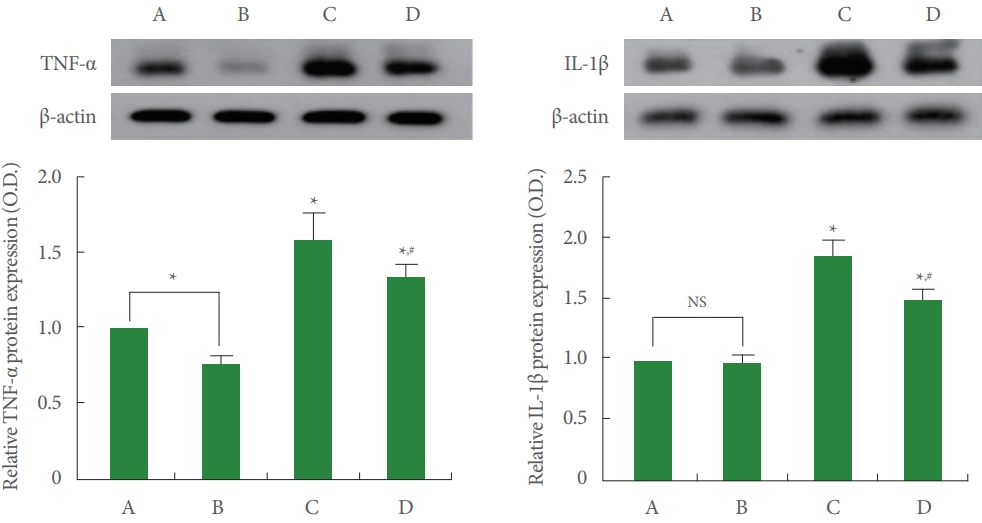
Tumor necrosis factor-alpha (TNF-α) and interleukin-1 beta (IL-1β) expression in the hippocampus. Left upper panel: representative expression of TNF-α. Left lower panel: relative expression of TNF-α. Right upper panel: representative expression of IL-1β. Right lower panel: relative expression of IL-1β. A, control group; B, running group; C, Alzheimer disease (AD)-evoked group; D, ADevoked and running group. *P<0.05 compared to the control group. #P<0.05 compared to the AD-evoked group. NS, nonsignificant.
NF-κB and IκB Expression in Hippocampus
Data from the western blotting of NF-κB and IκB expression were exhibited in Fig. 4. Induction of AD increased NF-κB expression and enhanced IκB phosphorylation (P<0.05). Treadmill running inhibited NF-κB expression and suppressed IκBα phosphorylation in AD-evoked rats (P<0.05).

Nuclear factor kappa B (NF-κB) and inhibitory protein of NF-κB (IκB) expression in the hippocampus. Left upper panel: representative expression of NF-κB. Left lower panel: relative expression of NF-κB. Right upper panel: representative expression of IκBα. Right lower panel: relative expression of IκBα. A, control group; B, running group; C, Alzheimer disease (AD)-evoked group; D, ADevoked and running group. *P<0.05 compared to the control group. #P<0.05 compared to the AD-evoked group. NS, nonsignificant.
MAPK Cascade Expression in Hippocampus
Data from the western blotting of MAPK cascade expression were exhibited in Fig. 5. Induction of AD increased phosphorylation of ERK, JNK, and p38 of the MAPK cascade (P<0.05), and treadmill running inhibited phosphorylation of ERK, JNK, and p38 in AD-evoked rats (P<0.05).

Mitogen-activated protein kinase (MAPK) expression in the hippocampus. Left upper panel: representative expression of expression of extracellular signal-regulated kinase (ERK). Left lower panel: relative expression of ERK. Middle upper panel: representative expression of c-Jun N-terminal kinase (JNK). Middle lower panel: relative expression of JNK. Right upper panel: representative expression of p-38. Right lower panel: relative expression of p-38. A, control group; B, running group; C, Alzheimer disease (AD)-evoked group; D, AD-evoked and running group. *P<0.05 compared to the control group. #P<0.05 compared to the AD-evoked group. NS, nonsignificant.
Apoptosis in Hippocampus
Data from the apoptosis were exhibited in Fig. 6. Induction of AD increased TUNEL-positive and cleaved caspase-3-positive cell number in the dentate gyrus of hippocampus (P<0.05). Treadmill running suppressed TUNEL-positive and cleaved caspase-3-positive cell number in AD-evoked rats (P<0.05). Induction of AD increased Bax expression and suppressed Bcl- 2 expression, consequently increasing the Bax versus Bcl-2 ratio (P<0.05). Treadmill running inhibited Bax expression and increased Bcl-2 expression, consequently decreasing the Bax versus Bcl-2 ratio in AD-evoked rats (P<0.05).
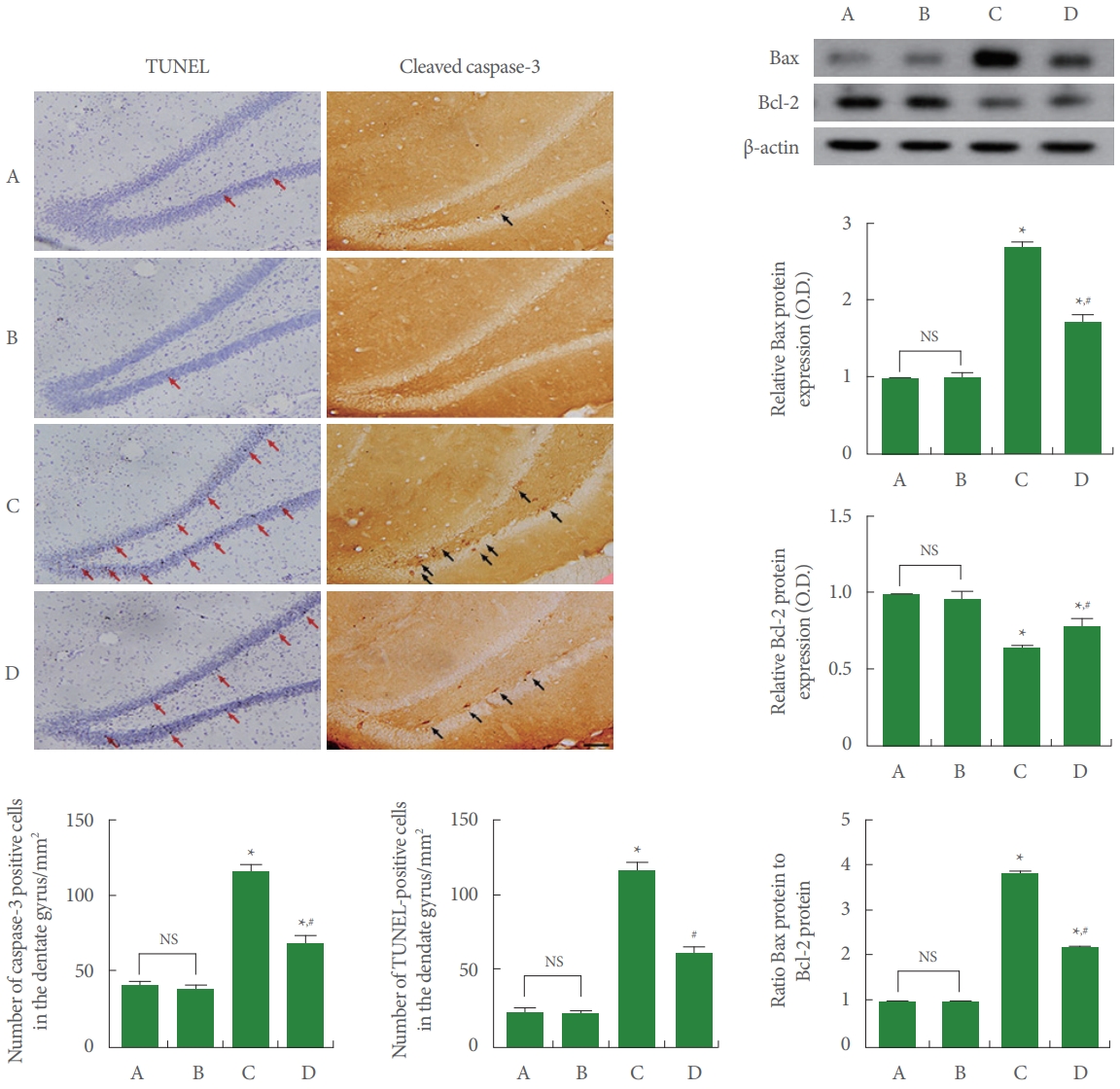
Apoptosis in the hippocampus. Left upper panel: photomicrographs of terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL)-positive cells (red arrows) and cleaved caspase-3-positive cells (black arrows) in the dentate gyrus of hippocampus. The scale bar represents 100 μm. Left lower panel: number of TUNEL-positive cells and cleaved caspase-3-positive cells. Right upper panel: representative expression of Bcl-2-associated X protein (Bax) and B-cell lymphoma-2 (Bcl-2) in the hippocampus. Right lower panel: expression of Bax, Bcl-2, and ratio of Bax to Bcl-2. A, control group; B, treadmill running group; C, Alzheimer disease (AD)-evoked group; D, AD-evoked and running group. *P<0.05 compared to the control group. #P<0.05 compared to the AD-evoked group. NS, nonsignificant.
DISCUSSION
The method of administering Aβ to experimental animals is a representative method of inducing AD. Accumulation of Aβ in the brain exerts devastating effects on the immune system [23]. Cells of the immune system release cytokines and chemokines that can influence the β-amyloid producing and/or aggregation [24]. Increased cytokine production is closely associated with decreased cognitive function [25]. Voluntary wheel running following brain inflammation inhibited proinflammatory cytokine production and apoptosis, then improved spatial learning memory [13]. Performing treadmill running improved spatial learning memory, short-term memory, and recognition memory [26-28]. In the current study, induction of AD was found to worsen the spatial learning memory in the Morris water maze task, whereas treadmill running improved AD-induced spatial learning memory.
The NF-κB/MAPK signaling pathway regulates inflammation through transcription of TNF-α and IL-1β [29]. The deposition of Aβ activates the MAPK cascade, including JNK, ERK, and p38, and then produces proinflammatory cytokines that are translocated to the nucleus in the NF-κB complex [30]. Tau hyperphosphorylation, Aβ toxicity, autophagy deregulation, and apoptosis are caused by activation of signaling pathways in the NF-κB and MAPK cascade [31]. Voluntary wheel running inactivated the signaling pathway of the NF-κB [13]. In the current study, induction of AD enhanced NF-κB expression and increased IκBα phosphorylation. Induction of AD also increased phosphorylation of JNK, ERK, and p38. The results mean that AD induction activated MAPK signaling pathway. Treadmill running suppressed MAPK expression and decreased IκBα phosphorylation. Treadmill running also suppressed phosphorylation of JNK, ERK, and p38. The results mean that treadmill running inactivated NF-κB/MAPK signaling pathway.
TNF-α and IL-1β production was increased during acute liver injury, and these proinflammatory cytokines exacerbated symptoms of acute liver injury [29]. Production of TNF-α and IL-6 in the hippocampus was suppressed by voluntary wheel running following brain inflammation [13]. In the current study, AD induction enhanced the production of TNF-α and IL-1β in the brain of AD-evoked rats. Treadmill running suppressed TNF-α and IL-1β production in AD-evoked rats.
In animal AD model, elevated Aβ caused apoptosis after injury or disease, and inhibition of caspases protected from apoptosis [32]. Apoptotic neuronal cell death and impaired spatial learning memory due to Aβ deposition in the hippocampus were observed in AD transgenic mice [33]. During acute liver injury, Bax expression was enhanced and Bcl-2 expression was suppressed, leading to increased apoptosis [29]. Voluntary wheel running inhibited the number of TUNEL-positive and caspase-3-positive cells in the hippocampus during brain inflammation [13]. Voluntary wheel running also suppressed Bax expression and increased Bcl-2 expression in the hippocampus after brain inflammation [13]. In socially isolated old rats, TUNEL- positive cell number and Bax expression were inhibited, and Bcl-2 expression was suppressed, indicating an accelerated apoptosis [16]. Meanwhile, swimming reduced TUNEL-positive cell number and Bax expression, and increased Bcl-2 expression, resulting in suppressed apoptosis [16]. In the current study, the number of TUNEL-positive and cleaved caspase- 3-positive cells, the expression of Bax, and the ratio of Bax to Bcl-2 were enhanced, while the expression of Bcl-2 was suppressed in AD-evoked rats. TUNEL-positive and cleaved caspase- 3-positive cell number, Bax expression, and Bax to Bcl-2 ratio were suppressed and Bcl-2 expression was increased by treadmill running in AD-evoked rats.
Current results show that treadmill running has an increasing impact on spatial learning memory through suppressing proinflammatory cytokine production and apoptosis via inactivation of the NF-κB/MAPK signaling pathway. Treadmill exercise can be considered as an effective intervention for alleviating AD symptoms.
Notes
Fund/Grant Support
This work was supported by the Ministry of Education of the Republic of Korea and the National Research Foundation of Korea (NRF-2018S1A5A2A01032722).
Research Ethics
This experiment was deliberated by the Animal Care and Use Committee and acquired the approval number (KSNU-2020-165).
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION STATEMENT
- Conceptualization: SHK
- Data curation: YJK, JYK
- Formal analysis: YJK, JYK
- Funding acquisition: YJS
- Methodology: YJK, JYK
- Project administration: YJS
- Visualization: YJS
- Writing-original draft: SHK
- Writing-review & editing: SHK

