INTRODUCTION
Stress urinary incontinence (SUI) is defined by the International Continence Society as involuntary leakage of urine on effort or exertion, such as during sneezing or coughing [
1]. In woman, SUI typically occurs after multiple child births and menopause; in men, SUI may occur after radical prostatectomy or, occasionally, after transurethral prostatectomy [
2]. Intrinsic sphincter deficiency (ISD) may also result from neurological lesions such as spinal cord injury, myelomeningocele, or spinal bifida, and due to complicated urethral rupture associated with pelvic fracture [
3].
SUI impairs the quality of life of the afflicted individual owing to the shame and embarrassment caused by incontinence. Conservative management includes the use of pads, external appliances for urine collection, penile clamps, behavioral modifications, and pelvic floor exercises [
4,
5]. Suburethral sling procedures are commonly performed to treat SUI in women; the reported success rate is approximately 90% at 3 years and 60% at 20 years [
6]. Surgical procedures for male SUI include periurethral injections of bulking agents, suburethral sling procedures, artificial urinary sphincter implantation, periurethral constrictors, and stem cell therapy [
7-
10]. Many patients with SUI require a surgical intervention for incontinence since nonsurgical modalities for postprostatectomy incontinence (PPI) offer limited and unsatisfactory improvement [
9,
11]. However, patients with mild urinary incontinence may not accept invasive surgical management. Injection therapy with urethral bulking agents or stem cell formulations have been developed as minimally invasive alternatives [
10]. Currently, there is no simple treatment modality to cure this distressing disorder in male incontinent patients.
Autologous platelet-rich plasma (PRP) is growing in popularity as a therapy to augment wound healing and hasten recovery after muscle and joint injuries and surgical repair [
12]. PRP is extremely rich in growth factors and cytokines that regulate tissue reconstruction; the use of PRP has been extensively studied in trauma patients and trauma experimental models [
13-
16]. The local application of autologous bone marrow-derived progenitor cells and PRP was shown to improve tissue regeneration [
15]. PRP is an easily produced and relatively inexpensive biological material. It is produced directly from the patient’s blood, and is thus superior to synthetic materials in terms of potential adverse effects such as foreign body reaction.
During wound healing, biologically active molecules are released from platelets. Platelets play an essential role in promoting tissue regeneration and wound healing [
12]. Recent studies have demonstrated the immunomodulatory effect of PRP on the inflammatory response [
15,
16]. Platelet-rich concentrates are used as a source of growth factors to improve the healing process. PRP can also eliminate neuropathic pain by the direct effect of some factors acting directly on neurons to promote axonal regeneration [
16]. Preliminary studies have also shown that repeated PRP injections into the suburothelium improved bladder pain and decreased frequency urgency episodes in patients with interstitial cystitis/bladder pain syndrome [
17,
18].
Owing to the potential regenerative effect of PRP, it seems rational to inject PRP into the urethral sphincter to increase the sphincter muscle bulk and urethral resistance. Because autologous PRP does not have antigenicity to the patient’s own tissue and has been proven safe for facilitating wound healing and in the treatment of interstitial cystitis [
15-
18], we performed a clinical trial to investigate the use of autologous PRP for treatment of SUI due to ISD of neurogenic or nonneurogenic etiology. Our results may provide clinical evidence for a novel therapeutic regimen for the treatment of ISD and SUI.
MATERIALS AND METHODS
A total of 35 patients with SUI with urodynamically proven ISD who did not respond to conventional treatments were enrolled in this study. All these patients had been treated with an oral medication (e.g., methylephedrine, imipramine, or duloxetine) or surgical treatment with a suburethal sling or artificial sphincter device, but treatment had failed for at least 1 year. The diagnosis of ISD was established based on the characteristic symptoms and video urodynamic findings. All patients had urinary incontinence and required pad protection. All patients were investigated thoroughly at enrollment and were excluded if they did not satisfy the study-selection criteria (
Supplementary material 1).
The severity of SUI was reported by the patient’s self-assessment using a 10-point visual analogue scale (VAS) score (0 indicating no incontinence and 10 indicating severe incontinence) (
Table 1). The SUI VAS system was validated in the first 10 patients; the Cronbach alpha for test-retest reliability was 0.929, confirming the reproducibility of the reported grading of SUI. Patients were requested to maintain a 3-day voiding diary prior to treatment to record the functional bladder capacity, urinary frequency, nocturia, and daily urinary leakage episodes. Videourodynamic studies were performed by standard procedures using a 6F dual channel catheter and an 8F rectal balloon catheter [
19]. Videourodynamic studies, abdominal leak point pressure (ALPP) measurements, and urethral pressure profilometry (UPP) were routinely performed at baseline and after PRP treatment. ALPP measurements were performed at full bladder to measure the smallest ALPP during cough and abdominal straining maneuvers. If there was no leak in response to coughing or the Valsalva maneuver, the highest abdominal pressure was used to represent the ALPP. UPP parameters included maximum urethral closing pressure (MUCP) and functional profile length (FPL). All descriptions and terminology in this report are in accordance with the recommendations of the International Continence Society [
1]. In this study, all patients were proven not to have urodynamic ISD or detrusor overactivity.
This study was approved by the Institutional Review Board (IRB) and Ethics Committee (IRB No. 107-231-A). All patients were informed about the study rationale and procedures; written informed consent was obtained prior to enrollment and treatment. Eligible patients were admitted for PRP treatment. All subjects received a urethral sphincter injection of 5 mL of PRP (extracted from 50 mL of the patient’s own whole blood) under intravenous general anesthesia in the operating room. The procedure was repeated every 1 month for a total of 4 treatments within 3 months. Patients were informed of the possible complications of urethral sphincter injection, such as hematuria, micturition pain, difficulty urinating, transient urinary retention, and urinary tract infection (UTI).
The procedure of preparing PRP was described in a previous report and is presented in
Supplement material 2 [
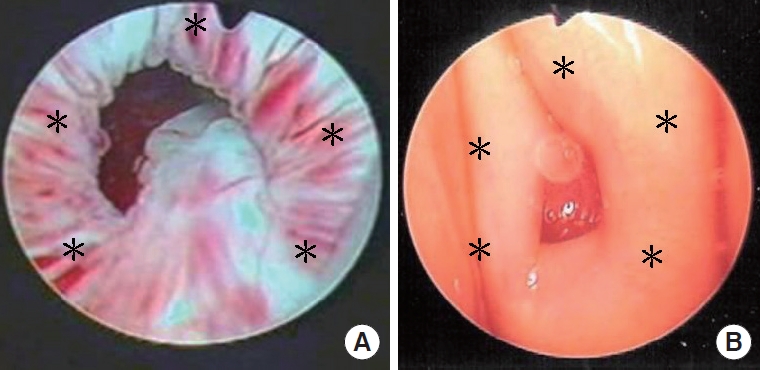
18]. In brief, a 50-mL whole blood specimen was withdrawn and subjected to centrifugation twice. The concentration of platelets in the PRP was 2.5–5 times that in the peripheral blood. Then, 5 mL of PRP in saline was sent to the operating room in a sterile test tube for urethral sphincter injection. The urethral sphincter PRP injections were administered at 5 sites via an injecting cystoscope for male patients and from the perineum around the urethral meatus in female patients (
Fig. 1). At each site, 1 mL of PRP was injected at each site. After the PRP injections, patients were followed up at the outpatient clinic every month, and at 3 and 6 months after the 4 PRP injection.
The treatment outcome was assessed by the Global Response Assessment (GRA). The treatment result was considered excellent (GRA=3) when patients reported complete dryness at the end of the study. A GRA score of 2 was considered indicative of moderate improvement. Excellent and moderately improved results were considered indicative of successful treatment. A GRA score of 1 or a reduction of 2 points in the VAS score was considered indicative of mild improvement. GRA improvement ≤1 was considered indicative of treatment failure.
The results of the voiding diary, urodynamic studies, and VAS scores were compared between baseline and the primary end-point at 6 months; urodynamic parameters were compared between baseline and after the PRP treatment. Data were compared between different patient groups and between baseline and post-PRP treatment. Continuous variables are expressed as mean±standard deviation, while categorical variables are presented as frequency (percentage). The paired t-test was used to analyze the data between baseline and posttreatment status, and the Wilcoxon rank-sum test was used for statistical comparisons of variables in the between-subgroup analysis. All statistical analyses were conducted using SPSS ver. 12 (SPSS Inc., Chicago, IL, USA). Two-sided P-values <0.05 were considered indicative of statistical significance.
RESULTS
In total, 35 patients with SUI were enrolled in this study. The mean age of patients was 68.7±12.0 years (range, 35–87 years); the median duration of SUI was 4 years (range, 1–10 years). The underlying diseases in the study population are listed in
Table 2. Neurogenic SUI was noted in 5 patients, while 30 patients had nonneurogenic SUI (21 with PPI, 6 with previous radical cystectomy for bladder carcinoma, and 3 with other etiologies of ISD).
Three months after the fourth PRP injection, 7 patients (20.0%) reported complete dryness, while moderate improvement was observed in 14 patients (40.0%); thus, the overall success rate was 60.0%. Among the 30 patients with nonneurogenic SUI, 28 patients (93.3%) had a GRA ≥1, while 20 patients (66.7%) had a GRA ≥2. However, only 1 out of the 5 patients (20%) with neurogenic SUI had a GRA ≥2 (P=0.044) (
Table 2). There was no significant difference between patients with successful treatment outcomes and those with failed treatment outcomes with respect to the baseline demographics and urodynamic variables (
Table 3).
Table 4 shows the changes in the SUI VAS score and urodynamic variables from baseline to the primary end-point in the overall patients. The SUI VAS score significantly decreased after PRP treatment. No significant difference was noted in any of the urodynamic measures after PRP treatment, with the exception of ALPP (98.3±55.8 cm H
2O vs. 157.3±79.3 cm H
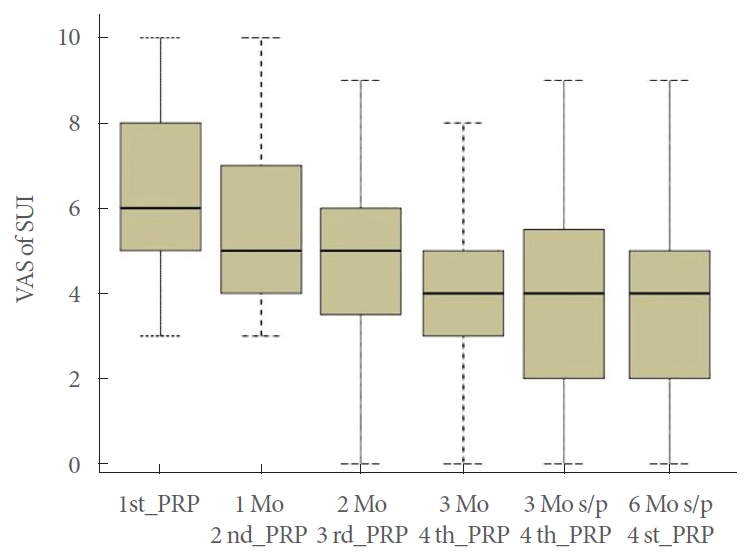
2O, P=0.000) and FPL length (26.2±6.59 mm vs. 28.7±7.46 mm, P=0.037). The mean SUI VAS score improved from 6.57±1.90 at baseline to 5.57±1.99 at 1 month, 4.74±2.19 at 2 months, 4.24±2.02 at 3 months, 3.9 ±2.20 at 3 months after the fourth PRP injection, and 3.77 ±2.41 at 6 months after the fourth PRP injection (P<0.05 for all, compared with baseline) (
Fig. 2). There was a significant difference between the successful and failed treatment groups with respect to changes in the SUI VAS score (−4.0±1.92 vs. −1.36±1.50, P=0.000) and ALPP (74.0±69.7 vs. 28.1±61.1, P=0.050). There was no urodynamic DO at baseline or
de novo DO in the follow-up urodynamic study. The other urodynamic parameters showed no significant difference between groups.
During the video urodynamic study, no urine leakage while performing the Valsalva maneuver was noted in 20 of 30 patients (66.7%) with nonneurogenic SUI; however, all patients with neurogenic SUI showed urine leakage during the stress test. We compared changes in urodynamic variables between patients with neurogenic and those with nonneurogenic SUI; the change in ALPP after PRP injection in nonneurogenic SUI patients (64.7±65.9 cm H2O) was greater than that in neurogenic SUI patients (1.0±71.0 cm H2O) (P=0.056). The change in the SUI VAS score from baseline to 6 months was 3.0±2.23 in the nonneurogenic SUI group and 2.60±2.07 in the neurogenic SUI group (P=0.614). On further analysis of the effect of baseline demographics and urodynamic measures on the treatment outcomes, patients with PPI who had successful treatment outcomes had significantly higher ALPP at baseline (123.0 ±51.0 cm H2O) than those with failed treatment outcomes (56.1±20.1 cm H2O, P=0.001).
After the fourth PRP injection, 3 patients were converted to a suburethral sling procedure and 2 received artificial sphincter implantation due to failed outcomes. The other 30 patients were followed up with a stable condition at 6 months after the fourth PRP injection. The median follow-up duration was 13 months (range, 6–24 months). Adverse events following PRP injection (mild hematuria and pain during micturition) were reported by 10 patients (28.6%). These adverse events resolved after conservative medical treatment. None of the patients developed UTI, difficult urination, or urinary retention after treatment.
DISCUSSION
This is the first study to demonstrate the efficacy of urethral sphincter PRP injections in alleviating SUI in patients with ISD caused by surgical trauma or nonneurogenic diseases. Twenty-one out of 35 patients (60.0%) showed moderate to excellent improvement; among these, 7 (20%) patients became dry after 4 injections of PRP. However, this treatment was not very effective in patients with neurogenic SUI. The therapeutic efficacy lasted for up to 1 year and no severe adverse events were reported.
Since the 1990s, platelet-derived products have been widely used in regenerative medicine. PRP has been applied to hasten healing of macular holes, dental implants, ulcers, burns, bone diseases, as well as for muscle repair and to enhance postoperative tissue recovery [
20-
22]. In our previous study, PRP injections into the bladder suburothelium were found to decrease tissue inflammation and improve urothelial barrier function in patients with interstitial cystitis [
17,
18]. Therefore, it is plausible that the deficient urethral sphincter in SUI may regain its innervation or striated muscle cells through repeat PRP injections.
In our study, 4 urethral sphincter injections of PRP effectively alleviated the severity of SUI by 3.0 points on the VAS in patients with nonneurogenic SUI; 66.7% of nonneurogenic patients were satisfied with this treatment. Although only 7 patients achieved complete dryness after PRP injections, our results demonstrate the efficacy of this novel minimally invasive therapeutic modality for SUI. However, the effects of PRP depend on the release of growth factors from the alpha-granules of platelets [
12], as well as on the response of the progenitor cells in the damaged tissue; therefore, patients with neuropathic urethral sphincter deficiency or those with severe intrinsic sphincter damage may not show an adequate response to 4 injections of autologous PRP. Therefore, PRP treatment should not be recommended to patients with severe ISD or neurogenic SUI.
The therapeutic effect of PRP on ISD was demonstrated by both the subjective improvement in symptoms and the objective increase in ALPP and FPL in urodynamic studies. Patients with successful treatment outcomes showed a greater increase in ALPP than those with failed outcomes. Higher ALPP indicates an increase in the urethral resistance, while a longer FPL suggests increased external sphincter volume. Most patients experienced a lesser degree of SUI immediately after the first PRP injection; this was likely attributable to the bulking effect of the injection. However, most patients perceived improvement before the next PRP injection. The wound healing process usually takes 1 month; therefore, an interval of 1 month between successive injections is rational to achieve further improvement in urethral tissue and an increase in urethral resistance.
In this study, the treatment success rate in patients with previous radical cystectomy and neobladder formation was 83.3%. These patients had lost bladder urethral sphincter coordination during bladder storage, and therefore, may experience SUI at an extremely full bladder. PRP injection increases the resistance of the urethral sphincter, and patients were educated to urinate at regular time intervals; this helped to improve the success rate. Among 21 men with radical prostatectomy, the success rate was 60.0%. The only variable associated with successful or failed treatment outcomes was a higher baseline ALPP; this suggests that a higher grade of SUI due to low urethral resistance may result in less effective treatment outcomes in this patient group. However, age and bladder condition were not associated with treatment outcomes.
Patients with SUI due to radical hysterectomy or radical prostatectomy may have detrusor underactivity in addition to ISD [
23]. The SUI in these patients can be treated with a suburethral sling or artificial sphincter implantation, However, difficulty urinating might be a
de novo issue that bothers patients after surgical treatment of SUI [
24]. If repeated PRP injections can improve the degree of SUI, these patients can be adequately treated with a balance between SUI and spontaneous urination. For patients with normal detrusor contractility and ISD, a trial of PRP urethral injection may also be performed prior to conducting a definitive surgical procedure such as a sling or artificial sphincter.
There are several limitations of this study. The first is the small case number and lack of a placebo control arm. However, because this is a proof-of-concept clinical trial, we should determine the safety and efficacy for this novel treatment before further investigation. Second, although this study demonstrated the clinical efficacy of PRP injections for ISD, the underlying mechanism remains to be elucidated. PRP contains several platelet-derived growth factors and cytokines, which promote revascularization, migration of fibroblasts, and regeneration of mesenchymal cells in the striated muscles [
25]. In this study, we did not observe an increase in the real urethral sphincter volume after PRP injection; however, there was a significant increase in the ALPP and FPL in patients with nonneurogenic SUI. Further imaging studies or electromyographic studies of the external sphincter may provide evidence of the true effect of PRP on urethral sphincter regeneration.
In conclusion, our results demonstrate the safety and efficacy of repeated urethral sphincter injections of autologous PRP in increasing urethral sphincter resistance and alleviating urinary incontinence in patients with SUI due to ISD. However, patients with neurogenic SUI and those with very low urethral resistance may not be good candidates for this treatment.