Advances in Liquid Biopsy for Diagnosis of Bladder Cancer
Article information
Abstract
Bladder cancer (BCa) is the most common malignancy of the urinary system. It has a high recurrence rate and requires longterm follow-up. Significant advances in BCa research have been made in recent years; however, the initial diagnosis and follow-up of BCa relies on cystoscopy, which is an invasive and expensive procedure. Over the past decade, liquid biopsies (e.g., blood and urine) have proven to be highly efficient methods for the discovery of BCa biomarkers. This noninvasive sampling method is used to analyze unique tumor components released into body fluids and enables serial sampling and longitudinal monitoring of tumor progression. Several liquid biopsy biomarkers have been studied extensively and have shown promising results in the clinical applications of BCa, including early detection, microscopic residual disease detection, recurrence prediction, and treatment response. Therefore, this review aims to provide an update on various new liquid biopsy markers and the advantages and current limitations of liquid biopsy in the diagnosis of BCa.
INTRODUCTION
Bladder cancer (BCa) is the leading cause of death and most common malignancy of the urinary system, with an estimated 573,000 new cases and approximately 212,000 deaths worldwide each year [1]. BCa is commonly classified into non–muscle-invasive bladder cancer (NMIBC), muscle-invasive bladder cancer (MIBC), and metastatic disease [2].
Approximately 75% of BCa patients are diagnosed with NMIBC, which can be effectively treated with transurethral resection of bladder tumors; 70% of these tumors recur and 15% progress to the next stage or grade [3]. Therefore, patients diagnosed with NMIBC require frequent treatment and monitoring. NMIBC is a superficial BCa, and its pathological stages include papillary (Ta), lamina propria infiltrate (T1), and carcinoma in situ (CIS). Among these patients, Ta, T1, and CIS accounted for approximately 70%, 20%, and 10%, respectively. Up to 80% of patients with NMIBC relapse within 5 years, 30% of Ta patients develop MIBC, and those with T1 and CIS are more likely to develop MIBC [4]. Due to the high recurrence rate in patients with NMIBC, it is important to develop noninvasive and sensitive methods to monitor recurrence. Currently, most patients with BCa are diagnosed during examinations due to hematuria. Thus, no diagnostic tools are available to screen for bladder cancer (BC). Cystoscopy is a key method for investigating hematuria and is an essential modality for diagnosing and identifying local disease recurrence in NMIBC [5]. However, cystoscopy is an invasive and expensive method that is sometimes associated with complications. Although cystoscopy has improved in sensitivity, the development of a noninvasive method to detect new and recurrent diseases remains a top priority for patients and healthcare professionals [6]. Liquid biopsy is a simple, noninvasive alternative to surgical biopsy that provides information about the disease through a sample of biological fluids such as blood, plasma, urine, or other biofluids. This review summarizes the current landscape of liquid biopsy biomarkers and their clinical applications in BCa.
COMMERCIAL KIT TO DETECT BLADDER CANCER USING LIQUID BIOPSY BIOMARKERS
Several commercial molecular tests have been approved by the U.S. Food and Drug Administration (FDA) for the specific diagnosis of BC [7]. These tests include the detection of soluble proteins such as nuclear matrix protein 22 (NMP22) and bladder tumor antigen (BTA); immunocytochemical measurement of the high molecular weight form of carcinoembryonic antigen (CEA) and 2 bladder tumor cell-associated mucins (ImmunoCyt/uCyt+, DiagnoCure Inc., Quebec, Canada); and chromosomal aneuploidy detected by fluorescence in situ hybridization (UroVysion, Abbott Molecular Inc., Des Plaines, IL, USA).
Nuclear Matrix Protein 22
NMP22 is a protein included in a group of nuclear matrix proteins that plays a significant role in the nuclear mitotic apparatus [8]. Numerous NMPs are overexpressed in urothelial tumors and can be found in the urine after tumor cell apoptosis. NMP22 is a tumor marker for urothelial BC and stands out as a protein that elevated in the urine of patients with BCa [9]. Both the quantitative test NMP22 Bladder Cancer enzyme-linked immunosorbent assay (ELISA) and the point-of-care test NMP22 BladderChek have been approved for surveillance by the FDA. In a comparative test between the 2 methods, NMP22 ELISA kit showed a lower sensitivity compared to NMP22 BladderChek kit (40%– 42% vs. 59%–63%), whereas specificity of 2 kits was similar (93%–99%) [10]. A meta-analysis of 23 studies that evaluated and compared the accuracy of NMP22 BladderChek showed that the overall sensitivity of 56% and specificity of 88% [11].
Bladder Tumor Antigen
BTA is a human complement factor H-related protein. The BTA TRAK/stat is an FDA-approved test for the diagnosis and follow-up of BCa that measures the level of protein in urine. BTA TRAK is a quantitative ELISA assay that requires skilled personnel and a reference laboratory. BTA Stat is a qualitative immunochromatographic assay used as an adjunct to cystoscopy for monitoring BC recurrence. A side-by-side comparison of tumor markers showed that BTA Stat had significantly higher sensitivity for high-grade tumors (74%) than for low-grade tumors (25%) [12]. The BTA Stat has a range of sensitivity from 57%–82% and specificity from 68%–93%, whereas the sensitivity of BTA TRAK ranges from 66%–77% and the specificity ranges from 50%–75% [13-15].
ImmunoCyt/uCyt+
ImmunoCyt/uCyt was developed to improve the low sensitivity of cytology and is a product that combines 3 monoclonal antibodies [16]. Among these antibodies, M344 and LDQ10, labeled with the green fluorophore fluorescein, were raised against mucin-like antigens, while 19A211, labeled with Texas Red, recognized the high molecular weight form of CEA. M344 is expressed in 71% of Ta–T1 tumors and 19A211 is expressed in 90% of Ta–T1 tumors [17]. In a study of 870 urine analyses, the combined cytology and ImmunoCyt/uCyt assay had an overall sensitivity of 84%, specificity of 61%, and negative predictive value of 95% [18]. Another study including patients with 85 recurrent and 58 newly diagnosed bladder tumors confirmed using cystoscopy combined both tests to show sensitivities of 66.7%, 78%, and 87% for different tumor grades, including G1, G2, and G3 transitional cell carcinomas [19].
UroVysion
UroVysion is an FDA-approved test for detecting BC that uses a DNA probe complex labeled at specific chromosomal foci to identify changes in chromosomes 3, 7, 17, and 9p21 loci in specific cells. It uses labeled DNA probes to identify specific chromosomal changes in cells found in urine. In many studies, this test has a sensitivity of 69%–74% and a specificity of 65%–95% [20, 21]. A meta-analysis of 13 studies showed that the specificity of UroVysion was 83% and sensitivity was 72%, compared to urine cytology whose specificity and sensitivity was 96% and 42%, respectively [22]. In another study, UroVysion showed sensitivity and specificity of 75.6% and 84.8% in high-grade BCa, whereas it showed sensitivity and specificity of 40.8% and 87.8% in low-grade BCa, respectively [23].
LIQUID BIOPSY IN CLINICAL TRIALS
Recently, liquid biopsy has attracted attention as a less invasive method with broad applicability. However, further research is required to validate the clinical application of liquid biopsy. Several clinical trials are currently underway in this field. We have summarized the clinical trials of liquid biopsy for BC in the last 5 years in Table 1.
Many clinical trials have attempted to diagnose BC using various methods including the detection of mutations and methylation in DNA; levels of microRNA (miRNA), messenger RNA (mRNA), and protein; and magnetic resonance imaging. However, liquid biopsy devices and kits face challenges in obtaining approval for diagnostic use owing to technical limitations and issues in standardizing procedures across laboratories. There is a lack of standardized methods for testing samples, setting thresholds, and interpreting data. Extensive clinical trials and feasibility studies with large sample sizes and multiple centers are necessary to address these issues and provide accurate clinical answers.
DIAGNOSTIC BIOMARKERS FOR BLADDER CANCER CAN BE IDENTIFIED FROM BLOOD SAMPLES
Circulating Tumor Cells
Circulating tumor cells (CTCs) are cancer cells shed from primary tumors and enter the bloodstream [24]. CTCs play a crucial role in metastasis by allowing cancer cells to travel through the bloodstream and establish secondary tumors in distant organs. CTCs can be released into circulation at an early stage of cancer, sometimes even before a tumor is clinically detectable. Therefore, the detection of CTCs may allow for the diagnosis of BCa at an earlier and potentially more treatable stage [25]. Qi et al. [26] reported that the quantitation of CTCs through folate receptor α ligand-polymerase chain reaction (PCR) could be a promising noninvasive method for diagnosing bladder transitional cell carcinoma. Its reasonably high sensitivity and specificity suggest that this test has potential utility in identifying individuals with BCa [27].
The epithelial cell adhesion molecule (EpCAM) is a transmembrane glycoprotein commonly expressed on the surface of epithelial cells, including cancer cells of epithelial origin. EpCAM has been widely used as a marker for the detection and isolation of CTCs since many solid tumors such as breast, colorectal, and prostate cancers, are of epithelial origin [28]. Moreover, there was a significant association between highgrade advanced stages of BCa and the expression of EpCAMs; the correlation between EpCAM expression and poor overall survival (OS) implies a potential role for EpCAM in tumor progression [29]. Thus, the use of EpCAM as an imaging target for the diagnosis of lymph node metastases in migratory cell carcinoma of the bladder is valuable application [30].
Rink et al. [31] suggested that CTCs are more frequently detected in patients with metastatic disease than in those with nonmetastatic disease. The presence of CTCs in individuals with nonmetastatic disease could indicate a subset of patients at higher risk or with more aggressive disease characteristics. The high detection rate in the metastatic group may also suggest a potential correlation between CTC presence and disease progression [32]. CTC detection as a prognostic tool has potential utility in BCa management. These findings suggest that approximately one-quarter of the 188 patients with BCa had detectable CTCs prior to treatment, which could include surgery, chemotherapy, or other interventions. Importantly, patients who tested positive for CTCs and subsequently underwent treatment showed poorer progression-free survival, cancer-specific survival, and OS compared to those who were CTC-negative [33]. These investigations have consistently reported correlations between the presence of CTCs and various clinical parameters such as tumor stage, lymph node metastasis, and survival outcomes [34]. Additionally, higher levels of CTCs were detected in MIBC compared with NMIBC. This finding suggests that CTCs may predominate during the invasive stages of the tumor, particularly when the cancer infiltrates the muscle layer of the bladder [35].
Circulating Tumor DNA/Cell-Free DNA
Tumor cells undergo various processes such as apoptosis, necrosis, and active release, which can result in the release of DNA into the bloodstream. Circulating DNA is often referred to as cell-free DNA (cfDNA) or circulating tumor DNA (ctDNA) when it originates from tumor cells [36]. Liquid biopsy techniques, which involve analyzing cfDNA in bodily fluids such as blood, have gained importance in cancer research and clinical practice owing to their minimally invasive nature and potential for real-time monitoring [37]. Researchers and clinicians can use techniques such as quantitative PCR, digital PCR, nextgeneration sequencing, and other molecular biology methods to analyze genetic alterations in ctDNA, thereby providing valuable information about the genetic makeup of tumors without requiring invasive procedures such as tissue biopsies [38]. There is often a correlation between the amount of ctDNA in the bloodstream and the size and stage of the tumor [39]. As tumors grow or progress, they shed more DNA into the circulation.
The association between ctDNA methylation status and the aberrant methylation of tumor suppressor genes in preoperative plasma samples from patients with BCa is indicative [40]. p16, specifically its promoter region, undergoes alterations in DNA methylation in BCa [41]. The results of a small cohort study indicate that the hypermethylation status of serum ctDNA could be a potential biomarker for BC detection [42]. This study suggests that the hypermethylation status of specific genes in serum cfDNA, particularly APC, GSTP1, and TIG1, may serve as a promising biomarker panel to distinguish patients with BCa from healthy individuals. Additionally, ctDNAs are often detected in metastatic BCa, thereby reflecting the presence of tumor-derived genetic material.
The identification of gene mutations in ctDNA, such as FGFR3, PIK3CA, ERBB2, and EGFR, is an important approach in the field of liquid biopsy for BCa [43]. Additionally, the identification of potential mutations in ctDNAs from patients with metastatic BCa (including RB1, CDKN2A, and ERBB2), MAPK/ERK or PI3K/AKT/mTOR pathway-associated mutations, and chromatin remodeling-associated mutations provides crucial insights into the disease [44].
CtDNA testing has potential value as a tool for the early identification of disease recurrence after RC or triple therapy for MIBC [45]. CtDNA analysis can also identify specific genetic alterations such as FGFR3, DRCC2, ERBB2, and TMB which have been proposed as biomarkers for BCa treatment response [46]. Recognizing the association between specific ctDNA mutations and BCa prognosis offers a new reference point and foundation for personalized BC treatment [47].
Cell-Free RNA (mRNA, miRNA, Long Noncoding RNA)
CfRNA stands for “cell-free RNA,” which refers to RNA molecules that are found outside of cells. CfRNA is usually present in body fluids such as blood, saliva, urine, and cerebrospinal fluid [48]. There are different types of cfRNAs including circulating miRNAs, mRNA, and long noncoding RNA (IncRNA). Techniques such as liquid biopsy, which involves the analysis of cfRNA in bodily fluids, have been explored for cancer detection and monitoring, as tumors release specific cfRNA signatures into the bloodstream [49].
Circulating miRNAs hold promise as noninvasive biomarkers for BCa diagnosis and prognosis. It has been reported that miR-19a expression is significantly increased in both tumor tissues and plasma in patients with BC, while miR-99a expression is significantly decreased [50]. Motawi et al. [51] suggested that miRNAs (miR-92a, miR-100, and miR-143) may show differential expression patterns in the plasma of individuals with BCa, thereby making them potential candidates for use as biomarkers to detect or monitor diseases. A subsequent study found that the expression levels of 7 miRNA panels (miR-6087, miR-6724-5p, miR-3960, miR-1343-5p, miR-1185-1-3p, miR6831-5p, and miR-4695-5p) may show patterns indicative of early stages, which allows for timely and accurate diagnosis [52].
Marin-Aguilera et al. [53] tested a panel of bladder-specific mRNA markers, including FXYD3, C10orf116, AGR2, KRT19, KRT20, and UPK2. There were differences in the expression of C10orf116 and KRT19 between patients and controls. Uroplakin II, cytokeratin (CK) 20, CK-19, and epidermal growth factor receptor mRNA in the blood may be useful markers for BCa [54] and matrix metalloproteinase (MMP)-2, MMP-9, and tissue inhibitors of metalloproteinase-2 mRNA levels are increased in the peripheral blood of BC patients [55].
Exosomes
Exosomes are a specific subtype of extracellular vesicles (EVs), which are membrane-bound vesicles released by cells into the extracellular environment [56]. Exosomes carry a variety of molecular cargo, including proteins, nucleic acids (DNA, miRNA, IncRNA, and circular RNA), lipids, and metabolites, and play a role in oncogenesis [57]. The specific proteins CD9, CD81, CD63, ALIX, TSG101, and HSP70 are commonly associated with exosomes and are often used as potential cancer diagnostic biomarkers [58].
Serum exosomes have been studied as potential noninvasive biomarkers for the early diagnosis of BCa. Exosomal IncRNA H19, lncRNA-rich uroepithelial carcinoma-associated 1 exosomes, and exosomal IncRNA PTENP1 are potential diagnostic biomarkers for BCa [59-61]. Furthermore, some studies have demonstrated the combined application of exosomal noncoding RNAs, such as exosomal IncRNAs (PCAT-1, UBC1, and SNHG16) in the diagnosis of BCa [62].
Numerous studies have explored the potential of blood-based exosomes as noninvasive prognostic biomarkers for BCa. For example, miR-663b and miR-4644 levels were elevated in the plasma exosomes of patients with BCa [63, 64]. Conversely, dysregulation of specific miRNAs such as miR-375-3p and miR-133b in the serum has been reported in BCa [65, 66]. Moreover, it was confirmed that the downregulation of miR-185-5p and miR1061-5p or the upregulation of miR-10b-5p in exosomes from BCa serum was significantly associated with cancer progression [67].
Metabolites/Proteins/Peptides
Serum metabolomic and proteomic studies offer valuable insights into the molecular changes associated with BCa. The identification of specific biomarkers can contribute to the development of noninvasive diagnostic tools and personalized treatment strategies for individuals with BCa [68].
Metabolomics plays a crucial role in cancer research by providing valuable information for risk assessment, real-time monitoring, prognosis prediction, and evaluation of treatment responses [68]. Metabolomics involves the systematic study of small molecules, often referred to as metabolites, and can provide critical information regarding cancer stage [69]. For example, a combination of dimethylamine, glutamine, and malonic acid was isolated between different grades of BCa using nuclear magnetic resonance (NMR) spectroscopy-based serum metabolomics [70]. The identification of specific combinations of serum metabolites, such as inosine, N1-acetyl-N2-formyl-5-methoxykynurenine, and phosphatidylserine, as predictors of high-level BCa further exemplifies the potential of metabolomics in developing diagnostic tools [71]. More recently, 4 potential serum biomarkers (isobutyrate, pyroglutamate, choline, and acetate) for BCa were quantified using proton NMR spectroscopy and a metabolic pathway impact analysis was conducted through NMR and laser desorption/ionization-mass spectrometry. Seven pathways (glycine, serine and threonine metabolism, glycerophospholipid metabolism, propanoate metabolism, glutathione metabolism, pyruvate metabolism, glyoxylate and dicarboxylate metabolism, and glycolysis/gluconeogenesis) were associated with BCa [72].
Proteomics-based analyses of liquid biopsies play a crucial role in advancing BCa research and clinical applications. The identification of differentially expressed proteins, including S100A8 and S100A9, as accurate discriminators between BCa and healthy controls underscores the potential of proteomic approaches for advancing diagnostic strategies [73]. The identification of abnormally expressed serum levels of S100A4, S100A8, S100A9, CA-I, and annexin V as effective protein markers for BCa offers promise for BCa diagnosis and management [74] and high levels of S100A12 have been detected in BCa diagnosis [75]. The levels of MMP23B protein in the blood are increased in BCa, thereby suggesting its potential as a biomarker for BCa [76]. During the clinical course, serum alpha-fetoprotein levels in patients with BCa have been reported to correlate with the treatment response including chemotherapy, surgery, and immunotherapy, such as pembrolizumab [77].
DIAGNOSTIC BIOMARKERS FOR BLADDER CANCER CAN BE IDENTIFIED FROM URINE SAMPLES
A voided urine specimen is an ideal biological source of normal bladder and exfoliated tumor cells [78]. Therefore, urine is considered the best specimen for the noninvasive diagnosis of BCa. Various molecules can be measured in urine, including DNA, RNA, and proteins.
Genome-Based Biomarkers
Urinary DNA molecules are classified into cellular DNA, cfDNA, and exosomal DNA. Several studies have confirmed that urinary DNA biomarkers can be used to predict the risk of BCa in patients with hematuria [79, 80]. Considering the genomic complexity of BCa, urinary DNA provides the most powerful tumor-specific information for noninvasive BCa detection. Epigenetic modifications, including DNA methylation, are fundamental mechanisms of carcinogenesis, particularly in BCa [81]. DNA methylation primarily occurs at the CpG dinucleotides. These CpG dinucleotides are spread throughout the genome and most of them are methylated. Some regions of the genome have a high CpG density and are called CpG islands. Generally, hypermethylation of abnormally unmethylated CpG islands in the promoter regions of tumor suppressor genes suppresses transcription in human tumors [82]. Therefore, aberrant DNA methylation is a potential biomarker for the diagnosis and monitoring of BC. DNA methylation can be detected in urinary DNA of patients with BCa [83]. Recently, genomic methylation of CFTR, SALL3, and TWIST1 was compared between 111 BCa and 57 control urinary cells [84]. In this study, methylation of the 3 genes achieved an area under the curve (AUC) of 0.741, whereas urinary cytology reached an AUC of 0.696. TWIST1 and NID2 were also independent predictors of BCa, which supports the results of several previous studies. Previously, Renard et al. [85] have showed a sensitivity and specificity of >90% for detecting BCa. Regarding the methylated genes that differentiate between normal and cancerous urothelium, TWIST1 is also part of a panel of urinary biomarkers in a large hematuria cohort assessed for the presence or absence of urothelial carcinoma. In other cases, the combination of SOX1, IRAK3, and L1-MET has been shown to provide better resolution than cytology or cystoscopy in detecting early recurrence [86]. Hypermethylation of several genes, including APC, RARb2, and GSTP1 has been observed in patients with urothelial BCa. These methylated genes allowed identification of BCa patients with a sensitivity of 62% and a specificity of 89% [87]. In another study, the UroMark assay showed sensitivity, specificity, and negative predictive value of 98%, 97%, and 97%, respectively for the detection of primary BCa compared to urine from normal counterparts [88].
In addition, genomic mutations can be detected in urinary DNA, including cellular, cell-free, and exosomal DNA. Previously, deep sequencing enabled large-scale identification of somatic mutations (SMs) in BCa and sensitive detection of SMs in urine DNA [89]. However, whole-exosome and whole-genome sequencing remain expensive at depths sufficient to detect SM at low mutant allele frequencies (MAFs). Therefore, targeted sequencing of a minimum number of SM panels that provide sufficient information is desirable to make the test inexpensive and interpretable.
Most urine DNA-based studies use DNA extracted from centrifuged urine cell pellets (cpDNA). The multiplex assays including intragenic mutations in 10 frequently mutated genes in urothelial tumors (FGFR3, TP53, PIK3CA, HRAS, KRAS, CDKN2A, ERBB2, MET, VHL, and MLL) detected mutations in 68% of the 175 urinary cell pellet samples with BCa [86]. A total of 246 SMs were detected in 8 of the 10 target genes. The median MAF of urinary cellular DNA with detectable mutations was 8% (8.14%). The most commonly altered genes were TP53 (45% of all mutations) and FGFR3 (20% of all mutations). In a multicenter study, sensitivity of the combination assays for detection of BCa recurrence was 57% in patients with primary low-grade NMIBC. However, sensitivity for recurrence, pT1, and MIBC was 83% [90].
CfDNA is a short DNA fragment released into body fluids through natural cell death. Recently, several studies reported that cfDNA extracted from centrifuged urine supernatants revealed genomic changes in BCa [91, 92]. In a pilot study of 377 biological fluid samples from 12 patients with recurrent, progressive, and metastatic BCa, patients with advanced disease had significantly higher levels of tumor DNA in their plasma and urine before disease progression than patients with recurrent disease [91]. Additionally, a comparison of genomic profiles of cellular DNA and cfDNA from urine with matched tumor DNA from 23 patients with BCa showed that urine DNA was highly representative of patient tumors and capable of detecting recurrent genomic abnormalities [89]. Diagnosis consisting of 5 target genes (TERT, FGFR3, TP53, PIK3CA, and KRAS) or 7 genes (TERT, FGFR3, TP53, PIK3CA, KRAS, HRAS, and KRAS) through bioinformatics analysis of urinary DNA mutations has also shown successful identification of malignancy in patients with hematuria [93]. Recently, to detect molecular residual disease in patients with BC, urinary cfDNA from 74 patients with localized BCa was analyzed using ultra-low-pass whole-genome sequencing and urine cancer personalized profiling by deep sequencing. In this study, variant allele frequency, estimated tumor mutation burden, and copy number-derived tumor fraction levels of urinary cfDNA significantly predicted pathological complete response status better than plasma ctDNA. These findings demonstrate that urine cfDNA can sensitively detect MRD and accurately predict patient survival [94].
Protein and Peptides
Urinary proteins and peptides are easily preserved and are more stable than other urinary biomarkers. Another advantage of protein biomarkers is that they are easy to use and can be used in point-of-care tests that provide rapid results. Several new tests and biomarkers based on proteins found in the urine have been investigated for the early diagnosis of BCa. Among recently introduced protein biomarkers, the URO17 test utilizing keratin 17 (K17) has shown particularly promising results. In this retrospective study, the sensitivity and specificity of URO17 showed 100% were confirmed using urine samples from BCa patients [95]. Another study observed high concentrations of MMP-2 and MMP-9 proteins in the blood and urine of BCa patients and showed 100% sensitivity and specificity for BCa diagnosis through receiver operating characteristic curve analysis [96]. A diagnostic study involving 3 protein biomarkers (AIB1, EIF5A2, and NMP22) was conducted with 92% sensitivity and specificity in a study of 210 subjects [97].
Exosomes
Additionally, urinary exosomes have emerged as promising diagnostic biomarkers for BCa [98]. Exosomes are released from cells and discharged into various body fluids such as blood and urine. Exosomes are a subset of EVs that contain a variety of proteins, lipids, and nucleic acids and may be potential biomarkers of malignancy. Most studies on the utilization of nucleic acids in exosomes as biomarkers have focused on miRNAs or mRNAs; however, exosomes containing DNA fragments and genomic alterations have been identified in various cancers. Several studies have shown that urinary exosomal DNA can be detected in patients with BCa [99, 100]. Targeted deep sequencing of 9 genes and copy number variation analysis of the entire genomic region revealed that urine cfDNA and exosomal DNA are representative of the entire human genome and enable the genomic profiling of BCa [99]. Moreover, urine from patients with BC is characterized by significantly higher exosome concentrations and exosomal DNA content than normal samples. A study of whole-exosome sequencing of urine exosomal DNA-matched DNA from serum, peripheral blood mononuclear cells, and tumor tissues from 6 patients, demonstrated that urine exosomal DNA captures SMs in several genes and several unique driver variants. showed an outstanding potential [100].
Metabolites
Recently, several studies have used urine metabolomic analysis to identify useful biomarkers for BCa diagnosis and monitoring. In a urine study of 51 patients with BCa and 49 healthy volunteers, several differential metabolites were identified through comparative urine metabolomics, and 8 putative markers were selected to distinguish early stage BCa, with an AUC of 0.97 [101]. A recent study showed that a metabolic panel based on 11 biomarkers (glycochenodeoxycholic acid, adenosine monophosphate, 5-Aminolevulinic acid, myristic acid, chenodeoxycholic acid, salicyluric acid, proline, N-acetylserine, picolinic acid, hydroxypropionic acid, and 4-Hydroxybenzoic acidcan) distinguished between BCa patients and healthy volunteers, with a sensitivity of 95.3% and a specificity of 100% [102]. Another study identified a 4 urinary biomarker panel (5-hydroxyvaleric acid, cholesterol, n3-phosphoglyceric acid, and glycolic acid) with a sensitivity of 88.0% and specificity of 85.7% for the diagnosis of BCa [103].
CONCLUSION
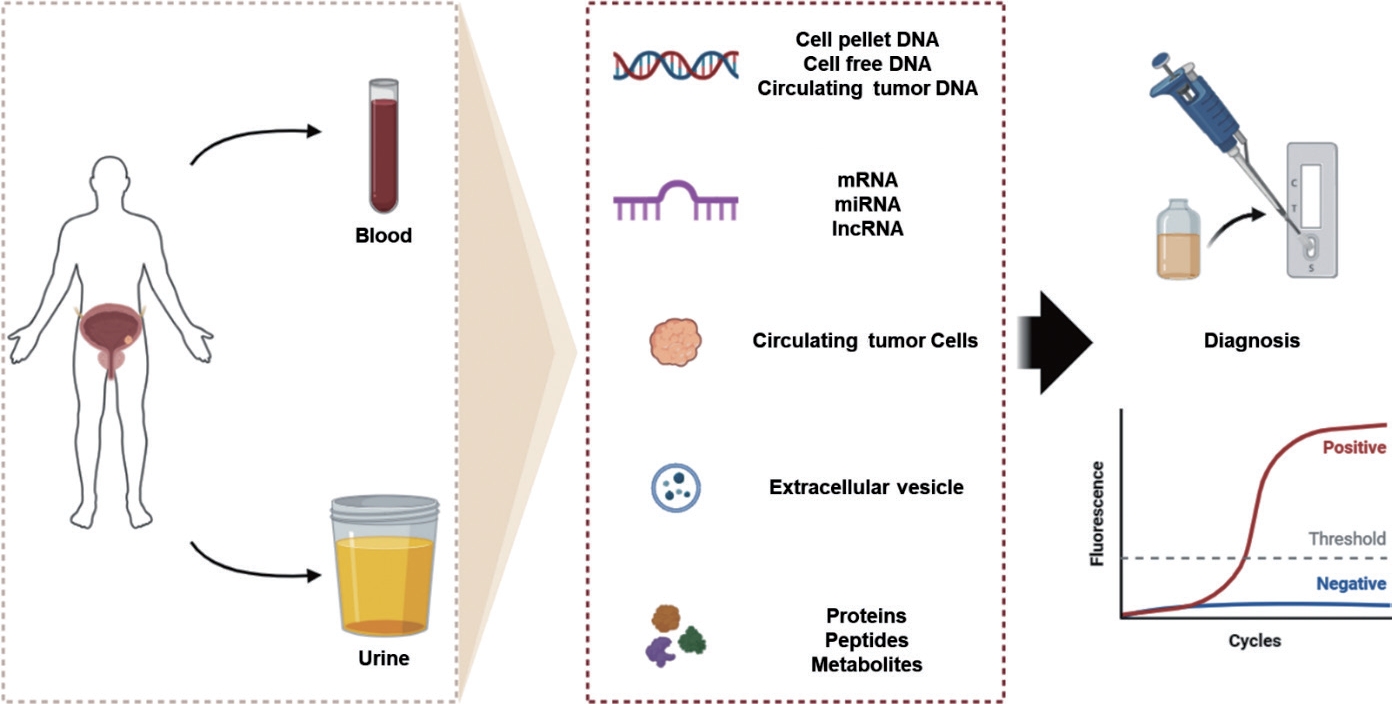
Management strategies for patients with BC face challenges associated with invasive biopsy methods, patient anxiety, and financial burden. Liquid biopsy has emerged as a promising alternative that offers effectiveness in cases where tissue samples are unavailable, and can potentially reduce patient discomfort by deferring invasive procedures. Although various blood and urinary diagnostic markers have been reported, most FDA-approved markers lack sufficient diagnostic capabilities such as sensitivity to low-grade BCa and susceptibility to false positives. Non-FDA-approved markers show promise but often exhibit inconsistent performance and low reproducibility. To date, no urinary marker has replaced cystoscopy for the detection of low-grade BCa. However, panel tests that assess multiple genomic or transcriptomic targets demonstrate high sensitivity for high-grade BCa, thereby potentially reducing the need for cystoscopy. The feasibility of implementing these panel tests depends on cost reduction. In addition, while noninvasive liquid biopsy holds the potential for early BC diagnosis, precise drug administration, disease monitoring, and predictive assessment, large-scale clinical trials are essential to validate its clinical effectiveness (Fig. 1).
Notes
Grant/Fund Support
This study was supported by a grant from the National Research Foundation (NRF), funded by the Korean Government (Ministry of Science and ICT) under Grant numbers RS-2023-00211812.
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION STATEMENT
· Conceptualization: JS, YK, SK, HGK, EMK, AK
· Funding acquisition: AK
· Project administration: AK
· Visualization: JS, AK
· Writing - original draft: JS, EMK, AK
· Writing - review & editing: JS, EMK, AK