|
 |


- Search
| Int Neurourol J > Volume 27(3); 2023 > Article |
|
ABSTRACT
Purpose
We evaluated the therapeutic effects on overactive bladder (OAB) symptoms and sexual function of behavioral therapy with or without mirabegron in sexually active male patients with OAB. Mirabegron, a selective ╬▓3 adrenoceptor agonist for the treatment of OAB, has been shown to induce corpus cavernosum relaxation.
Methods
In this 4-site, randomized controlled trial, 150 sexually active men with OAB were enrolled between June 2020 and May 2022. Participants were randomly allocated (1:2) into 2 treatment groups: (1) behavioral therapy alone (n = 50) and (2) a combination of mirabegron 50 mg daily and behavioral therapy (n = 100). The evaluation was based on the overactive bladder symptoms score (OABSS), the International Index of Erectile Function, the ejaculatory domain short form, the International Prostate Symptom Score, patient perception of bladder condition, quality of life, and urodynamic parameters. The therapeutic outcomes were assessed at baseline, 4 weeks, and 12 weeks.
Results
There were 65 patients (65%) in the combination subgroup and 36 patients in the behavioral therapy who completed all 12 weeks of treatment. Both groups had a statistically significant improvement in OABSS after 12 weeks of treatment. The combination therapy group achieved a statistically significant improvement in all 4 subscores of OABSS, however, the urinary frequency (P = 0.120) and urinary incontinence (P = 0.234) subscores in the behavioral therapy only group did not show a significant change. Additionally, the combination group had a significant improvement in functional bladder capacity, which was not seen in the behavioral therapy group. However, both groups did not have a significant change in erectile or ejaculatory function.
Overactive bladder (OAB) syndrome is defined by the International Continence Society as urgency, with or without incontinence, usually associated with urinary frequency and nocturia. Some papers have reported that the prevalence rate for men with OAB is higher in some Asian countries than in Europe, the USA, and Canada [1-3]. OAB was reported to be significantly associated with an increased prevalence of erectile dysfunction, and reduced sexual activity and satisfaction due to lower urinary tract symptoms (LUTS) [4,5].
Mirabegron, a selective beta-3 adrenoceptor agonist, is the first drug of its type to be clinically available for the treatment of OAB. Beta-3 adrenoceptors are the predominant beta receptors expressed on detrusor smooth muscle cells, and their stimulation is thought to induce detrusor relaxation. In addition, the activation of beta-3 adrenoceptors initiates relaxation of strips from the corpus cavernosum (CC) in humans and rats via the nitric oxide (NO)-cyclic guanosine monophosphate (cGMP) signaling pathway [6]. A previous study proposed that erectile function (EF) was improved by neurogenic relaxation after intracavernous administration of mirabegron in diabetic rats [7]. In previous human studies, 2 research groups demonstrated that mirabegron improves both OAB symptoms and EF in male OAB patients without serious adverse effects [8,9]. In 1 systemic review article, combination therapy of phosphodiesterase type 5 inhibitor (PDE5I) and mirabegron showed significant improvement on urological symptom scores or quality of life (QoL) compared to PDE5I therapy only [10]. Nevertheless, nearly all of the reviews did not explicitly state the effect of mirabegron on sexual function.
According to the updated treatment guidelines published by the American Urological Association and European Association of Urology, behavioral therapy is considered the first-line treatment for patients with OAB, followed by pharmacotherapy. However, one earlier study has shown that combining behavioral and drug therapy yields greater improvements in OAB symptoms compared with drug therapy alone [11]. However, there was no report on male EF and ejaculatory function in OAB patients in that randomized controlled trial (RCT). Consequently, in the present study, we aimed to evaluate the effectiveness of behavioral therapy alone and a combination of behavioral therapy and mirabegron medication for reducing OAB symptoms and effects on sexual function in male OAB patients.
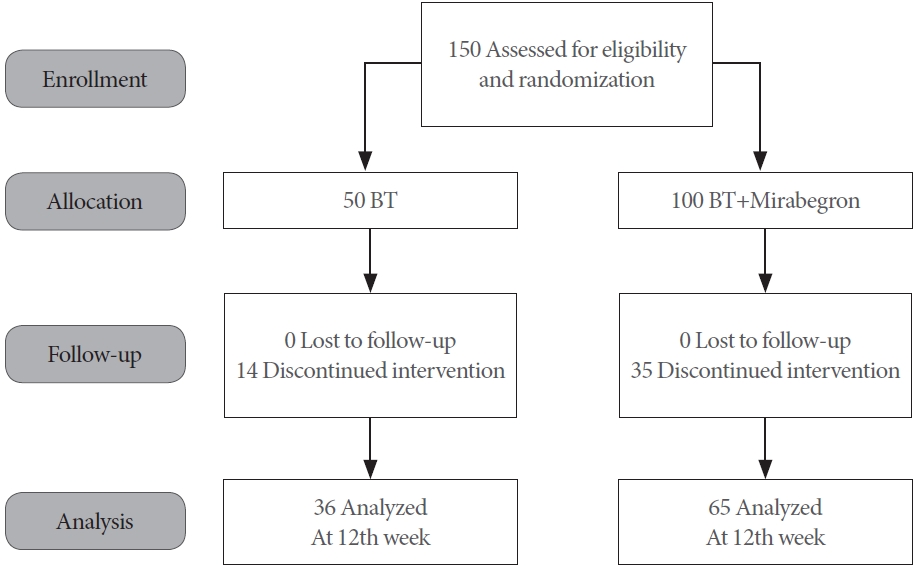
Sexually active men aged Ōēź 20 years old with OAB symptoms were enrolled between June 2020 and May 2022. The overactive bladder symptoms score (OABSS) questionnaire was used to diagnose OAB; a positive diagnosis was considered if the OABSS urgency score was Ōēź 2 and the sum scores were Ōēź 3. These recruited patients should be able to have sexual activity during study period. Written informed consent was obtained from the participants prior to their inclusion within the study. This trial followed the CONSORT (Consolidated Standards of Reporting Trials) guidelines (Fig. 1).
A total of 150 patients were recruited and allocated into the 2 study subgroups in a 2:1 ratio. The 2 study subgroups were ŌĆ£combined behavioral therapy and mirabegronŌĆØ and ŌĆ£behavioral therapy aloneŌĆØ (Fig. 2). The permuted block randomization method was used and each randomization number was assigned according to the time-sequence for when the screened patient became eligible for inclusion.
The exclusion criteria were concurrent use of phosphodiesterase type 5 inhibitor (PDE5I) or testosterone therapy during the study period, a history of stress urinary incontinence, evidence of an active urinary tract infection or urinary tract stone, confirmed genitourinary tract or pelvic malignancy, genitourinary tract operation during 3 months prior to baseline, postvoid residual urine (PVR) volume Ōēź 100 mL, history of uncontrolled hypertension (systolic > 180 mmHg and/or diastolic > 110 mmHg), history of intolerance to mirabegron, and a history of intravesical onabotulinum toxin A treatment within the last 6 months.
After randomization, all patients in both groups received 12 weeks of behavioral therapy, urge control strategy, conducted by verbal instruction of each physician investigators or nurse practitioners at baseline visit. The urge control strategy consisted of a reduction in fluid intake at specific times, avoidance of caffeine and alcohol, usage of relaxed and double-voiding techniques, urethral milking, distraction techniques, and bladder retraining to increase bladder capacity and the time interval between voids. In addition, avoidance or substitution of medications with urinary effects, and treatment for constipation were recommended. In second visit, success or failure with bladder control strategies discussed in detail to improve results and adherence.
In the combination subgroup, all participants received mirabegron 50 mg once daily for 12 weeks in addition to the behavioral therapy. In both subgroups, patients who were previously prescribed alpha-blockers and/or 5-alpha reductase inhibitors continued to receive their medication.
All participants received a thorough urological check-up before they started treatment. This included taking a history of all personal underlying diseases, measuring serum prostate-specific antigen (PSA) levels, a transrectal sonography of the prostate to measure prostate size, completion of the International Prostate Symptom Score (IPSS) questionnaire, a uroflowmetry test, completion of a 3-day voiding diary and measurement of PVR volume by ultrasonography.
There were 2 primary outcomes in this study; the first was change in OAB symptoms as determined by the OABSS, and the second was change in sexual function as determined by the abridged 5-item version of the International Index of Erectile Function (IIEF-5). The others were secondary outcomes, such as patient perception of bladder condition (PPBC) and QoL, and the Male Sexual Health Questionnaire - Ejaculatory Domain (MSHQ-EjD) Short Form. The therapeutic outcomes (IPSS, OABSS, PPBC, QoL, IIEF-5, maximum flow rate, and PVR volume) were assessed at baseline, 4 weeks, and 12 weeks. All outcome measures were assessed and scored in a blinded fashion and all adverse effects were recorded.
Based on our previous publication [12], the IIEF-5 score decreased from 16.4 ┬▒ 5.5 to 15.6 ┬▒ 5.6 (P = 0.106) and 14.9 ┬▒ 5.9 (P = 0.077) at 4- and 12-week follow-up, respectively. Group sample sizes of 78 and 37 achieve 80% power to detect a difference of 2.600 in a design with 3 repeated measurements having a compound symmetry covariance structure when the standard deviation (SD) is 5.600, the correlation between observations on the same subject is 0.500, and the alpha level is 0.050. With 30% drop out rate, the estimated sample size would be around 150.
All data are described as the mean ┬▒ SD for continuous variables or as n with percentage for categorical variables. P < 0.05 was considered to indicate a statistically significant difference. All changes in symptom scores, including IPSS, IIEF-5, OABSS, QoL, uroflowmetry parameters, and PVR volume were recorded at baseline and at every subsequent visit. All statistical analyses were performed using SPSS ver. 14.0 (SPSS Inc., Chicago, IL, USA).
A total of 100 patients were allocated into the combined behavioral therapy and mirabegron subgroup however 35 (35%) withdrew during the 12 weeks of treatment. A total of 50 patients were allocated into the behavioral therapy alone subgroup however 14 (28%) withdrew during the 12 weeks of treatment (Fig. 1). There was no statistically significant difference in the age, prostate size, PSA level, and testosterone level at baseline between the 2 groups (Table 1). Furthermore, the percentage of comorbidity and prescription of alpha-blockers and 5╬▒ reductase inhibitors were not significantly different between the 2 groups.
There was no statistically significant difference in OABSS, urodynamic parameters, PPBC, 3-day voiding diary, and IPSS at baseline between the 2 subgroups (Table 2). The OABSS questionnaire showed a significant improvement for all symptoms and the total score 4 weeks after combination treatment (Table 2). In the behavioral therapy alone group, total score for the OABSS also showed a statistically significant improvement (from 8.1 ┬▒ 2.7 to 6.5 ┬▒ 2.0, P = 0.011) after 4 weeks treatment; OABSS nocturia (from 2.2 ┬▒ 0.9 to 1.7 ┬▒ 1.2, P = 0.022) and urgency subscores (from 3.5 ┬▒ 1.4 to 2.8 ┬▒ 2.0, P = 0.035) also showed a significant improvement after 12 weeks of treatment. However, the OABSS frequency subscore showed no significant improvement after behavioral therapy alone. Bladder capacity in the combination subgroup was significantly increased after mirabegron (4 weeks and 12 weeks) treatment. However, in the behavioral therapy only subgroup, there was no significant difference in bladder capacity after 12 weeks of therapy.
The functional bladder capacity as determined by the voiding diary, revealed no significant increase after both treatment approaches (Table 2). In the behavioral therapy alone subgroup, all parameters in the 3-day voiding diary, except for daytime frequency, showed no significant change after treatment. Whereas the combination subgroup reported a statistically significant decrease in voiding times (daytime from 8.8 ┬▒ 3.4 to 7.9 ┬▒ 2.3, P = 0.044; night time from 1.5 ┬▒ 1.0 to 1.2 ┬▒ 0.8, P = 0.041) after 12 weeks treatment. Both the behavior only and combination subgroups revealed a significant improvement in bladder condition as measured by the PPBC (from 2.4 ┬▒ 1.2 to 1.5 ┬▒ 1.1, P = 0.001; from 2.6 ┬▒ 1.2 to 1.4 ┬▒ 1.0, P < 0.001, respectively).
IPSS total score, QoL, and voiding subscores showed statistically significant decreases after 4 weeks of treatment in both the behavior only and combination subgroups (Table 2). The combination subgroup had a significantly lower nocturia subscore at 4 weeks compared with the behavior only subgroup. Interestingly, there was no significant difference in the treatment effect after 12 weeks of treatment between the combination subgroup and the behavior only subgroup (storage subscore, 4.4 ┬▒ 2.5 and 4.9 ┬▒ 2.8, P = 0.376, nocturia subscore, 1.5 ┬▒ 1.0 and 1.7 ┬▒ 1.1, P = 0.373).
As for erectile and ejaculatory functions, there were no significant changes in the IIEF-5 score, and the 4 items of the MSHQ-EjD short form in both subgroups at 4 weeks and 12 weeks compared with baseline (Table 3).
Behavioral therapy along with pelvic floor muscle training and urge suppression strategies, are recognized by treatment guidelines as a conservative first-line therapy for OAB [13-15]. As shown by previous studies, the advantages of behavioral therapy are avoidance of adverse effects and the effective reduction of OAB symptoms, especially urgency urinary incontinence and urinary frequency, by counseling patients to control urgency and inhibit detrusor contractions [16,17]. In most studies regarding behavioral therapy, researchers demonstrated that a combination of behavior and drug therapy had an additive effect for improving OAB symptoms such as urgency and urinary frequency [11,17-19]. While there are more strong evidences for the use of combination therapy in women, the effectiveness of combined behavioral and drug therapy for men is less clear [18,20-22]. Recently, some studies demonstrated that a combination of antimuscarinic medication and behavioral therapy was better than antimuscarinic medication alone for men [11,18,19].
Mirabegron, is an alternative treatment option to antimuscarinics for LUTS associated with OAB [23]. In 1 meta-analysis, Sebastianelli et al. [24] reported that mirabegron 50 mg provides a reduction in urinary frequency, urgency, and urgency urinary incontinence, and an improvement in voided volume and nocturia. Kuo et al. [25] conducted a double blind, 3 arm (placebo, mirabegron, and tolterodine), RCT among Asian countries, and demonstrated that mirabegron 50 mg once daily for 12 weeks was superior to a placebo for increasing voided volume and reducing the frequency of micturition in patients with OAB symptoms. To the best of our knowledge, this study is the first RCT to explore the effect of combined behavioral therapy and mirabegron on male sexual function in addition to an evaluation of OAB symptoms. In this study, a combination of behavioral and drug therapy demonstrated improved effectiveness on the OABSS, PPBC, IPSS, and the voiding diary after 4 weeks treatment compared with behavioral therapy alone. Interestingly, both treatment approaches showed no statistically significant differences in treatment outcomes for OABSS total score, PPBC, and IPSS after 12 weeks treatment.
Furthermore, OAB is considered to have a significant association with reduced sexual activity and enjoyment due to storage symptoms [4]. Irwin et al. demonstrated that patients with OAB (n = 502) have a statistically higher incidence of ED than controls (n = 502) (25.2% vs. 15.6%, P Ōēż 0.05, respectively). Chuang et al. [1] proposed that increased symptom severity was significantly associated with lower sexual QoL in both men and women. Another study demonstrated that the initiation, maintenance and rigidity of a penile erection were under parasympathetic and somatotopic control [7]. Some authors further concluded that the sympathetic nervous system plays an important role in the subsidence and suppression of an erection [26,27]. Cirino et al. [27] demonstrated that ╬▓3-adrenoceptors are localized mostly in the smooth muscle cells of human CC and can mediate a penile erection. In human CC, they reported that activation by a selective ╬▓3-adrenergic receptor agonist (BRL37344), elicited a cGMP-dependent, NO-independent vasorelaxation that was selectively blocked by a specific ╬▓3-receptor antagonist (SR59230A) [27].
On the basis of these findings, Wu et al. [12] conducted a clinical trial to evaluate the EF of sexually active men with OAB after 12-week treatment with mirabegron. They demonstrated that mirabegron treatment significantly improved male OAB symptoms but not male sexual function, like IIEF-5. Unexpectedly, the results revealed that 12 weeks of mirabegron treatment had a significantly negative impact on men with better baseline EF (IIEF-5 Ōēź 17; 11.7% decrease; P = 0.044). There was no improvement in EF after treatment with mirabegron, especially for the patient group with mild ED (mean IIEF-5 scores 16.3 and 17.3, respectively) in our trial which was also based in an Asian country. However, Karakus et al. [8] revealed that mirabegron may improve EF in some patients with OAB in addition to OAB-related symptoms. Elbaz et al. [9] conducted another study which compared the use of mirabegron and tolterodine for the treatment of patients with ED concomitant with LUTS. In their study, mirabegron resulted in increased improvements in urinary symptoms and the associated sexual dysfunction in patients with LUTS compared with tolterodine [9]. In these 2 studies, the recruited patients showed lower baseline IIEF compared with those in this current study, implying that mirabegron treatment may have a better treatment outcome on sexual function in patients with more severe ED. However, unlike previous studies, our results did not reveal an effect on erectile and ejaculatory functions. Although the ╬▓3-adrenoceptor agonist has been reported to facilitate smooth muscle relaxation in the resting filling phase in the penis in some studies [6,7], the ╬▓3 adrenoceptor agonist may not play a role during actual erection.
Although this present study was an RCT, it still had some limitations. The major limitation of this study was the small number of patients, especially those with different grades of ED. However, the current sample size is still enough to show the positive therapeutic effects of mirabegron on OAB symptoms and the lack of therapeutic effects on EF and ejaculatory function by both treatment approaches. The other limitation in the study was that there was no placebo to mirabegron and this was not one blind research. Another limitation of this research was that we could not offer a good explanation for the finding of a lack of improvement in EF in the combined behavioral therapy and mirabegron subgroup. We also acknowledge that prior reports of ED in mirabegron registry studies about male sexual function did not include large numbers of patients and a well-organized study design. Additional, multi-institutional RCTs with a larger number of participants, and different combination therapies, might be required to consolidate the possible effects of mirabegron on sexual parameters in patients with LUTS and OAB.
In summary, this research shows that both behavioral therapy alone and combination mirabegron and behavioral therapy improve male OAB symptoms but do not benefit EF and ejaculatory function. For male OAB, combination treatment achieved a greater improvement in OAB symptoms than behavioral therapy alone 4-week posttreatment. Future studies should prioritize determining the effect of mirabegron treatment on sexual function by combining different medications, which act on distinct signaling pathways related to penile erection.
NOTES
Grant/Fund Support
The study was funded by Taiwan Continence Society. Fund Ref ID:2020-06-A. In addition, this study was supported, in part, by a grant (Project number: V112C-20) from Taipei Veterans General Hospital to Chih-Chieh Lin.
Research Ethics
This study was performed according to the Helsinki Declaration and approved by the Research Ethics Committee or the Institutional Review Board (IRB) of the institution where the experiment was performed (IRB Reg. No. 202000790A3). A written informed consent should be obtained from all subjects. This is a randomized clinical trial registered with Clinicaltrials.gov (no. NCT04420533).
AUTHOR CONTRIBUTION STATEMENT
┬Ę Conceptualization: YCC
┬Ę Data curation: CCL, HCK, JRL, YCC
┬Ę Formal analysis: CCL, YCC
┬Ę Funding acquisition: YCC
┬Ę Methodology: YCC
┬Ę Project administration: CCL, HCK, JRL, YCC
┬Ę Visualization: JRL, YCC
┬Ę Writing - original draft: CCL
┬Ę Writing - review & editing: CCL, HCK
REFERENCES
1. Chuang YC, Liu SP, Lee KS, Liao L, Wang J, Yoo TK, et al. Prevalence of overactive bladder in China, Taiwan and South Korea: results from a cross-sectional, population-based study. Low Urin Tract Symptoms 2019;11:48-55. PMID: 28967230

2. Irwin DE, Milsom I, Hunskaar S, Reilly K, Kopp Z, Herschorn S, et al. Population-based survey of urinary incontinence, overactive bladder, and other lower urinary tract symptoms in five countries: results of the EPIC study. Eur Urol 2006;50:1306-14 discussion 1314-5. PMID: 17049716


3. Stewart WF, Van Rooyen JB, Cundiff GW, Abrams P, Herzog AR, Corey R, et al. Prevalence and burden of overactive bladder in the United States. World J Urol 2003;20:327-36. PMID: 12811491



4. Irwin DE, Milsom I, Reilly K, Hunskaar S, Kopp Z, Herschorn S, et al. Overactive bladder is associated with erectile dysfunction and reduced sexual quality of life in men. J Sex Med 2008;5:2904-10. PMID: 19090944


5. Coyne KS, Sexton CC, Thompson C, Kopp ZS, Milsom I, Kaplan SA. The impact of OAB on sexual health in men and women: results from EpiLUTS. J Sex Med 2011;8:1603-15. PMID: 21492396


6. Gur S, Peak T, Yafi FA, Kadowitz PJ, Sikka SC, Hellstrom WJ. Mirabegron causes relaxation of human and rat corpus cavernosum: could it be a potential therapy for erectile dysfunction? BJU Int 2016;118:464-74. PMID: 27124860


7. Yilmaz-Oral D, Kaya-Sezginer E, Askin D, Hamurtekin Y, Gur S. Mirabegron, a selective ╬▓3-adrenoceptor agonist causes an improvement in erectile dysfunction in diabetic rats. Exp Clin Endocrinol Diabetes 2019;129:296-302. PMID: 30978726


8. Karakus S, Musicki B, Burnett AL. Mirabegron improves erectile function in men with overactive bladder and erectile dysfunction: a 12-week pilot study. Int J Impot Res 2022;34:588-92. PMID: 34215861



9. Elbaz R, El-Assmy A, Zahran MH, Hashem A, Shokeir AA. Mirabegron for treatment of erectile dysfunction concomitant with lower urinary tract symptoms in patients with benign prostatic obstruction: a randomized controlled trial. Int J Urol 2022;29:390-6. PMID: 35043484



10. Kang TW, Kim SJ, Kim MH, Jung JH. Beta 3 adrenoreceptor agonist for the management of lower urinary tract symptoms in men with benign prostatic hyperplasia: a systematic review. Int Neurourol J 2021;25:182-91. PMID: 34610712




11. Burgio KL, Kraus SR, Johnson TM 2nd, Markland AD, Vaughan CP, Li P, et al. Effectiveness of combined behavioral and drug therapy for overactive bladder symptoms in men: a randomized clinical trial. JAMA Intern Med 2020;180:411-9. PMID: 31930360



12. Wu TH, Shen YC, Lee WC, Wang HJ, Chuang YC. Effect of mirabegron on erectile function in sexually active men with bothersome overactive bladder symptoms. J Chin Med Assoc 2020;83:55-9. PMID: 31567878


13. Lightner DJ, Gomelsky A, Souter L, Vasavada SP. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU Guideline Amendment 2019. J Urol 2019;202:558-63. PMID: 31039103


14. Nambiar AK, Arlandis S, Bo K, Cobussen-Boekhorst H, Costantini E, de Heide M, et al. European Association of Urology guidelines on the diagnosis and management of female non-neurogenic lower urinary tract symptoms. Part 1: diagnostics, overactive bladder, stress urinary incontinence, and mixed urinary incontinence. Eur Urol 2022;82:49-59. PMID: 35216856


15. Gacci M, Sakalis VI, Karavitakis M, Cornu JN, Gratzke C, Herrmann TRW, et al. European Association of Urology guidelines on male urinary incontinence. Eur Urol 2022;82:387-98. PMID: 35697561


16. Goode PS, Burgio KL, Johnson TM, Clay OJ, Roth DL, Markland AD, et al. Behavioral therapy with or without biofeedback and pelvic floor electrical stimulation for persistent postprostatectomy incontinence: a randomized controlled trial. JAMA 2011;305:151-9. PMID: 21224456



17. Burgio KL, Goode PS, Johnson TM, Hammontree L, Ouslander JG, Markland AD, et al. Behavioral versus drug treatment for overactive bladder in men: the Male Overactive Bladder Treatment in Veterans (MOTIVE) Trial. J Am Geriatr Soc 2011;59:2209-16. PMID: 22092152


18. Mattiasson A, Masala A, Morton R, Bolodeoku J, Group SS. Efficacy of simplified bladder training in patients with overactive bladder receiving a solifenacin flexible-dose regimen: results from a randomized study. BJU Int 2010;105:1126-35. PMID: 19818077


19. Klutke CG, Burgio KL, Wyman JF, Guan Z, Sun F, Berriman S, et al. Combined effects of behavioral intervention and tolterodine in patients dissatisfied with overactive bladder medication. J Urol 2009;181:2599-607. PMID: 19375110


20. Burgio KL, Locher JL, Goode PS. Combined behavioral and drug therapy for urge incontinence in older women. J Am Geriatr Soc 2000;48:370-4. PMID: 10798461


21. Mattiasson A, Blaakaer J, H├Ėye K, Wein AJ; Tolterodine Scandinavian Study Group. Simplified bladder training augments the effectiveness of tolterodine in patients with an overactive bladder. BJU Int 2003;91:54-60. PMID: 12614251


22. Burgio KL, Kraus SR, Menefee S, Borello-France D, Corton M, Johnson HW, et al. Behavioral therapy to enable women with urge incontinence to discontinue drug treatment: a randomized trial. Ann Intern Med 2008;149:161-9. PMID: 18678843



23. Lin CT, Chiang BJ, Liao CH. Perspectives of medical treatment for overactive bladder. Urol Sci 2020;31:91-8. 
24. Sebastianelli A, Russo GI, Kaplan SA, McVary KT, Moncada I, Gravas S, et al. Systematic review and meta-analysis on the efficacy and tolerability of mirabegron for the treatment of storage lower urinary tract symptoms/overactive bladder: comparison with placebo and tolterodine. Int J Urol 2018;25:196-205. PMID: 29205506



25. Kuo HC, Lee KS, Na Y, Sood R, Nakaji S, Kubota Y, et al. Results of a randomized, double-blind, parallel-group, placebo- and active-controlled, multicenter study of mirabegron, a beta3-adrenoceptor agonist, in patients with overactive bladder in Asia. Neurourol Urodyn 2015;34:685-92. PMID: 25130281

26. Diederichs W, Stief CG, Benard F, Bosch R, Lue TF, Tanagho EA. The sympathetic role as an antagonist of erection. Urol Res 1991;19:123-6. PMID: 1853514



27. Cirino G, Sorrentino R, di Villa Bianca RdE, Popolo A, Palmieri A, Imbimbo C, et al. Involvement of ╬▓3-adrenergic receptor activation via cyclic GMP- but not NO-dependent mechanisms in human corpus cavernosum function. Proc Natl Acad Sci U S A 2003;100:5531-6. PMID: 12707413



Table┬Ā1.
Baseline characteristics of all participants
Table┬Ā2.
Changes in OABSS, urodynamic parameters, PPBC, voiding diary, and IPSS from baseline to weeks 4 and 12
| Variable |
Data at each timepoint |
Difference compared to baseline |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Behavior (n=36) | P-valuea) | Combination (n=65) | P-valuea) | P-valueb) | Behavior (n=36) | Combination (n=65) | P-valueb) | |||
| OABSS | ||||||||||
| Q1: Daytime frequency | ||||||||||
| Baseline | 1.0 ┬▒ 0.5 | 0.9 ┬▒ 0.6 | 0.389 | |||||||
| 4 Weeks | 1.0 ┬▒ 0.8 | 0.881 | 0.6 ┬▒ 0.5 | 0.001 | 0.002 | 0.0 ┬▒ 0.7 | -0.3 ┬▒ 0.6 | 0.010 | ||
| 12 Weeks | 0.8 ┬▒ 0.5 | 0.120 | 0.7 ┬▒ 0.5 | 0.007 | 0.344 | -0.2 ┬▒ 0.6 | -0.2 ┬▒ 0.6 | 0.775 | ||
| Q2: Nocturia | ||||||||||
| Baseline | 2.2 ┬▒ 0.9 | 2.1 ┬▒ 0.9 | 0.612 | |||||||
| 4 Weeks | 1.9 ┬▒ 1.1 | 0.155 | 1.5 ┬▒ 0.9 | < 0.001 | 0.013 | -0.3 ┬▒ 0.8 | -0.6 ┬▒ 0.9 | 0.081 | ||
| 12 Weeks | 1.7 ┬▒ 1.2 | 0.022 | 1.4 ┬▒ 0.9 | < 0.001 | 0.263 | -0.5 ┬▒ 0.8 | -0.7 ┬▒ 0.9 | 0.449 | ||
| Q3: Urgency | ||||||||||
| Baseline | 3.5 ┬▒ 1.4 | 3.6 ┬▒ 1.1 | 0.884 | |||||||
| 4 Weeks | 3.0 ┬▒ 1.8 | 0.153 | 2.4 ┬▒ 1.8 | < 0.001 | 0.065 | -0.5 ┬▒ 1.5 | -1.1 ┬▒ 1.6 | 0.034 | ||
| 12 Weeks | 2.8 ┬▒ 2.0 | 0.035 | 2.2 ┬▒ 1.8 | < 0.001 | 0.162 | -0.6 ┬▒ 1.6 | -1.2 ┬▒ 1.7 | 0.069 | ||
| Q4: Urinary incontinence | ||||||||||
| Baseline | 1.5 ┬▒ 1.6 | 1.3 ┬▒ 1.5 | 0.471 | |||||||
| 4 Weeks | 0.9 ┬▒ 1.3 | 0.046 | 0.5 ┬▒ 1.1 | < 0.001 | 0.086 | -0.6 ┬▒ 1.2 | -0.8 ┬▒ 1.4 | 0.437 | ||
| 12 Weeks | 1.1 ┬▒ 1.4 | 0.234 | 0.7 ┬▒ 1.2 | 0.004 | 0.137 | -0.5 ┬▒ 1.2 | -0.6 ┬▒ 1.3 | 0.877 | ||
| Total score | ||||||||||
| Baseline | 8.1 ┬▒ 2.7 | 7.8 ┬▒ 2.5 | 0.615 | |||||||
| 4 Weeks | 6.5 ┬▒ 2.9 | 0.011 | 5.0 ┬▒ 2.9 | < 0.001 | 0.005 | -1.6 ┬▒ 2.2 | -2.8 ┬▒ 2.7 | 0.010 | ||
| 12 Weeks | 6.0 ┬▒ 3.3 | 0.002 | 5.0 ┬▒ 3.0 | < 0.001 | 0.104 | -1.9 ┬▒ 2.7 | -2.7 ┬▒ 2.9 | 0.194 | ||
| Urodynamic parameters | ||||||||||
| Bladder capacity (mL) | ||||||||||
| Baseline | 283.7 ┬▒ 122.0 | 252.5 ┬▒ 118.8 | 0.136 | |||||||
| 4 Weeks | 300.4 ┬▒ 126.5 | 0.516 | 293.2 ┬▒ 132.6 | 0.030 | 0.770 | 14.8 ┬▒ 124.1 | 49.6 ┬▒ 122.8 | 0.135 | ||
| 12 Weeks | 319.3 ┬▒ 120.1 | 0.199 | 295.3 ┬▒ 131.3 | 0.041 | 0.388 | 24.3 ┬▒ 109.0 | 41.7 ┬▒ 111.8 | 0.472 | ||
| Qmax (mL/sec) | ||||||||||
| Baseline | 17.5 ┬▒ 8.4 | 15.5 ┬▒ 7.6 | 0.158 | |||||||
| 4 Weeks | 17.7 ┬▒ 7.6 | 0.899 | 16.6 ┬▒ 6.9 | 0.351 | 0.411 | 0.0 ┬▒ 4.8 | 1.5 ┬▒ 6.1 | 0.160 | ||
| 12 Weeks | 17.9 ┬▒ 6.8 | 0.802 | 16.7 ┬▒ 8.3 | 0.346 | 0.476 | 0.2 ┬▒ 4.2 | 0.5 ┬▒ 5.5 | 0.800 | ||
| Residual urine (mL) | ||||||||||
| Baseline | 30.9 ┬▒ 28.0 | 26.3 ┬▒ 23.8 | 0.291 | |||||||
| 4 Weeks | 34.8 ┬▒ 35.8 | 0.549 | 37.1 ┬▒ 39.7 | 0.019 | 0.750 | 6.2 ┬▒ 35.3 | 10.7 ┬▒ 39.7 | 0.522 | ||
| 12 Weeks | 41.5 ┬▒ 29.7 | 0.132 | 29.8 ┬▒ 28.0 | 0.489 | 0.062 | 10.4 ┬▒ 30.7 | 5.4 ┬▒ 27.8 | 0.431 | ||
| PPBC | ||||||||||
| Baseline | 2.4 ┬▒ 1.2 | 2.6 ┬▒ 1.2 | 0.329 | |||||||
| 4 Weeks | 1.9 ┬▒ 1.3 | 0.074 | 1.6 ┬▒ 1.1 | < 0.001 | 0.145 | -0.5 ┬▒ 1.0 | -1.0 ┬▒ 1.2 | 0.020 | ||
| 12 Weeks | 1.5 ┬▒ 1.1 | 0.001 | 1.4 ┬▒ 1.0 | < 0.001 | 0.740 | -0.9 ┬▒ 1.2 | -1.2 ┬▒ 1.1 | 0.354 | ||
| Voiding diary | ||||||||||
| Daytime frequency | ||||||||||
| Baseline | 9.1 ┬▒ 2.5 | 8.8 ┬▒ 3.4 | 0.556 | |||||||
| 4 Weeks | 8.4 ┬▒ 2.2 | 0.129 | 8.1 ┬▒ 2.4 | 0.082 | 0.484 | -0.6 ┬▒ 1.8 | -0.9 ┬▒ 2.4 | 0.474 | ||
| 12 Weeks | 8.1 ┬▒ 2.1 | 0.038 | 7.9 ┬▒ 2.3 | 0.044 | 0.715 | -0.6 ┬▒ 1.8 | -1.3 ┬▒ 2.7 | 0.198 | ||
| Nocturia | ||||||||||
| Baseline | 1.7 ┬▒ 1.3 | 1.5 ┬▒ 1.0 | 0.184 | |||||||
| 4 Weeks | 1.5 ┬▒ 1.1 | 0.316 | 1.2 ┬▒ 0.9 | 0.063 | 0.128 | -0.3 ┬▒ 0.9 | -0.3 ┬▒ 0.7 | 0.795 | ||
| 12 Weeks | 1.4 ┬▒ 1.0 | 0.161 | 1.2 ┬▒ 0.8 | 0.041 | 0.239 | -0.3 ┬▒ 0.7 | -0.3 ┬▒ 0.8 | 0.815 | ||
| FBC (mL) | ||||||||||
| Baseline | 382.8 ┬▒ 135.1 | 362.1 ┬▒ 121.2 | 0.359 | |||||||
| 4 Weeks | 394.5 ┬▒ 166.2 | 0.711 | 374.5 ┬▒ 109.4 | 0.496 | 0.416 | 12.7 ┬▒ 95.9 | 9.3 ┬▒ 93.0 | 0.848 | ||
| 12 Weeks | 387.7 ┬▒ 145.9 | 0.884 | 364.5 ┬▒ 138.1 | 0.907 | 0.449 | 9.5 ┬▒ 84.0 | 1.3 ┬▒ 100.3 | 0.695 | ||
| IPSS | ||||||||||
| Voiding subscore | ||||||||||
| Baseline | 5.9 ┬▒ 4.8 | 5.8 ┬▒ 5.0 | 0.981 | |||||||
| 4 Weeks | 4.0 ┬▒ 3.7 | 0.026 | 3.4 ┬▒ 3.8 | < 0.001 | 0.447 | -2.1 ┬▒ 3.7 | -2.7 ┬▒ 4.4 | 0.409 | ||
| 12 Weeks | 4.0 ┬▒ 3.9 | 0.046 | 3.7 ┬▒ 4.3 | 0.003 | 0.740 | -3.1 ┬▒ 4.9 | -2.7 ┬▒ 4.5 | 0.721 | ||
| Storage subscore | ||||||||||
| Baseline | 6.5 ┬▒ 3.3 | 6.6 ┬▒ 2.7 | 0.890 | |||||||
| 4 Weeks | 5.6 ┬▒ 3.0 | 0.169 | 4.7 ┬▒ 2.8 | < 0.001 | 0.067 | -1.0 ┬▒ 2.3 | -1.9 ┬▒ 2.6 | 0.036 | ||
| 12 Weeks | 4.9 ┬▒ 2.8 | 0.019 | 4.4 ┬▒ 2.5 | < 0.001 | 0.376 | -1.3 ┬▒ 3.4 | -2.3 ┬▒ 2.8 | 0.118 | ||
| Total score | ||||||||||
| Baseline | 12.4 ┬▒ 6.4 | 12.4 ┬▒ 6.3 | 0.971 | |||||||
| 4 Weeks | 9.6 ┬▒ 5.5 | 0.023 | 8.2 ┬▒ 5.5 | < 0.001 | 0.149 | -3.1 ┬▒ 4.5 | -4.6 ┬▒ 5.4 | 0.101 | ||
| 12 Weeks | 8.9 ┬▒ 5.9 | 0.008 | 8.2 ┬▒ 5.5 | < 0.001 | 0.532 | -4.4 ┬▒ 6.9 | -5.1 ┬▒ 5.6 | 0.634 | ||
| Nocturia score | ||||||||||
| Baseline | 2.4 ┬▒ 1.2 | 2.3 ┬▒ 1.2 | 0.557 | |||||||
| 4 Weeks | 2.0 ┬▒ 1.2 | 0.114 | 1.5 ┬▒ 1.1 | < 0.001 | 0.015 | -0.4 ┬▒ 1.0 | -0.7 ┬▒ 1.1 | 0.163 | ||
| 12 Weeks | 1.7 ┬▒ 1.1 | 0.004 | 1.5 ┬▒ 1.0 | < 0.001 | 0.373 | -0.8 ┬▒ 1.2 | -0.8 ┬▒ 1.0 | 0.874 | ||
| Quality of life | ||||||||||
| Baseline | 3.6 ┬▒ 1.4 | 3.8 ┬▒ 1.5 | 0.557 | |||||||
| 4 Weeks | 3.0 ┬▒ 1.4 | 0.036 | 2.6 ┬▒ 1.5 | < 0.001 | 0.072 | -0.7 ┬▒ 1.5 | -1.2 ┬▒ 1.6 | 0.067 | ||
| 12 Weeks | 2.6 ┬▒ 1.4 | < 0.001 | 2.4 ┬▒ 1.4 | < 0.001 | 0.589 | -1.2 ┬▒ 1.6 | -1.3 ┬▒ 1.5 | 0.728 | ||
Table┬Ā3.
Changes in sexual functions from baseline to weeks 4 and 12
| Variable |
Data at each timepoint |
Difference compared to baseline |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Behavior (n=36) | P-valuea) | Combination (n=65) | P-valuea) | P-valueb) | Behavior (n=36) | Combination (n=65) | P-valueb) | |||
| IIEF-5 | ||||||||||
| Baseline | 16.3 ┬▒ 5.3 | 17.3 ┬▒ 4.6 | 0.270 | |||||||
| 4 Weeks | 16.1 ┬▒ 5.6 | 0.855 | 17.5 ┬▒ 4.7 | 0.708 | 0.131 | -0.4 ┬▒ 2.5 | 0.3 ┬▒ 3.0 | 0.167 | ||
| 12 Weeks | 17.1 ┬▒ 6.1 | 0.560 | 17.7 ┬▒ 4.8 | 0.560 | 0.561 | 0.6 ┬▒ 2.9 | 0.3 ┬▒ 3.5 | 0.606 | ||
| MESH EjD SF | ||||||||||
| Q1: Frequency | ||||||||||
| Baseline | 4.1 ┬▒ 1.1 | 4.1 ┬▒ 1.2 | 0.759 | |||||||
| 4 Weeks | 4.1 ┬▒ 1.2 | 0.967 | 4.1 ┬▒ 1.2 | 0.855 | 0.860 | 0.0 ┬▒ 0.6 | 0.1 ┬▒ 0.8 | 0.663 | ||
| 12 Weeks | 3.8 ┬▒ 1.4 | 0.200 | 4.0 ┬▒ 1.2 | 0.883 | 0.356 | -0.3 ┬▒ 1.0 | 0.1 ┬▒ 0.8 | 0.063 | ||
| Q2: Strength | ||||||||||
| Baseline | 3.2 ┬▒ 1.2 | 3.2 ┬▒ 1.2 | 0.883 | |||||||
| 4 Weeks | 3.3 ┬▒ 1.3 | 0.879 | 3.4 ┬▒ 1.3 | 0.184 | 0.483 | 0.0 ┬▒ 1.1 | 0.2 ┬▒ 1.2 | 0.430 | ||
| 12 Weeks | 3.4 ┬▒ 1.4 | 0.622 | 3.3 ┬▒ 1.2 | 0.512 | 0.871 | 0.1 ┬▒ 0.9 | 0.1 ┬▒ 1.5 | 0.953 | ||
| Q3: Volume of semen | ||||||||||
| Baseline | 3.2 ┬▒ 1.1 | 3.2 ┬▒ 1.3 | 0.814 | |||||||
| 4 Weeks | 3.2 ┬▒ 1.3 | 0.795 | 3.3 ┬▒ 1.2 | 0.405 | 0.459 | -0.0 ┬▒ 0.8 | 0.1 ┬▒ 1.1 | 0.499 | ||
| 12 Weeks | 3.4 ┬▒ 1.4 | 0.656 | 3.3 ┬▒ 1.3 | 0.754 | 0.694 | 0.2 ┬▒ 1.0 | 0.1 ┬▒ 1.4 | 0.616 | ||
| Q1ŌĆōQ3 total scores | ||||||||||
| Baseline | 10.6 ┬▒ 2.8 | 10.4 ┬▒ 3.1 | 0.786 | |||||||
| 4 Weeks | 10.3 ┬▒ 3.6 | 0.724 | 10.7 ┬▒ 3.5 | 0.537 | 0.530 | -0.2 ┬▒ 2.5 | 0.2 ┬▒ 2.8 | 0.448 | ||
| 12 Weeks | 10.5 ┬▒ 3.7 | 0.913 | 10.7 ┬▒ 3.4 | 0.649 | 0.805 | 0.0 ┬▒ 2.5 | 0.3 ┬▒ 3.3 | 0.631 | ||
| Q4: Bother/satisfaction | ||||||||||
| Baseline | 1.5 ┬▒ 1.3 | 1.4 ┬▒ 1.4 | 0.669 | |||||||
| 4 Weeks | 1.5 ┬▒ 1.6 | 0.788 | 1.4 ┬▒ 1.3 | 0.921 | 0.533 | 0.1 ┬▒ 1.0 | 0.0 ┬▒ 1.1 | 0.734 | ||
| 12 Weeks | 1.5 ┬▒ 1.6 | 0.904 | 1.2 ┬▒ 1.3 | 0.385 | 0.263 | -0.0 ┬▒ 1.1 | -0.3 ┬▒ 1.3 | 0.341 | ||
Values are presented as mean┬▒standard deviation.
IIEF-5, abridged 5-item version of the International Index of Erectile Function; MSHQ-EjD SF, Male Sexual Health Questionnaire-Ejaculatory Domain Short Form.
MSHQ-EjD SF: Q1: How often have you been able to ejaculate or ŌĆ£cumŌĆØ when having sexual activity? Q2: How would you rate the strength or force of your ejaculation? Q3: How would you rate the amount or volume of semen or fluid when you ejaculate? Q4: If you have had any ejaculation difficulties or have been unable to ejaculate, have you been bothered by this?