• HIGHLIGHTS
- We explored the possibility of transcutaneous electrical stimulation of the abdominal wall, ear, and ankle for the treatment of overactive bladder.
INTRODUCTION
Overactive bladder (OAB) is a global health concern affecting millions of people, with an estimated prevalence of 11.8% [1]. This condition is characterized by a symptom complex that includes frequency and urgency, with or without urgency incontinence [2]. Treatment options for OAB include behavioral modifications and medication [3]. Additionally, physiotherapy, with or without biofeedback, can be provided. Percutaneous tibial nerve stimulation is another treatment option, although it necessitates frequent clinic visits for repeated stimulation to maintain the treatment’s effectiveness. Other options include botulinum toxin A injections into the detrusor muscle or sacral neuromodulation. Notably, guidelines from the European Association of Urology highlight percutaneous tibial nerve stimulation and sacral neuromodulation as neuromodulation treatment options. Despite the range of available treatments, many patients remain untreated. Sacral neuromodulation, for instance, is offered to a limited number of patients due to financial constraints and a shortage of trained physicians. Consequently, many patients are unable to access this treatment. Some individuals are reluctant to opt for percutaneous tibial nerve stimulation, due to the necessity of frequent hospital visits. Therefore, a pressing need exists for more widely available and accessible treatment methods.
One such potential treatment is noninvasive transcutaneous electrical stimulation, which can be administered at home. This form of electrical stimulation does not necessitate the implantation of an electrode, thereby eliminating the need for highly skilled surgeons and expensive implantable devices. Research has demonstrated comparable efficacy between transcutaneous and percutaneous tibial nerve stimulation [4,5]. The advantage of transcutaneous tibial nerve stimulation lies in its superior comfort level, which stems from its less invasive nature [6,7]. Other peripheral nerves may serve as promising alternative targets for peripheral nerve stimulation in treating bladder dysfunction. The impact of neuromodulation of the peroneal and saphenous nerves on bladder function has been previously investigated [8-10]. We hypothesize that in addition to the ankle, the abdominal wall and the ear could serve as effective targets for transcutaneous electrical stimulation in the treatment of OAB. This proposition is based on the ear’s innervation by the vagus nerve, which maintains neural connections to the bladder. The afferent fibers of the pelvic and pudendal nerves are located within the dorsal root ganglia at the sacral level, while those of the hypogastric nerve are found at the rostral lumbar level [11]. The latter dorsal root ganglia also house afferent fibers from the abdominal wall. Therefore, we propose that transcutaneous electrical stimulation of both the ear and the abdominal wall could modulate bladder activity, as they share neural control pathways with the urinary bladder.
The target of electrical stimulation is not the only critical factor; stimulation parameters also play an important role. The effectiveness of electrical stimulation is known to hinge on parameters such as frequency and amplitude [12-14]. We hypothesize that different stimulation frequencies may have varying impacts on bladder contraction. The optimization of these stimulation parameters is crucial for maximizing the treatment effect and extending the battery life of the simulator. Consequently, in this study, we not only explored various targets of transcutaneous electrical stimulation but also examined multiple frequencies.
In rats, detrusor overactivity can be induced by infusing acetic acid into the bladder [15]. We employed this method to simulate detrusor overactivity in these animals, while concurrently applying electrical stimulation to assess its impact on bladder contraction. As such, we examined the efficacy of low-frequency (20 Hz) transcutaneous stimulation of the abdominal wall, ear, and tibial nerve. In addition, we compared low-frequency and high-frequency (in the kilohertz range) transcutaneous stimulation of the tibial nerve, to ascertain whether varying frequencies of transcutaneous stimulation have differential effects on bladder function.
MATERIALS AND METHODS
Animals
We adhered to the guidelines of the local Animal Experiment Committee for all animal experiments. The animals were housed under standard laboratory conditions, in compliance with European laws, guidelines, and policies pertaining to animal experimentation, housing, and care. The animals were kept in a facility with a 12-hour light/dark cycle and given unrestricted access to food and water. Three female Sprague-Dawley murine pathogen-free rats (mean weight, 230 ± 22 g) were utilized. The mean age of the rats was 59 ± 13 days.
Procedures
The rats were anesthetized using a solution of ketamine (90 mg/kg) and xylazine (10 mg/kg) [16]. This solution, which contained 10% ketamine and 2% xylazine, was injected intraperitoneally. To prolong the anesthesia, we also administered 0.13 mL of a 50% urethane solution intraperitoneally [16]. The dosages of ketamine and xylazine were determined based on the rat’s weight, while the urethane dosage remained constant across animals. Following the administration of anesthesia, each rat was positioned on a heated pad, with the ventral side facing upwards. For transcutaneous tibial nerve stimulation, we placed 2 clip electrodes just above the medial malleolus, with 2 mm between them. For abdominal wall stimulation, the electrodes were positioned 1 centimeter below the umbilicus and spaced 2 mm apart. For ear stimulation, the electrodes were placed on the auricle, again with a 2-mm separation.
Electrical Stimulation
A ventral midline incision was performed to expose the urinary bladder. Saline was utilized to maintain tissue moisture throughout the procedure. A 23G catheter was inserted into the bladder dome and connected to a disposable pressure transducer and an infusion pump. The pump filled the bladder at a rate of 0.06 mL/min. Initially, the bladder of each rat was filled with saline (0.9% sodium chloride), followed by 0.5% acetic acid to simulate detrusor overactivity [15]. Intravesical pressure was gauged using a Statham SP1400 blood pressure monitor (Gould Inc., Oxnard, CA, USA). All experiments were conducted at room temperature. Two stimulators (Hameg HM 8130 and SI-Heidelberg) were employed to deliver rectangular monophasic (low-frequency) and rectangular biphasic (kilohertz-frequency) stimulation. Voltage stimulation was administered at frequencies of 20 Hz, 1 kHz, 3 kHz, 10 kHz, and 20 kHz at an amplitude of 10–20 V, with unilateral stimulation of the ear and tibial nerve. Each stimulation session was followed by a 2-minute rest period, during which no electrical stimulation was applied but bladder infusion continued. Each rat underwent stimulation twice. At the conclusion of the experiment, the rat was euthanized with a lethal dose of potassium chloride, injected into the heart.
Signal Processing
Pressure and stimulation signals were captured using LabVIEW (National Instruments Corporation, Austin, TX, USA) at a sampling frequency of 25 kHz. These signals were processed and analyzed with a custom script for MATLAB (MathWorks, Inc., Natick, MA, USA).
Statistical Analysis
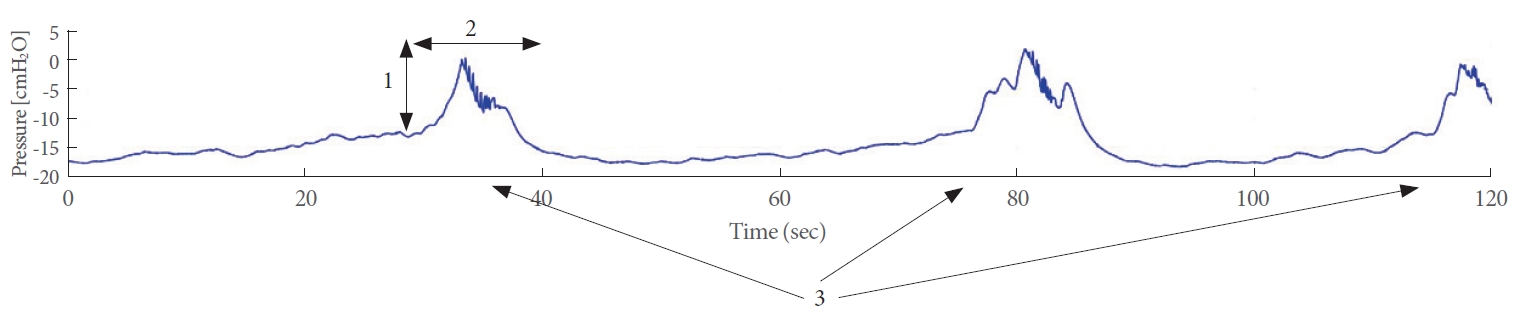
The primary outcome measures of interest included (1) the change in intravesical pressure from the onset to the peak of contraction, (2) the average duration of contraction, and (3) the number of contractions within a 2-minute period. These outcome variables are depicted in Fig. 1. The statistical analysis was conducted using SPSS (SPSS Inc., Chicago, IL, USA). All findings are reported as the mean ± standard error of the mean. To compare the results, we utilized the dependent t-test. A P-value of less than 0.05 was considered to indicate statistical significance.
RESULTS
Transcutaneous Stimulation of the Abdomen, Ear, and Tibial Nerve
Transcutaneous stimulation was applied to the abdominal wall, ear, and ankle using a 20-Hz stimulus for 2 minutes. Simultaneously, intravesical pressure was measured. For each stimulation trial, we calculated the average pressure change during bladder contraction, the average duration of these contractions, and the number of bladder contractions.
Saline
During all forms of transcutaneous stimulation, while the bladder was being infused with saline, no significant differences were observed in bladder pressure, duration, and volume when compared to the condition without stimulation (Fig. 2A–C).
Acetic acid
When the bladder was infused with acetic acid, stimulation of the ear, abdominal wall, and tibial nerve resulted in an increased pressure change during contractions compared to instances without stimulation (Fig. 2D). However, the pressure change during contractions with ear stimulation was not significantly higher than the pressure changes during contractions without stimulation (P = 0.374). The duration of bladder contractions also lengthened with ear stimulation, but this increase did not significantly differ from the duration of contractions without stimulation (P = 0.224) (Fig. 2E). Moreover, the number of contractions within a 2-minute period appeared to decrease with stimulation of the ear, abdominal wall, and tibial nerve, but this change was not statistically significant.
Transcutaneous Kilohertz Stimulation of the Tibial Nerve at Multiple Frequencies
Saline
During the infusion of the bladder with saline, transcutaneous tibial nerve stimulation across all examined frequencies (20 Hz to 20 kHz) did not significantly influence the pressure alterations during bladder contractions. However, it did reduce the duration of these contractions (Fig. 3A, B). Stimulation of the tibial nerve at 20 Hz resulted in a decrease in the number of contractions, while stimulation within the kilohertz range led to an increase in the number of contractions (Fig. 3C) over a 2-minute interval.
Acetic acid
When the bladder was infused with acetic acid, tibial nerve stimulation at 20 Hz led to an increase in both the pressure change during contractions and the duration of these contractions (Fig. 3D, E). No significant difference was observed in the pressure change during contractions between 1-kHz and 20-Hz stimulation (P = 0.072) (Fig. 3D). The duration of contractions was significantly shorter during 1-kHz and 3-kHz stimulation relative to 20-Hz stimulation (P = 0.025 and P = 0.044, respectively) (Fig. 3E). However, no significant difference was observed in the duration of contractions between 10-kHz and 20-Hz stimulation (P = 0.085) (Fig. 3E). Over 2 minutes of stimulation, the number of contractions decreased during 20-Hz stimulation, but no consistent pattern was associated with stimulation in the kilohertz range (Fig. 3F).
DISCUSSION
In this pilot study, we demonstrated that the effects of transcutaneous 20-Hz stimulation of the ear and abdomen differed from those of transcutaneous tibial nerve stimulation in a rat model of detrusor overactivity. Specifically, differences were found in the pressure changes during contraction and contraction duration. Transcutaneous stimulation of the ear, abdomen, and tibial nerve reduced the number of contractions. The impact of transcutaneous tibial nerve stimulation was found to be frequency-dependent. In general, transcutaneous tibial nerve stimulation in the kilohertz range produced different results in the evaluated outcome parameters compared to stimulation at 20 Hz.
The impact of tibial nerve stimulation on bladder function can be elucidated through the neuroanatomical correlation with nerve fibers that innervate the bladder. The tibial nerve originates in the L4-S3 nerve roots, which also provide innervation to the pelvic floor, including the urethral sphincters and the detrusor muscle [17]. Tibial nerve stimulation at a frequency of 20-Hz influences the frequency of bladder contractions by inhibiting spinal micturition centers.
The influence of ear stimulation on bladder function can be explained through the activation of the vagus nerve, which exercises parasympathetic control over various organs such as the heart and digestive tract. The ear extends afferents to the vagus nerve, which in turn innervates the bladder. This connection was suggested by a prior study in which esophageal distension was associated with an increase in micturition frequency in rats [18]. The impact on the bladder was nullified by vagotomy, implying that the vagus nerve innervates the bladder. In another previous study conducted on rats, dissection of the vagus nerve led to an increase in intravesical pressure, extended the interval between bladder contractions, augmented functional bladder capacity, and improved the compliance of the detrusor muscle [19]. Furthermore, electrical stimulation of the vagus nerve at a frequency of 20 Hz has been found to decrease intravesical pressure in dogs [20].
The impact of transcutaneous abdominal wall stimulation on bladder function could potentially be attributed to the common innervation of the abdominal wall and the bladder. The abdominal wall surrounding the umbilicus receives innervation from thoracic segments, rostral lumbar segments, and the iliohypogastric nerve [21]. These nerves, along with the afferents of the parietal peritoneum that travel to the dorsal root ganglia, may explain the effects of transcutaneous stimulation of the abdominal wall on intravesical pressure and bladder contractions. This is because the rostral lumbar dorsal root ganglia also house afferents from the bladder [11].
Neuromodulation in the treatment of OAB is hypothesized to act through the modulation of afferent pathways. These include spinal micturition centers and forebrain areas [8,22-24]. This proposed mechanism of action accounts for the effects of both sacral neuromodulation and electrical stimulation of peripheral nerves, such as the tibial nerve. We hypothesize that the impact of sacral neuromodulation is more robust than that of peripheral transcutaneous nerve stimulation. This is due to the greater number of nerve fibers stimulated and the continuous nature of sacral neuromodulation, as opposed to the intermittent stimulation of peripheral nerves. Furthermore, the proximity of the electrode to the nerve exerts an influence, contributing to the superior efficacy of sacral neuromodulation. However, both transcutaneous and invasive neuromodulation can provide benefits to different patients with OAB. Transcutaneous stimulation may be an effective and appropriate treatment for patients who are not suitable for surgery or who reside in resource-limited areas. In contrast, invasive neuromodulation can be offered to patients with access to a specialized urological clinic and to those with a reasonable life expectancy and good overall health.
A previous study revealed no acute effects of sacral neuromodulation on urodynamic parameters in patients with OAB [25]. In contrast, the present study indicated that bladder parameters were impacted by transcutaneous electrical stimulation at several sites. This difference in results could potentially be attributed to the variations in stimulation parameter settings. Therefore, this preliminary study indicates that different stimulation frequencies may influence bladder function in diverse ways. At present, electrical nerve stimulation is typically performed at a fixed frequency. Minimal attempts have been made to optimize stimulation parameters in order to enhance efficacy and extend the device’s battery life. This preliminary study suggests that further research to refine neuromodulation modalities and stimulation parameter settings could alleviate patients’ symptom burden and decrease the expenses associated with treatment.
The limitations of the present study include its small sample size and the absence of a direct comparison between transcutaneous and percutaneous tibial nerve stimulation. Subsequent studies should incorporate a direct comparison and possibly extend the scope to include sacral neuromodulation.
In conclusion, this preliminary study has generated the following hypotheses for future research: (1) transcutaneous low-frequency (20 Hz) stimulation of the abdominal wall and ear may offer treatment alternatives for OAB, and (2) transcutaneous stimulation of the tibial nerve within the kilohertz range may present an effective treatment for OAB.