Time-Dependent Changes in the Bladder Muscle Metabolome After Traumatic Spinal Cord Injury in Rats Using Metabolomics
Article information
Abstract
Purpose
The main treatment options of neurogenic bladder remains catheterization and long-term oral medications. Metabolic interventions have shown good therapeutic results in many diseases. To date, no studies have characterized the metabolites of the detrusor muscle during neurogenic bladder. Using metabolomics, new muscle metabolomic signatures were identified to reveal the temporal metabolic profile of muscle during disease progression.
Methods
We used 42 Sprague-Dawley rats (200±20 g, males) for T10 segmental spinal cord injury modeling and collected detrusor tissue and performed nontargeted metabolomics after sham surgery, 30-minute, 6-hour, 12-hour, 24-hour, 5-day, and 2-week postmodelling, to identify the dysregulated metabolic pathways and key metabolites.
Results
By comparing mzCloud, mzVault, MassList, we identified a total of 1,271 metabolites and enriched a total of 12 metabolism-related pathways with significant differences (P<0.05) based on Kyoto Encyclopedia of Genes and Genomes analysis. Metabolites in several differential metabolic pathways such as ascorbate and aldarate metabolism, Steroid hormone biosynthesis, and carbon metabolism are altered in a regular manner before and after ridge shock.
Conclusions
Our study is the first time-based metabolomic study of rat forced urinary muscle after traumatic spinal cord injury, and we identified multiple differential metabolic pathways during injury that may improve long-term management strategies for neurogenic bladder and reduce costs in long-term treatment.
• HIGHLIGHTS
- The incidence of neurogenic bladder increases gradually after spinal cord injury.
- The crosstalk of metabolites can aggravate the occurrence of injury; Metabolic intervention therapy has achieved some success, but lacks comprehensive research.
- Regular changes of multiple metabolic pathways during disease formation.
- Metabolomics research provides a basis for metabolic intervention therapy and possible biomarkers.
INTRODUCTION
Hydronephrosis and urinary tract infections due to neurogenic bladder are important contributors to mortality in traumatic spinal cord injury (TSCI) survivors [1,2]. However, the clinical manifestations of neurogenic bladder are complex, and the pathological process of the disease has undergone a transition from neural dominance to the joint involvement of nerve detrusor muscle. So far, the process of detrusor injury is still being studied.
The traditional treatments for neurogenic bladder in patients with TSCI are long-term care and drug intervention [3,4]. However, the life expectancy of patients continues to decline [2].
The new concept of neurogenic bladder treatment is to delay tissue damage through chronic disease management, and it is feasible to partially restore tissue function. Metabolic intervention has shown high efficacy in animal models. Metabolic intervention is a targeted intervention treatment for metabolic disorders based on the results of metabolomics research as the core, in order to regulate and alleviate the dominant pathophysiological mechanisms in the occurrence and development of diseases. Several studies have suggested that metabolic intervention can ameliorate spinal cord injury and restore neurological function. For example, selenium-doped carbon quantum dots can improve the recovery of urinary function and reduce the degree of bladder tissue fibrosis, bladder endometriosis, and cell vacuolization in rats after TSCI [5]. NADPH oxidase 2 can promote the regeneration and functional recovery of sensory axons after TSCI by scavenging reactive oxygen species and enhancing redox signaling [6]. Studie shows that serine is involved in the remodeling of lipid metabolism. Abnormal serine homeostasis is related to diabetes peripheral neuropathy, and serine supplementation can slow down neuropathy [7]. Another notable example of metabolic intervention is s-nitrosoglutathione (GSNO) treatment, which promotes neurorepair to restore bladder function and improve the recovery of locomotor function in rats after TSCI [8]. Since glutathione can reduce fibrosis in inflamed tissues by inhibiting ferroptosis [9,10], it is speculated that timely administration of GSNO can greatly attenuate bladder muscle damage in rats after TSCI.
The studies above have shown promising outcomes of metabolic intervention in rats with TSCI, it remains unclear why bladder function impairment in some patients with spinal cord injury progresses to neurogenic bladder despite the recovery of lower limb function. Normal voiding depends not only on the coordinated opening of the sphincter through the nerve but also on the cooperated contraction of the detrusor [2,3]. Detrusor instability occurs in most patients with neurogenic bladder. Therefore, it is necessary to elucidate the pathological process of spinal cord injury for metabolic interventions. Time-dependent metabolomics studies can uncover the changes in the levels of metabolites in neurogenic bladder after TSCI and shed new light on the pathological mechanisms.
Here, for the first time, we used time-series-based untargeted metabolomics to systematically examine the metabolic status of bladder muscle at different time points following TSCI. A large number of metabolites involved in injury and repair were identified. The results may provide useful evidence for metabolic interventions in the treatment of neurogenic bladder in patients with TSCI.
MATERIALS AND METHODS
Animals
Male Sprague-Dawley rats (180–220 g, aged 8 weeks) were purchased from the Animal Center of Shanxi Medical University, with free access to irradiated chow and water.
Establishment of the Rat Model of TSCI
Rats were randomly divided into 7 groups (6 rats per group), including 1 control group (DDZ0) and 6 TSCI groups (SSZ1, SSZ2, SSZ3, SSZ4, SSZ5, and SSZ6). The rat model of TSCI was established using a weight-drop method [11]. Briefly, rats were anesthetized with pentobarbital (1%, 40 mg/kg, Merck, Sigma-Aldrich; St. Louis, MO, USA) and the lamina was removed at T9–10 using a laminectomy. Then, the spinal cord was confused by an 18-g weight that was dropped from a height of 15 cm. The rats in the sham operation group were treated similarly without the contusion of the weight. Bladder tissues were dissected from the rats at 30 minutes, 6 hours, 12 hours, 24 hours, 5 days, and 2 weeks after injury (corresponding to SSZ1, SSZ2, SSZ3, SSZ4, SSZ5, and SSZ6, respectively) (Fig. 1A). After removing the serous membrane and mucosa at 4°C, due to the varying degrees of congestion in mucosal tissue during the disease process and the ability to secrete hormones, bladder tissues were frozen in liquid nitrogen and stored at -80°C. The time points after SCI were selected based on our preliminary study and relevant literature [12-15]. The acute-phase response likely occurred within 2 days after spinal cord injury, and the chronic and long-term response likely occurred after 2 weeks.

Clusters of the differential metabolites in the detrusor muscle at different time points in fats after traumatic spinal cord injury (TSCI) using untargeted metabolomics. (A) Schematic overview of the experimental design. The rat model of TSCI was established using the weight-drop method. In the control group, rats were anesthetized and only the skin on the back was removed. (B) The tree diagram showing the distribution between the superclass (color) and class (square) of 1,272 metabolic characteristics for statistical analysis. Each number corresponds to a class listed in the source data file. (C) The results of detrusor muscle strips in rats after 2 weeks of injury. (D) Heatmap and cluster tree of differential metabolites at different time points in bladder muscle after TSCI. 6 TSCI groups: SSZ1, SSZ2, SSZ3, SSZ4, SSZ5, and SSZ6; 1 control group: DDZ0. The relative levels of differential metabolites were normalized, converted, and clustered using hierarchical clustering analysis.
Metabolite Extraction From the Bladder Tissue
To extract metabolites, the bladder tissue was homogenized in ice-cold 80% methanol for 5 minutes, followed by centrifugation (15,000 g, 20 minutes) at 4°C. The supernatant was diluted with ultrapure water until the final concentration of methanol was 53%. The diluted supernatant was centrifuged (15,000 g, 20 minutes, 4°C) again, and the resulting supernatant was collected and subjected to metabolomics analysis.
Untargeted Metabolomics
Metabolomic profiling of bladder muscle was performed using liquid chromatography-mass spectrometry (LC-MS) [16]. Quality control samples were prepared by an equal volume mix of experimental samples, which were tested before, during, and after the measurement of the experimental samples to ensure the accuracy and reliability of the data. The LC-MS data were analyzed using Compound Discoverer 3.1 (ThermoFisher Scientific, Thermo Fisher, Waltham, MA USA) to determine the levels of metabolites.
For more accurate identification of metabolites, peak alignments for different samples were performed at a retention time deviation of 0.2 minutes and a mass deviation of 5 ppm. Then, peak extraction and the quantification of peak area were carried out with the following parameters: mass deviation 5 ppm, signal intensity deviation 30%, signal-to-noise ratio 3, and minimum signal intensity of adduct ions. After the integration of target ions, molecular formulas were predicted based on molecular ion peaks and fragment ions, followed by comparison with mzCloud (https://www.mzcloud.org/), mzVault, and MassList databases. Background ions were removed by subtracting the signals of blank samples, and the raw quantitative results were normalized to identify and quantify metabolites.
RESULTS
Differential Metabolites in TSCI Rats Compared to Control Rats
We detected a total of 1,271 metabolites in bladder muscle in rats after TSCI. By using Classyfire, a total of 988 metabolites were annotated and classified into different groups, including organic acids and derivatives, organic oxygen compounds, benzenoids, lipids and lipid-like molecules, nucleosides, nucleotides and analogues, phenylpropanoids and polyketides, organoheterocyclic compounds, organic nitrogen compounds, alkaloids and derivatives, and others (Fig. 1B). The heatmap showed significant changes in the levels of differential metabolites in muscle tissue over time (Fig. 1C). The number of differential metabolites varied at different time points after TSCI, with 161, 352, 436, 521, 199, and 345 differential metabolites detected at 30 minutes, 6 hours, 12 hours, 24 hours, 5 days, and 2 weeks, respectively (P<0.05), by comparing the Kyoto Encyclopedia of Genes and Genomes (KEGG) database, LIPID MAPS database and HMDB database. Rats develop urinary incontinence around 5 days after TSCI. Before and after 5 days, the coincidence of changes in the number of differential metabolites and the occurrence time of urinary incontinence suggests a significant impact of bladder capacity on detrusor function (Fig. 1D).
Dynamics of Muscle Metabolome Associated With the Progression of Neurogenic Bladder After TSCI
After completing the detection of metabolites, in order to reflect the overall metabolic differences between each group of samples and the degree of variation between samples within the group (Fig. 2). Data were subjected to principal component analysis and partial least squares discriminant analysis (PLSDA) using metaX [6], and further validation was performed. In the univariate analysis, the difference in the levels of each metabolite between TSCI rats and control rats was compared using t-test, and the fold change (FC) of metabolite levels in rats with TSCI was calculated compared to the control rats. The criteria for differential metabolite screening were variable importance in projection>1, P-value<0.05 and FC≥2 or FC≤0.5. The results showed that the levels of all metabolic groups changed significantly after 6 hours in TSCI rats compared to the control rats. The 200 interaction test trials showed that the model used here was good and all parameters met the requirement.
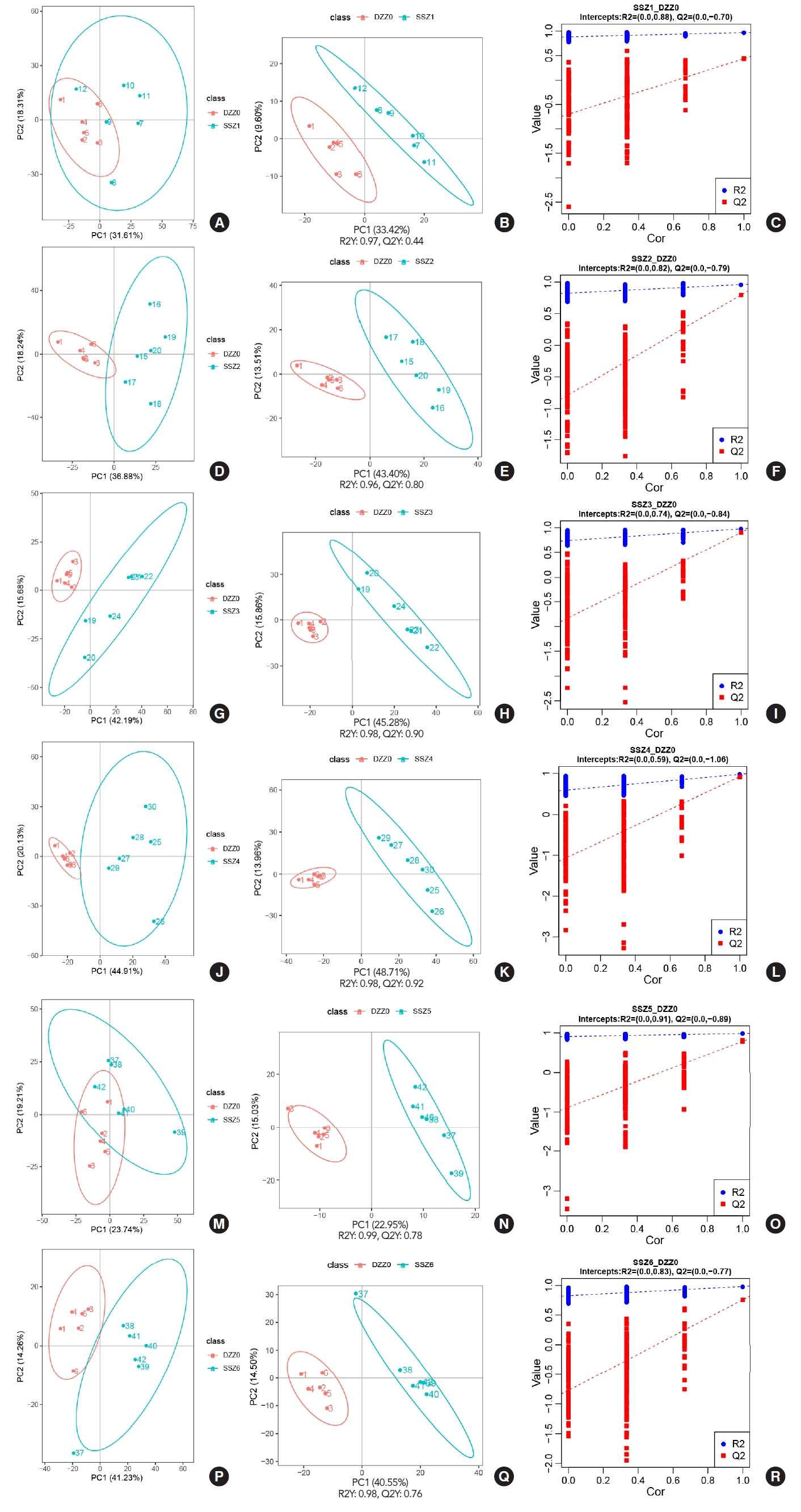
Principal component analysis (PCA), partial least squares discriminant analysis (PLS-DA), and valid test results for the control group and the model group of the detrusor at different time points. (A, D, G, J, M, P) PCA results of 30 min/6 hr/12 hr/24 hr/5 days/2 weeks group. (B, E, H, K, N, Q) PLS-DA results of 30 min/6 hr/12 hr/24 hr/5 days/2 weeks group. (C, F, I, L, O, R) Valid test results of 30 min/6 hr/12 hr/24 hr/5 days/2 weeks group.
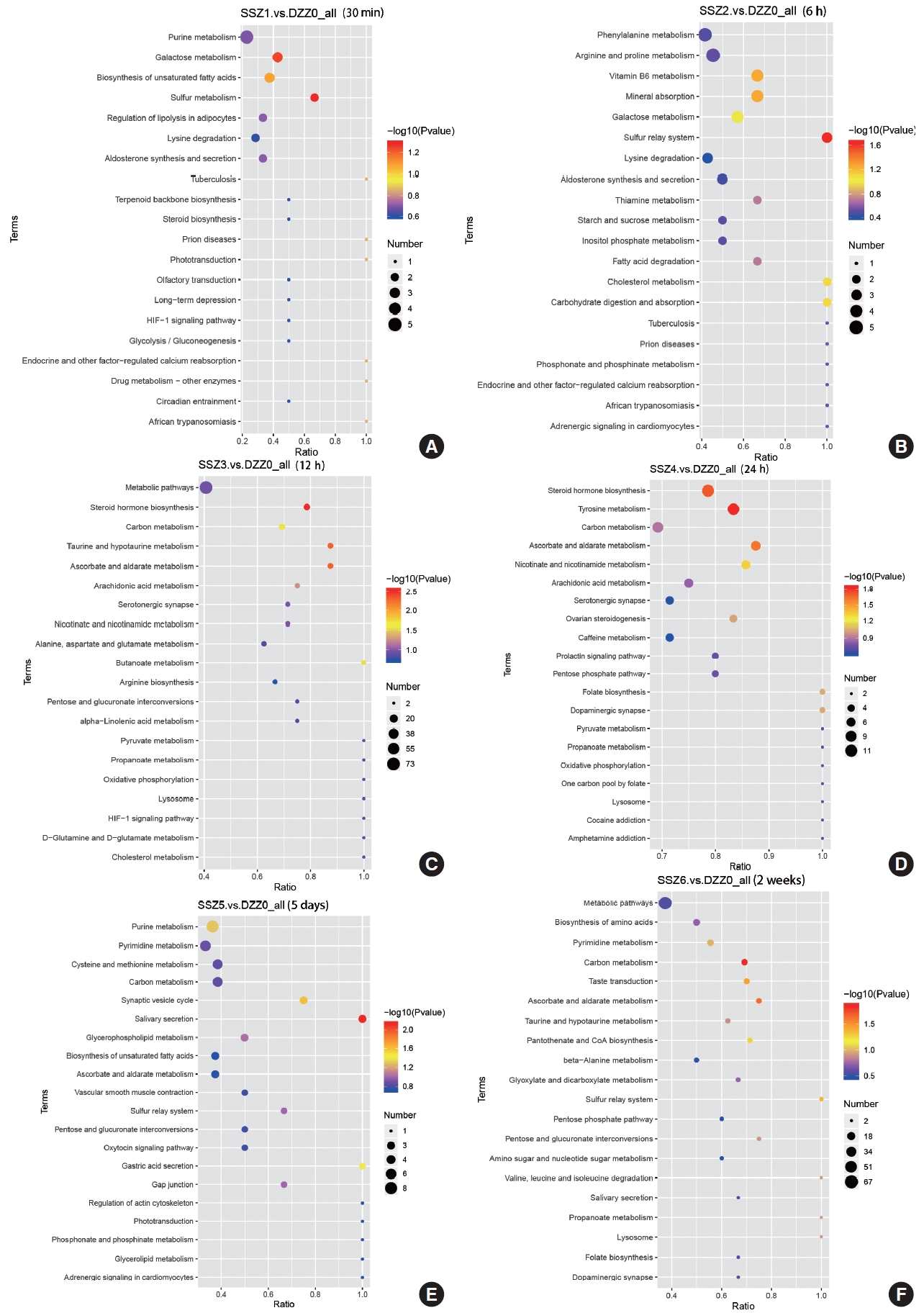
Time-Dependent Pathway Analysis of Bladder Muscle After TSCI
The results of the KEGG metabolic pathway analysis indicate that ascorbic acid and aldehyde metabolic pathways continue to play an important role in the stabilization phase of an injury or disease (Fig. 3). Multivitamins are involved in the injury process. In the acute phase of injury, the synthesis of steroid hormones, is an important stress response in the face of volume expansion stress. Abnormalities in carbon metabolism during the duration of injury may mark a shift from functional to organic injury, and this shift may be accompanied by impaired energy circulation. Differences in the course of injury at 30-minute postinjury, caused by varying degrees of bladder volume expansion, may account for the inability to obtain significant metabolic pathways from KEGG enrichment analysis, suggesting laterally that bladder volume expansion is a major driver of disease progression, at least in the early stages of injury.
Trend Characterization of Differential Metabolites
Based on the muscle contraction characteristics of neurogenic bladder (unstable detrusor contraction) and the results of KEGG pathway analysis, we focused on the metabolic characteristics of the following 4 aspects: (1) changes in energy supply patterns during disease occurrence; (2) secretion of steroid hormones during bladder expansion; (3) changes in the content of muscle neurotransmitters during the occurrence of diseases; (4) antioxidant stress and metabolic protection during the disease process.
There are 3 energy supply pathways for muscles: aerobic metabolism, anaerobic metabolism, and phosphocreatine pathway. After literature review and comparison, we have finally determined α-Ketoglutaric acid, fructose 1,6-phosphate, and creatine phosphate are markers of 3 energy metabolism modes. They are either substrates of rate limiting enzymes in the process of energy metabolism, or direct energy sources of adenosine 5’-triphosphate production (phosphocreatine pathway), which have strong representativeness, stability, and relatively high content and are easy to detect in metabolic testing. During the comparison process, we found an interesting phenomenon that there is a significant difference in the relative content of phosphocreatine within the same group. There was no significant difference in the relative content of ketoglutaric acid and fructose 6-phosphate within the group. After excluding modeling reliability factors, we believe that its instability may be related to the state of the detrusor muscle (detrusor hyperreflexia and detrusor reflex delay), and may serve as a biomarker for detrusor function to differentiate bladder function in patients with neurogenic bladder.
We found that steroid hormones maintain high levels of secretion within 12–24 hours after injury. The enriched metabolites include both glucocorticoids represented by corticosterone and a series of sex hormones represented by estrogen, estradiol, and 17a hydroxyprogesterone. Their changes over time during the process of disease occurrence are shown in Fig. 4A, and their secretion characteristics are as follows: (1) during the acute injury period, secretion intensifies with time progression (increased bladder volume dilation pressure); (2) in the early stage of urinary incontinence, with the occurrence of filling urinary incontinence, the bladder capacity pressure decreases and secretion decreases instantly; (3) As the disease enters the chronic stage, the amount of residual urine increases (despite urinary incontinence), and its content increases; Here, we found that the release of steroid hormones is significantly controlled by bladder capacity.
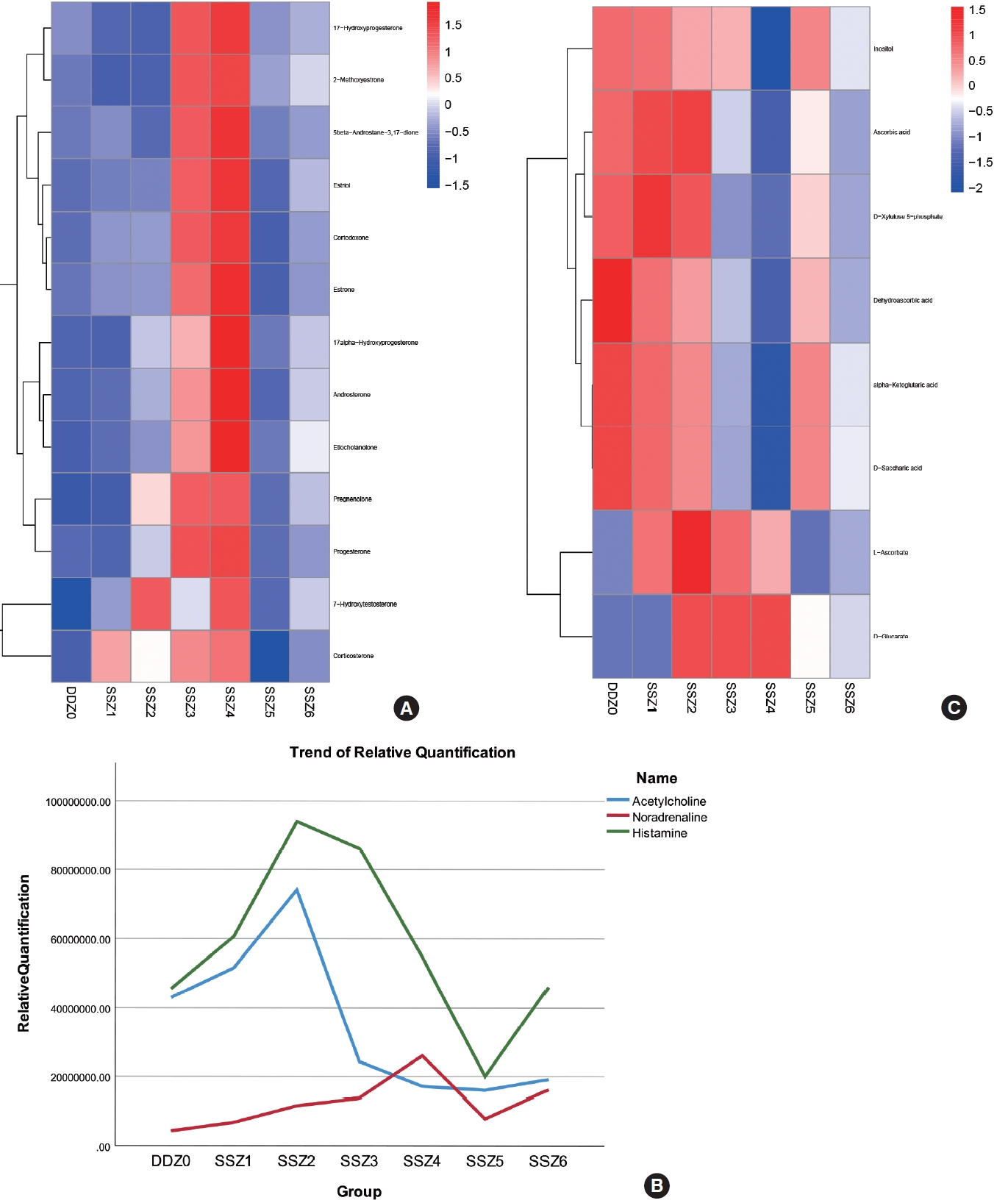
Trend of major differential metabolites over time. (A) Heat map of Steroid hormone biosynthesis. (B) Trend chart of the relative content of acetylcholine, histamine and noradrenaline. (C) Heat map of Ascorbate and aldarate metabolism.
Changes in the content of muscle neurotransmitters during the process of disease occurrence: After TSCI, neurogenic bladder patients experienced denervation of the bladder, so we focused on the relative content of neurotransmitters in muscle tissue over time. The neurotransmitters we have enriched include 2 types: monoamine neurotransmitters such as noradrenaline, and amino acid neurotransmitters such as histamine and acetylcholine. Their time variation curves are shown in Fig. 4B. We can intuitively observe that the trends of acetylcholine and histamine are similar, while the relative content of noradrenaline increases with the increase of bladder volume dilation pressure before urinary incontinence. However, due to the destruction of the central nervous system, the relative content of acetylcholine continues to decrease after reaching its peak. Even after urinary incontinence, the upward trend is very slight in the face of volume expansion caused by residual urine. The relative content of norepinephrine is almost unaffected. This may have caused detrusor sphincter dyssynergia.
In the KEGG enrichment analysis results, the metabolic pathways of ascorbic acid and aldehydes have attracted our attention. This pathway is significantly enriched at almost every time period of injury (P<0.05), and the content of differential metabolites enriched by this metabolic pathway in the group samples and the temporal trend of different differential metabolites maintain good consistency. This suggests that the biological functions involved in the compounds in this pathway should be relatively single and stable during the disease process, as shown in the Fig. 4C, May be fully involved in the occurrence of neurogenic bladder after spinal cord injury. Based on our understanding of the biological function of ascorbic acid and the mechanism of disease occurrence, we believe that ascorbic acid may mainly participate in antioxidant stress damage here.
DISCUSSION
In this study, we identified a larger number of important metabolites in the bladder detrusor during the acute/subacute and chronic phases of neurogenic bladder in rats after TSCI using untargeted metabolomics. We also found time-dependent changes in major differential metabolite pathways.
Firstly, there may be a time window for metabolic intervention. In the early stage of spinal shock, especially within 6 hours, the number of differential metabolites gradually increases in rats. For example, L-ascorbic acid reaches its peak at 12 hours after injury. Although it is still higher than normal at 24 hours after injury, it shows a decrease compared to 12 hours after injury. This reflects the rapid depletion of reserve materials in the face of capacity expansion pressure. At around 5 days, spinal shock ended and the levels of differential metabolites decreased rapidly, indicating that the decreased bladder volume due to urinary incontinence attenuated metabolic damage. At 2 weeks, however, the injury worsened again, as evidenced by clustered heatmap and the number of differential metabolites (Fig. 1D), indicating the recurrence of the above pathological process and some differential metabolic pathways. This result suggested the necessity of long-term metabolic intervention.
Secondly, there are significant differences in the relative content of phosphocreatine among different samples within the same group. Early research on neurogenic bladder in diabetes found that the unstable contraction of neurogenic bladder detrusor might be related to the energy metabolism mode [17]. The results of this experiment indicate that this relationship may be related to the phosphocreatine metabolism pathway in muscles, while the aerobic and anaerobic respiration of detrusor muscle cells are not related to this change. Unfortunately, in order to ensure the stability of sampling after modeling, we did not retain muscle tissue for muscle electrophysiological testing during the same period.
Besides, we found that the number of steroid hormones increased consistently in 12–24 hours after injury, including 17-hydroxyprogesterone (17-OHP), progesterone, androgen, estrogen, corticosterone, hydrocortisone, etc. 17-OHP is an intermediate steroid in the adrenal biosynthetic pathway that converts cholesterol to cortisol by binding to the glucocorticoid receptor [18]. Though steroid hormones (such as 17-α-hydroxyprogesterone and progesterone) are commonly found in females during pregnancy, they are also secreted in males under normal conditions. They can relax smooth muscle, modulate the inflammatory response to smooth muscle injury, and exert an antiendotoxin effect in local tissues [19,20]. Here, the secretion of a large amount of steroid hormones may be caused by the response of the detrusor muscle to the enlargement of the bladder during the acute phase of spinal shock. In addition, the increased progesterone level may also have a protective effect by inhibiting endoplasmic reticulum stress-induced apoptosis [21]. Since rats already had urinary incontinence during spinal shock, it can be assumed that the elevation of progesterone was likewise due to the enlarged bladder. High levels of progesterone can suppress sexual desire [22], which explains why some patients with urinary retention from neurogenic bladder are often accompanied by sexual dysfunction. Nevertheless, we believed that sexual function is also controlled by the spinal center. The changes in steroid hormones explained the enlargement of bladder capacity following injury and the release of steroids from the local bladder muscle. To maintain bladder compliance for an increased residual urine volume, the steroids could inhibit muscle contraction and protect the injured muscle cells from excessive apoptosis and proliferation of fibrous connective tissue. However, the results also indicated that this compensatory mechanism was not sufficient to compensate for the consequences of the injury.
In addition, its involvement in various physiological processes, ascorbic acid (also known as vitamin C) plays an important role in healing muscle injuries and provides a favorable microenvironment for the recovery of peripheral nerves [23,24]. Muscle rupture and scar-forming fibroblasts are found in the bladder after spinal cord injury [25]. In this study, we found significant alterations in taurine metabolism during the acute phase of neurogenic bladder. It is reported that the combination of taurine and ascorbic acid decreases the mRNA expression of inflammatory factors (such as interleukin-6) and reduces lipid peroxidation [26]. In addition, detrusor instability is often accompanied by ischemia and reperfusion, and oxidative stress induced by chronic bladder ischemia can promote the urination/micturition reflex [27]. As an important antioxidant, ascorbic acid (vitamin C) could be actively involved in the progression of neurogenic bladder, which provided a new direction for metabolic interventions.
In conclusion, LC-MS-based metabolomics revealed time-dependent changes in the metabolome of bladder muscle. Based on PLS-DA analysis and enrichment analysis of the KEGG database, important pathways associated with the development of neurogenic bladder after TSCI were identified, including sulfur metabolism, steroid hormone biosynthesis, ascorbate and aldehyde metabolism, taurine and hypotaurine metabolism, butyrate metabolism, tyrosine metabolism, ascorbate and aldehyde metabolism, nicotinic acid and nicotinamide metabolism, carbon metabolism, and synaptic vesicle cycle. Of these, the ascorbic acid metabolism played a prominent role. Further studies using more specific approaches (such as targeted metabolomics) are needed to elucidate the exact metabolic pathways of key differential metabolites.
Notes
Grant/Fund Support
This work was supported by the Science and Technology Department of Shanxi Province No. YDZJSX2022A065.
Research Ethics
This study was approved by the Animal Experimentation Ethics Committee of Shanxi Medical University and was in accordance with the National Academy of Sciences Guide for the Care and Use of Laboratory Animals (IRB: DWYJ-2023-101).
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION STATEMENT
· Conceptualization: WBS
· Data curation: YHL, QG, YKW
· Formal analysis: YHL
· Funding acquisition: WBS
· Methodology: YHL, WBS
· Project administration: YHL, WBS
· Visualization: YHL
· Writing - original draft: YHL
· Writing - review & editing: WBS
Acknowledgements
LC-MS/MS testing was conducted by Novogene Co., Ltd. (Beijing, China).