Competitive Inoculation as an Effective Prophylaxis Against Recurrent Urinary Tract Infections: A Systematic Review
Article information
Abstract
Urinary tract infection (UTI) is a common condition defined as the presence of bacteria within the urine above a certain threshold (usually >100,000 m/L). The lifetime risk in women is estimated to be 50%, of whom 25% will develop recurrence within 6 months. Unfortunately, the use of antibiotics to treat and manage recurrent UTI (rUTI) is a growing problem, due to the burden of growing antibiotic resistance on public health. As such, new approaches to manage rUTI are being investigated and developed. Competitive inoculation via instillation of Escherichia coli 83972 or HU2117 in the bladder is a new prophylactic non-antimicrobial therapy for rUTIs. It utilizes the principle of the protective nature of asymptomatic bacteriuria to prevent recurrence of symptomatic UTIs. However, the effectiveness and safety of this technique remains unclear. This systematic review examined the current outcomes data on competitive inoculation as an effective and safe treatment for rUTI prophylaxis. Based on a limited number of studies, current evidence suggests that competitive inoculation is an effective and safe prophylactic measure against UTIs in a select group of patients with incomplete bladder emptying. However, administration of the technology is both resource and time intensive, and there is strong data demonstrating low successful colonisation rates. Competitive inoculation is an alternative to antibiotics only to rUTI patients with incomplete bladder emptying. There is no evidence to suggest that the technology would be suitable for other subsets of rUTI patients. Further randomized controlled trials should be conducted to improve the evidence base before drawing conclusions for clinical practice, and ideas to improve colonisation rates and simplify the administration process should be explored.
INTRODUCTION
Urinary tract infection (UTI) is a common condition defined as a symptomatic infection that involves any part of the urinary tract, such as the bladder and urethra [1]. The presence of bacteria within the urine is usually required to surpass a certain threshold (usually >100,000 colony forming unit [CFU]/mL) to meet microbiological criteria. Escherichia coli is the leading infectious agent, accounting for 80% of all community-acquired infections [2]. There is a propensity for the disease to recur, especially in women. A study conducted in young women with risk factors for UTI noted that 27% developed one episode of microbiologically confirmed recurrence within 6 months [3]. Ikäheimo et al. [4] observed that 53% of women above the age of 55 had experienced recurrent UTI (rUTI) at some point in their lives. Considering 150–300 million people are affected by UTIs worldwide each year, the burden of rUTI on public health is vast [5].
Standard prophylaxis of rUTI, defined by European Association of Urology guidelines as ≥2 episodes in the last 6 months or ≥3 episodes in the last 12 months, involves regular low dose antibiotics [6]. There is considerable concern this contributes to the rise of antibiotic resistance, a mounting global crisis. It is predicted that 2.4 million people in the western world will die from multidrug-resistant microorganisms in the next 30 years [7]. In recognition of the dangers antibiotic resistance poses to society, multiple nonantibiotic methods have been developed to combat rUTIs.
One unique approach is using the protective effects of asymptomatic bacteriuria (ABU), and henceforth the urinary microbiome, in combatting rUTIs [8]. ABU is defined by the asymptomatic carriage of >105 CFU/mL of urine in 2 consecutive urine cultures [9]. In clinical practice, there is debate about whether ABU should be treated with antibiotics. Current guidelines by the Infectious Diseases Society of America do not recommend treating ABU, aside from in pregnant women. This is because there is little evidence to suggest antibiotics lower the risk of symptomatic infection [10-12]. In fact, multiple studies have demonstrated ABU can be a preventative measure against symptomatic UTIs.
Two strains of E. coli that cause ABU have been explored in clinical trials. The first strain, E. coli 83972, was isolated from a Swedish child with ABU who had carried it for more than 3 years [13]. The second strain, E. coli HU2117, is a derivative of E. coli 83972 [14]. Both are capable of long-term bladder colonisation and are well-adapted to the urinary tract. Clinicians instil these strains with saline solution into the bladder as prophylaxis. This approach is known as competitive inoculation, or bacterial interference, as the subvariants aim to outcompete virulent E. coli for nutrients and ultimately survival.
Generally, the urinary microbiome prevents the establishment of uropathogenic strains in the bladder via competition for nutrients, host-cell binding molecules and inhibition of virulence factors [15]. Interestingly, E. coli 83972 and HU2117 outcompete uropathogens due to their immense growth rate in urine as opposed to other mechanisms [13]. Competitive inoculation is an attractive technology because it has several advantages over traditional antibiotic treatment. First, it does not create selection pressure for resistance as uropathogens are not killed as the primary mechanism. Second, the strains used can be engineered to improve capability for different scenarios.
There is a growing body of literature that suggests that competitive inoculation is an effective form prophylaxis against rUTIs. This review will examine the current outcome data on competitive inoculation as a treatment for rUTI. It will also aim to establish whether the technology poses any health risks and what steps can be taken for future research.
METHODOLOGY
The search strategy was formed to identify studies investigating the efficacy of competitive inoculation as prophylaxis for rUTIs. The following search terms were inputted into MEDLINE (1966– 2022) and Embase+Embase Classic (1947–2022): (E. coli 83972 OR Escherichia coli 83972 OR 83972 OR Asymptomatic Bacteriuria OR Bacterial Interference OR Competitive Inoculation OR antibiosis) AND (RUTI OR rUTI OR Recurrent Urinary Tract Infection or Chronic UTI or cUTI or Chronic Urinary Tract Infection). Additional eligible studies were identified from the references of included papers. Moreover, EU Clinical Trials Register and ClinicalTrials.gov were searched for any ongoing trials. The search was conducted and concluded on 10/02/22.
The inclusion criteria were: studies must be randomized controlled trials (RCTs) or nonrandomized studies of intervention (NRSI); the intervention must be bladder instillation of bacterial variants compared to placebo; the patient population must have rUTI. On the other hand, studies were excluded if they were conducted on animals or were not the study types mentioned previously.
The primary outcome measure was the number of symptomatic or microbiologically proven UTI within the author defined timeframe. Secondary outcomes were the frequency and type of adverse events recorded during treatment or follow-up, % of successful colonisations and mean duration of successful colonisations.
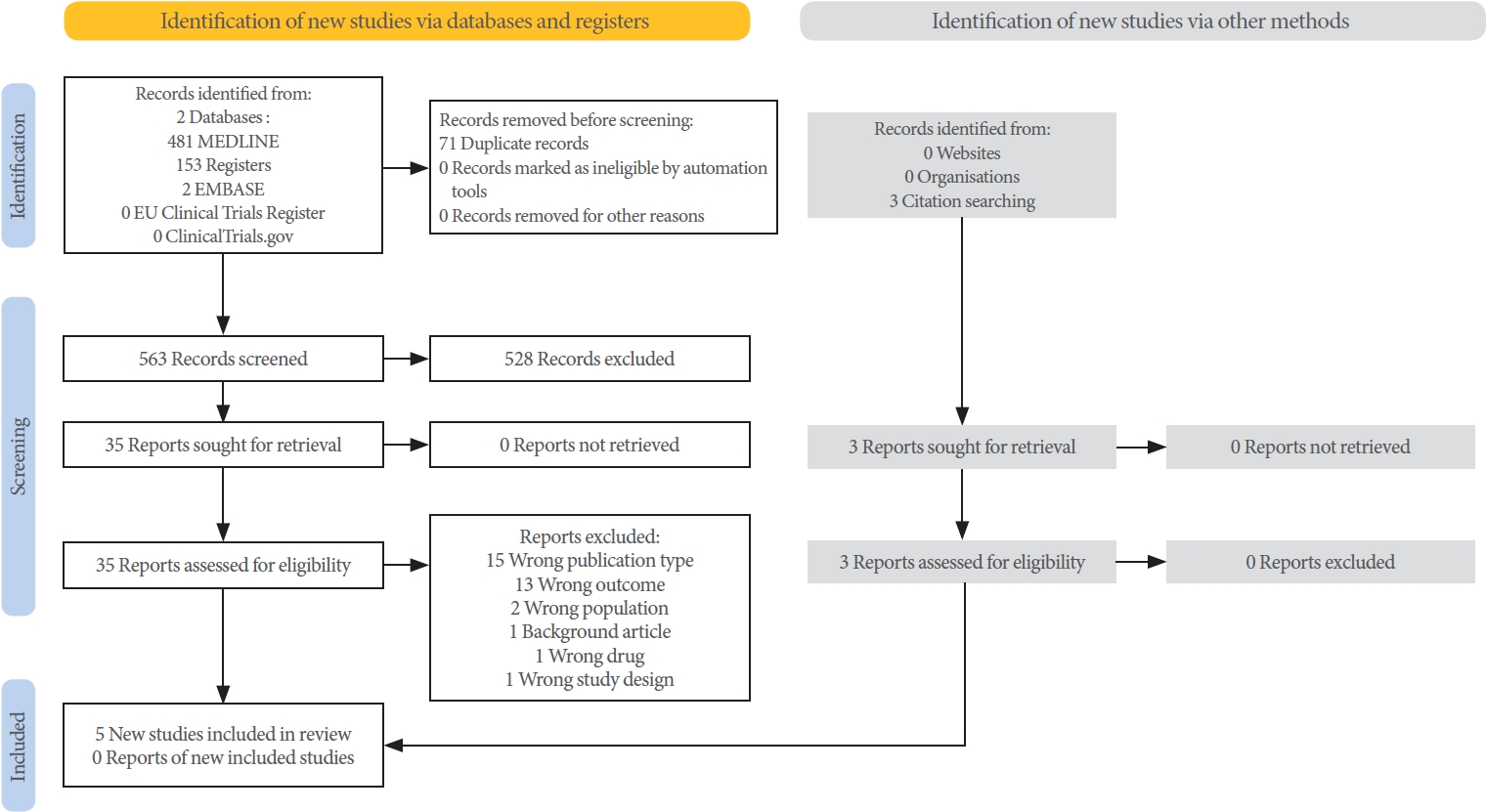
Two reviewers (QM and JG) screened and selected studies. Duplicates were removed using EndNote 20 (EndNote 20.3) software [16], and the remaining studies were uploaded to Rayyan [17]. The titles and abstracts were screened independently, and full-text analysis was conducted on records that passed the criteria, see PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) 2020 flow chart (Fig. 1). Any dis agreements were resolved by discussion.
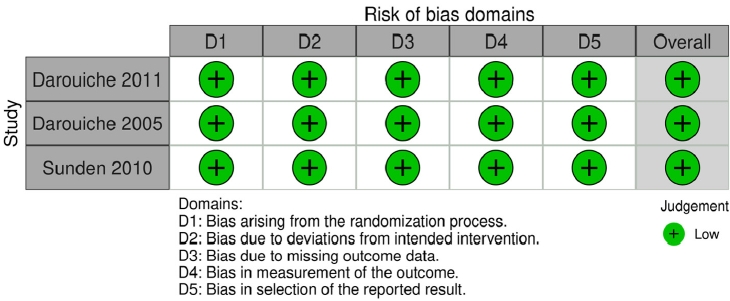
Quality assessments were conducted independently by QM and JG using Cochrane risk of bias tools. RCTs and NRSIs were evaluated using the RoB 2 and ROBINS-I tools respectively. Discrepancies were settled by discussion.
RESULTS
1. Study Characteristics
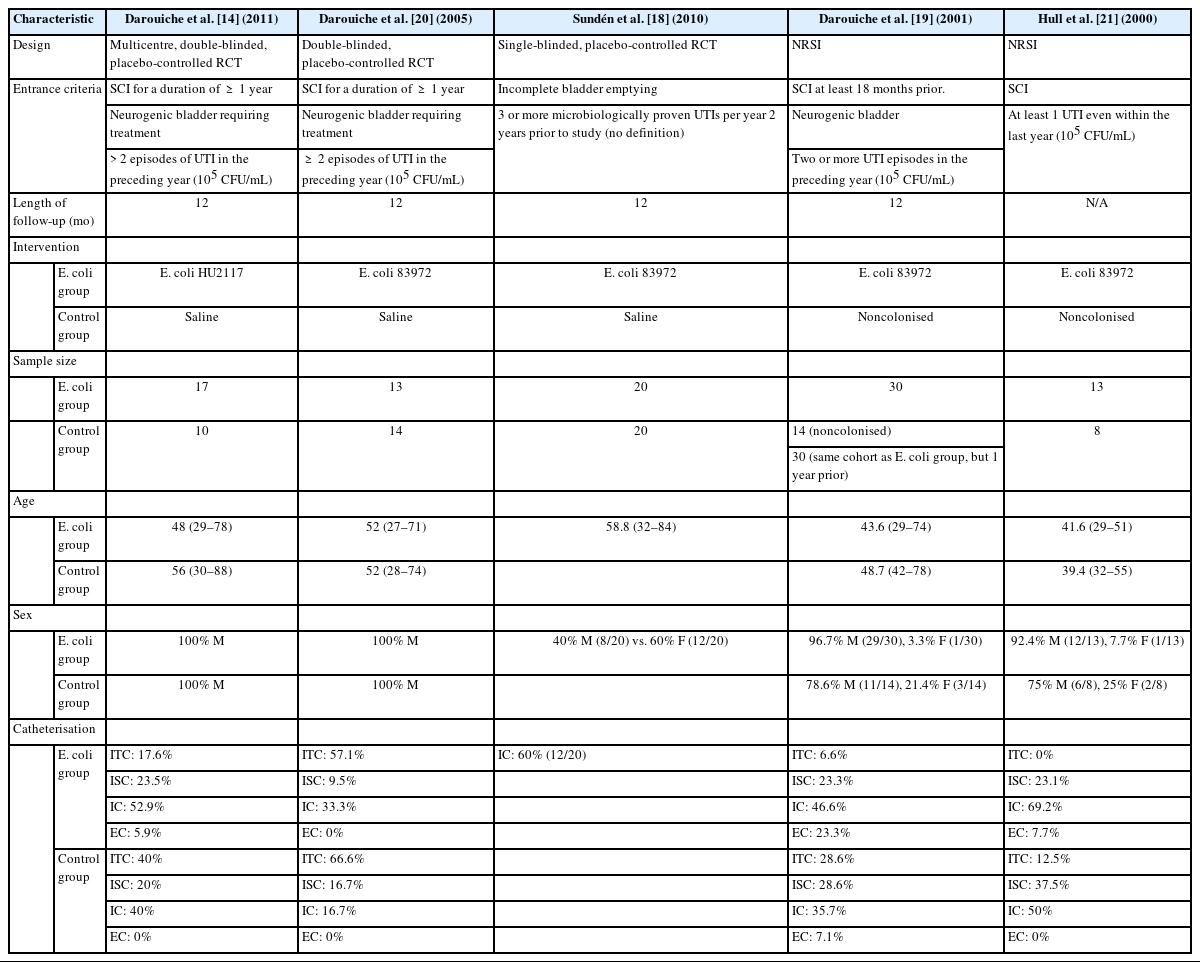
Five studies met the inclusion and exclusion criteria (Fig 1). Three studies were RCTs, and 2 studies were NRSIs. Fig. 2 (RCTs) and Fig. 3 (NRSIs) present an overview of the risk of bias analysis for the 5 studies. As shown, all but one study was of good quality: Hull 2000 was adjudged to have a moderate risk of bias. The authors did not resolve confounding concerns, and there may have been some selection bias. Sundén et al. [18] featured a crossover study design. The risk of bias arising from period and carryover effects was assessed to be low.
Darouiche et al. [14,19,20] and Hull et al. [21] explicitly recruited patients with spinal cord injury (SCI) and neurogenic bladder (4 of 5 studies). Additionally, key criteria regarding patient selection across all 5 studies were that patients had to be ≥18 yearsold and have incomplete bladder emptying. The definitions of rUTI differed, ranging from 1 episode of UTI in the past 12 months to 3 or more episodes of UTI in the past 12 months. The microbiological threshold for UTI was 105 CFU/mL in 4 of 5 studies. Sundén et al. [18] did not confirm their microbiological threshold for UTIs. E. coli 83972 was the chosen intervention in 4 of 5 studies, while E. coli HU2117 was used by Darouiche et al. [14]. Saline was chosen as a placebo in 3 of 5 studies, whilst no intervention was chosen as the control in 2 of 5 studies (noncolonised patients). All selected studies had relatively small sample sizes. The largest study was conducted by Darouiche et al. [19], whose team recruited 44 patients (Table 1). Across all studies, mean age ranged between 40–60 years.
Incomplete bladder emptying in included patients was managed via a variety of methods: intermittent catheterisation (IC), indwelling transurethral catheter (ITC), indwelling suprapubic catheter (ISC) and external convene catheters (EC). Generally, EC was the least popular method, used in only 3 of 5 studies (not utilized in Darouiche et al. [20] or Sundén et al. [18]) and proportionally always below 25%. On the other hand, IC was the most popular method. It was used in all 5 studies, and the median proportion across all eligible studies (excluding Sundén et al. [18]) was 43.3%, as opposed to ITC and USC which were seen in 4 of 5 studies and had medians of 23.1% and 23.2% respectively (Table 1).
The only exception is the study of Sundén et al. [18], where 40% of the participants used no adjuncts to aid bladder emptying. A high proportion of male participants were recruited in all studies except [18]. Darouiche et al. [14] and Darouiche et al. [20] recruited exclusively male participants while Darouiche et al. [19] and Hull et al. [21] included over 75% men. Only Sundén et al. [18] had a more balanced cohort (male to female ratio was 40:60).
2. Efficacy of Competitive Inoculation
The definition of symptomatic UTI also differed across the 5 studies. Darouiche et al. [14,19,20] all utilized the same criteria: Significant bacteriuria (105 CFU/mL), pyuria (10 leukocytes/high-power field), and fever plus ≥1 of the following signs and symptoms for which no other aetiology could be identified: suprapubic or flank discomfort, bladder spasm, increased spasticity, and worsening dysreflexia. Hull et al. [21] followed a similar set of rules. Sundén et al. [18] defined symptomatic UTIs as when patients reported symptoms and antibiotics were prescribed for treatment.
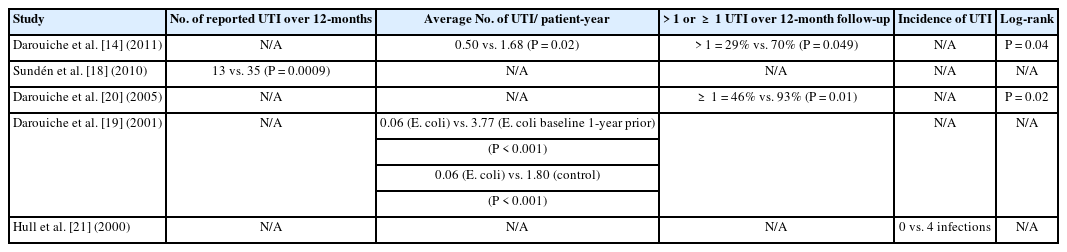
There was marked heterogeneity in primary outcome measures reported by the studies (Table 2). Four studies [14,18-20] reported statistically significant different reductions (P<0.01) in number of UTI during a 12-month follow-up period. Of note Hull 2000 did not conduct statistical analysis, but the results concur with the other studies; no patients in the intervention group developed UTI, as opposed to 4 incidences in the control group. Interestingly, Sundén et al. [18] also reported a reduced median time to infection in favour of the intervention group (11.3 months vs. 5.7 months, P=0.0129).
Complications during instillation or follow-up due to the 2 strains of E. coli were rare. Only Darouiche et al. [20] reported an adverse event, where one patient in the placebo group developed autonomic dysreflexia immediately after inoculation but resolved after 30 minutes. This is most likely associated with the method of delivery as opposed to the E. coli itself. There was no evidence of septicaemia or other adverse events related to competitive inoculation.
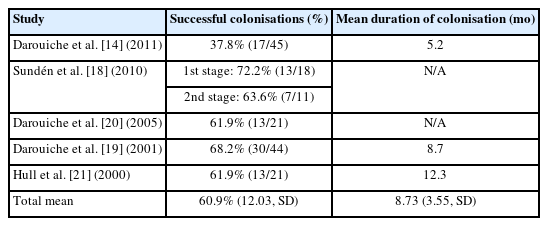
Rates of successful colonisations of the bladder were variable (Table 3). The instillation process was similar across the 5 studies. The solution is delivered through a catheter, which is then clamped for a variable amount of time (1–3 hours) to allow for retention. The procedure is repeated over 3 days should it be required, with the number of repeat instillations per day varying between studies. Sundén et al. [18] only conducted one inoculation daily, whilst Darouiche et al. [14,19,20] inoculated twice daily and Hull et al. [21] thrice daily. A successful colonisation was classified as detectable growth (>103 CFU/mL) of E. coli for one month or more. Overall, the mean of successful colonisations across all studies is 60.9% (±12.03, standard deviation [SD]). The mean duration of colonisation is short. On average, the mean across the eligible studies was 8.73 months (±3.55, SD).
Meta-analysis of the intervention effect was not possible as the primary outcome measures could not be standardised. For this reason, the quality of evidence was assessed in accordance with the approach Murad et al. [22] on performing GRADE analysis in narratively synthesised data. The overall judgment was the GRADE (certainty) of evidence for reduction in symptomatic UTI episodes was moderate (Table 4).
DISCUSSION
Results from this systematic review evaluating current evidence for the use of competitive inoculation using either E. coli 83972 or HU2117 demonstrates that it may be a safe and effective method for reducing rUTI in patients with incomplete bladder emptying. More specifically, competitive inoculation reduces the number of symptomatic UTI episodes in patients within the short term. Only one adverse event was reported in total between the 5 studies. There is also some limited evidence to suggest that the intervention delays UTI recurrence, which is unsurprising.
Cai et al. [8] demonstrated in an RCT that ABU in young women may have protective effects in preventing symptomatic recurrence. This conclusion was echoed by subsequent studies evaluating long-term outcomes of ABU in schoolgirls [23,24]. This observation can be linked to the human microbiome. The microbiome is the sum of all genomic information of resident microbiota in the human body. Recent research has indicated that the microbiome plays an important role in preserving host physiology and health. Disruption to the commensal flora can lead to inflammatory bowel disease, cancer and other disease. Over 100 species exist within the urinary tract, helping to maintain homeostasis and prevent pathology such as UTI [25]. The genus Lactobacillus is frequently found in the urinary tract and vagina [26,27], and some data suggest antimicrobial effects through the secretion of lactic acid [28,29]. However, 2 systematic reviews conducted in 2015 and 2021 noted probiotics provided no significant benefit in reducing UTI recurrence compared to placebo. It was concluded that the trials were or poor quality and there was insufficient evidence to support probiotics [30,31]. In another example of competitive inhibition, Ohlemacher et al. [32] discovered that enterobacteria secrete escherichelin that inhibits and prevents opportunistic symptomatic infections from Pseudomonas aeroguinosa. The infecting strains found in ABU may have an intrinsic ability to outcompete or inhibit more virulent variants.
E. coli 83972 and HU2117 were selected as the competitive agents as they are less-virulent strains of uropathogenic E. coli (UPEC). 83972 (HU2117 is a derivative) has been proven to grow in human urine and outcompete UPEC in-vivo [33]. The ability of UPEC to precipitate symptomatic UTIs lies with their production of adhesins which facilitate the attachment of bacteria to host cells. Type 1 fimbriae, PapG adhesins and P fimbriae which help in adhesion to the bladder mucosa are the main culprits in promoting virulence in UPE Only one adverse event was reported in total between the 5 studies. E. coli 83972 has a similar genome to UPEC albeit with mutated genes regulating bacterial adherence [34]. Type 1 and P fimbriae are nonfunctional [33], which explains the lack of host response and possibly the inability to establish itself in a patient with no voiding issues. It is hypothesised that the 2 subvariants outcompete UPEC because of its spectacular ability to replicate in urine. The expression of genes that are involved in iron processing (e.g., sidABCD) and sugar/carbohydrate metabolism (e.g., ara-ABD) are increased, which allows the organisms to thrive in the iron-low sugar-rich environment of urine [13]. It has been proposed that the strain has undergone reductive evolution to resemble a commensal flora [35].
Successful bladder colonisation relies on incomplete bladder emptying [36]. This link informed the patient selection for the current trials. An important observation to emerge from this systematic is the high failure rate of colonisation, even in this patient cohort with incomplete bladder emptying. In the trial of Darouiche et al. [19], only 68% of patients (30 of 44) were successfully colonised by 83972 for one month or longer. Similarly, only 62% (13 of 21) of patients were successfully colonised by 83972 according to the same criterion. This has important implications for the subset of patients who can receive this treatment. Moreover, the mean duration of successful colonisation across the studies was only 8.73 months. When compounded by the high failure rate, this calls into question whether this treatment is appropriate for clinical practice. Patients will require regular reinoculation to maintain colonisation.
Evidence from this review must be considered regarding the limitations in the current literature. Three studies were randomized controlled trials that were at least single-blinded (two were double-blinded) and placebo-controlled. The quality assessment did not uncover any potential risk of bias. However, remaining 2 studies were NRSIs with potential bias detected in one trial [21]. The trials were also limited by small patient numbers and short to medium-term follow-up only. Heterogeneity in study methods and outcomes prevented pooled analysis further limiting the extrapolation of current results to larger patient cohorts.
Feasibility and implementation are a principal factor for any new treatment or technology. Competitive inoculation is a difficult procedure that not only has a high failure rate but also requires multiple treatments over several days. The protocol for inoculation in all 5 studies consisted of bladder instillation 2 to 3 times per day for 3 consecutive days. The burden on patients of such a regime will be considerable and requires high compliance. The costs for such intensive treatment must also be considered. An interesting idea by Trautner et al. [37] considered using catheters coated with E. coli 83972 or HU2117 to treat catheter-associated UTIs. This could be a path forward because it eliminates the need for repeated clinic visits for inoculation and improves patient compliance but requires further study. Further research looking into genetic modifications to improve the ability for adherence in these strains should be conducted. This would rectify the issue of colonisation failure and allow for the technology’s introduction to patients with unencumbered bladder emptying.
The current consensus regarding ABU is it is safe and beneficial for the patient. However, there are concerns that chronic renal infections may present similarly, which can cause permanent scarring [38]. As competitive inoculation is by and large the artificial installation of ABU, distinguishing ABU from other forms of UTI is important to prevent complications should the technology become mainstream. One method that could work is by measuring the host immune response. The literature regarding the immune response to the many different forms of UTI are extensive [39-41]. Conversely, there is little research done on ABU. A study by Grönberg-Hernández et al. [42] characterised the host immune response generated for E. coli 83972 compared to placebo amongst patients. They showed that cytokines seen in innate immunity (IL-8, RANTES) were raised, whilst adaptive immunity associated cytokines were low or absent (IL-2, IL-3). Most importantly, IL-6 which is greatly increased in symptomatic UTI [43] remained low. The findings suggest quantitative measurement of the immune response can delineate ABU from other forms of UTI when subjective symptoms are not reliable, particularly in patients with incomplete bladder emptying as is this case for competitive inoculation.
Current evidence demonstrates that competitive inoculation can be a safe and effective in patients with incomplete bladder emptying, particularly following SCI, in the short term. However, the treatment is limited by considerable burden on healthcare resources of administration and low colonisation rates. Further research into improving colonisation and simplifying administration is required before drawing conclusions for clinical practice. The role of competitive inoculation in patients with normal bladder emptying is unnecessary.
Notes
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION STATEMENT
· Conceptualization: QM, NR
· Data curation: QM, JG, NR
· Formal analysis: QM, JG
· Methodology: QM, JG, NR
· Project administration: QM
· Visualization: QM
· Writing - original draft: QM, JG
· Writing - review & editing: QM, JG, KA, SK, PD, SM, NR
Acknowledgements
Firstly, I would like to thank my co-reviewer Julian Greig, who assisted me every step of the way from screening studies to conducting risk of bias. I wouldn’t have been able to do it without him. I would also like to extend my gratitude to Mr. Sachine Malde, Professor Shamim Khan, Professor Kamran Ahmed and Professor Prokar Dasgupta for their support. Lastly, I am also deeply indebted to Mr. Nicholas Raison, who took it upon himself to supervise the project and never failed to provide meticulous advice despite the many iterations this article has gone through.