|
 |


- Search
| Int Neurourol J > Volume 27(1); 2023 > Article |
|
ABSTRACT
Purpose
In this paper, we propose an optimal ureter stone detection model utilizing multiple artificial intelligence technologies. Specifically, the proposed model of urinary tract stone detection merges an artificial intelligence model and an image processing model, resulting in a multimethod approach.
Methods
We propose an optimal urinary tract stone detection algorithm based on artificial intelligence technology. This method was intended to increase the accuracy of urinary tract stone detection by combining deep learning technology (Fast R-CNN) and image processing technology (Watershed).
Results
As a result of deriving the confusion matrix, the sensitivity and specificity of urinary tract stone detection were calculated to be 0.90 and 0.91, and the accuracy for their position was 0.84. This value was higher than 0.8, which is the standard for accuracy. This finding confirmed that accurate guidance to the stones area was possible when the developed platform was used to support actual surgery.
Conclusions
The performance evaluation of the method proposed herein indicated that it can effectively play an auxiliary role in diagnostic decision-making with a clinically acceptable range of safety. In particular, in the case of ambush stones or urinary stones accompanying ureter polyps, the value that could be obtained through combination therapy based on diagnostic assistance could be evaluated.
Urolithiasis [1–7] is a disease in which substances in urine precipitate, harden like stones in the kidneys, and move along the urinary tract. The diagnosis of urolithiasis is confirmed based on the patient’s clinical symptoms and findings from a physical examination, urine examination, and radiography. A simple urinary tract scan can confirm the presence of stones, but simple radiography cannot diagnose invisible stones. In addition, ureter stones may be difficult to distinguish because they are covered by pelvic bones, feces, or other organs. However, ureter stones can be confirmed by urinary tract imaging or computed tomography. In many cases, it may be difficult to confirm the presence of stones, which is an important part of the clinical diagnosis. Therefore, this paper attempted to develop an artificial intelligence (AI) model that can help detect urinary tract stones and evaluated its accuracy.
AI has begun to be used in various medical fields. The urological system is no exception, and AI research is actively underway throughout the entire cycle of the analysis, diagnosis, treatment, and management of urological diseases. The application of AI technology has mainly focused on the tasks of diagnosis and analysis, and methods have been proposed to assist clinicians in the diagnosis. A recent analysis compared various AI models that assist in the diagnosis of urinary tract stones [8]. In this paper, we propose an optimal ureter stone detection model utilizing various AI technologies. Specifically, we compared and evaluated AI methods such as machine learning, deep learning, and image processing to find a way to detect urinary tract stones more effectively. For machine learning, we used a representative segmentation method (support vector machine [SVM]). For deep learning, we used a ResNet-50 [9–13] and Fast R-CNN as representative convolutional network (CNN) methods. Image processing was conducted using an efficient watershed method for circular objects considering the shape of stones.
Performance evaluations yielded the best results for ResNet-50, but some false positive (FP) results were derived from general images, especially when the foreign object portion or boundary and grayscale information were very ambiguous. To reduce the number of FP results, further studies are needed to improve weight optimization. Nevertheless, the high sensitivity of the proposed method indicates its accuracy and effectiveness. Fast R-CNN has been found to show a similar level ofperformance to ResNet-50. However, cross-validation indicated significantly lower sensitivity. Finally, SVM faces problems with the value of the function to be used as an input. Specifically, it seems to be too inefficient to perform detection tasks for stone materials due to difficulties in processing value information and data for the entire area. The morphological properties of stone and other objects appear to be inaccurate given differences in information density and height, requiring the processing of additional attribute information as a separate area of interest.
Among the image processing methods, the results of the efficient watershed method for detecting circular objects showed superior performance compared to SVM. This is thought to have been possible because marker-based feature information was used instead of a general gradient image. Although this method involves a relatively simple process, it seems that it yields superior performance compared to SVM algorithms, with clear results and few caveats.
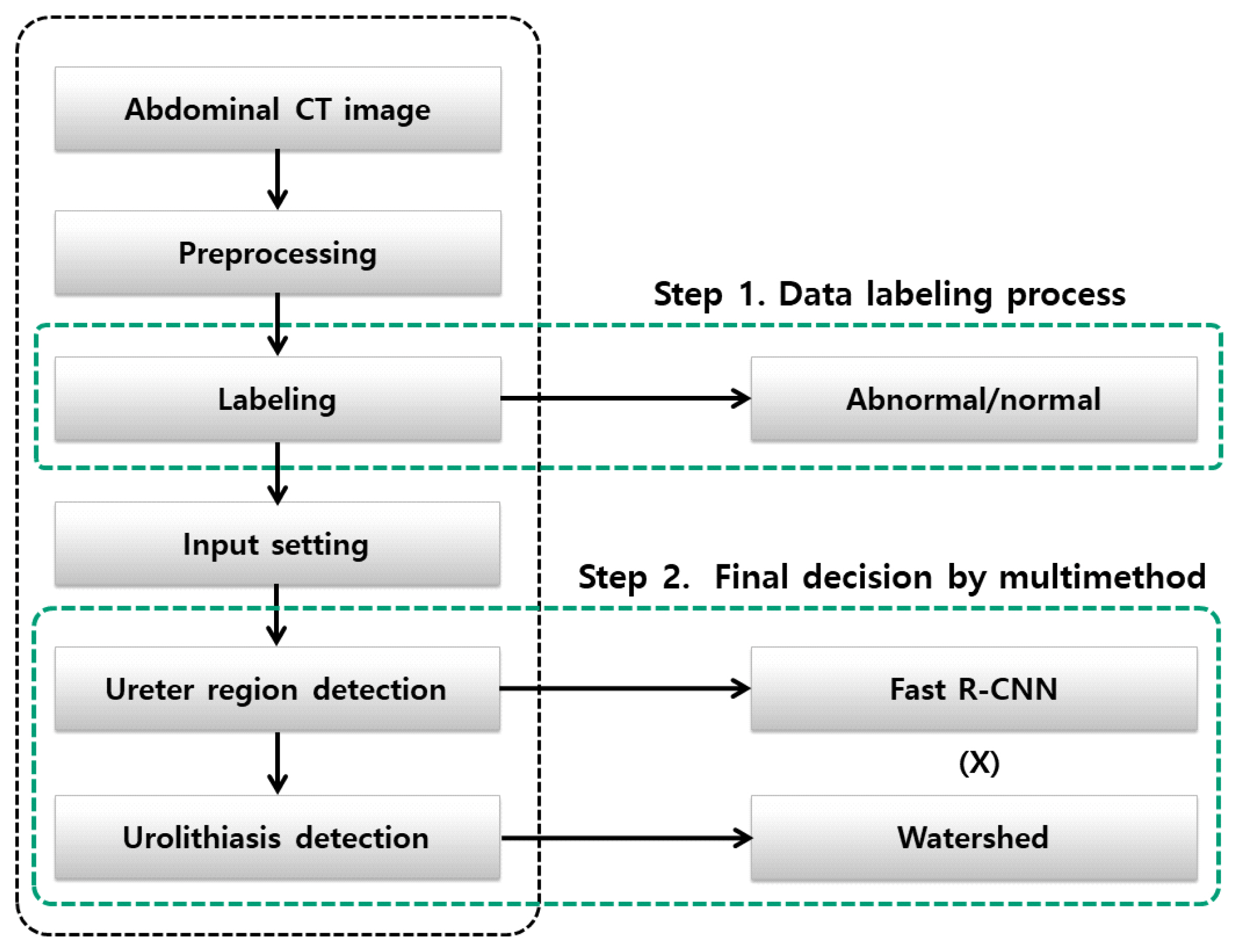
As such, AI research in various fields of urinary tract stone detection has been conducted. Therefore, this study extends the concepts of existing studies by conducting a multimethod application considering accuracy in detecting the location of urinary tract stones. Fig. 1 presents is a conceptual diagram of the whole research. This comparative approach will be described in detail in the next sections.
In this paper, we propose an optimal urinary tract stone detection algorithm based on AI technology. This method was intended to increase the accuracy of urinary tract stone detection by combining deep learning technology and image processing technology. We refer to this method as “polymorphism,” where 2 results are fused to identify the exact location of the urinary tract stone. For the AI technology, this study used Fast R-CNN as a CNN method, and image processing utilized the watershed method for circular objects considering the shape of stones. The results of these 2 methods were merged to determine the final location of the urinary tract stones. First, the ureter region was detected in continuous abdominal images through Fast R-CNN, and then the final position of the ureter stone was derived through comparison with the region detected through the watershed method. Fig. 2 presents a diagram of the proposed method.
Numerous studies in the medical field have demonstrated excellent performance in the field of machine learning with advances in high-performance hardware and big data by applying image recognition, voice classification, recognition, and object detection through deep learning. Among them, convolutional neural networks (CNNs) [14], which are widely used in image recognition, are deep learning models developed to overcome overfitting, local optimal convergence, and gradient extinction by modifying the structure of existing artificial neural networks. CNNs outperform conventional object detection and learning methods that automatically detect features from input images, and have the advantage of being able to extract and learn features from a single structure.
Among them, R-CNN performs affine image warping in each area, as a region-based deep learning technique with CNN functionality, and then generates a region proposal using selective search. An optional search algorithm [15] based on a bottom-up approach measures similarity for each region to integrate or eliminate the region. R-CNN has a complex structure and has the disadvantage of verifying each region; thus, its computation volume increases as it generates and provides 2,000 candidate regions. The CNN distorts and cuts each learning area of the input image; thus, it structures much of the learning area, leading to image deformation and loss problems. To overcome these shortcomings, Fast R-CNN [16] was proposed.
Fast R-CNN is a domain-based deep learning method that generates feature maps using CNNs, excluding fully connected layers of input images and region of interest (RoI). An RoI pooling layer is constructed that extracts fixed-length feature vectors from feature maps so that various learning regions can enter the fully connected layers. Features are extracted through the RoI pooling layer and then learned through the full connection layer. After learning, softmax and cross-entropy are used to classify the object detection part, the classifier and the bounding box regression, and predict the detection area. In R-CNN, the problem of not being able to update CNN features in response to linearly independent SVMs and object domain coordinates (i.e., the problem of complex learning structures), is solved using multi-task loss of Fast R-CNN and end-to-end learning structures. Fig. 3 presents a structure of the Fast R-CNN.
This paper analyzed several abdominal images of patients with urinary tract stones using the corresponding Fast R-CNN. This was done to find the ureter in the continuous images, and the final primary candidate region was detected using the learned ureter feature information.
The watershed method [17,18] was applied for image processing to detect the original shape of the stone. The principle of the watershed algorithm is that when calculating the slope from an original image, the gray level has a high value and a low value, and viewing the slope as having a topographical meaning enables an analysis of the image. The watershed algorithm also assumes that there is a hole in the minimum value of the inclined image, and the water begins to rise little by little through the hole and to merge into another puddle; at this point, a dam is formed to prevent this merger. A line composed of these dams is expressed as a watershed line, and an image included in the watershed line is divided.
The watershed algorithm has a problem of overdifferentiation because it expands the area based on the local minimum. As a method for controlling this oversegmentation, marker control catchment is a method of determining external and internal markers and correcting the calculated slope again to solve the problem of oversegmentation. Marker selection ranges from simple methods to size, shape, location relative distance, and texture range. The important point here is that the use of markers introduces prior knowledge related to segmentation problems.
The area detected through the watershed method was determined as the candidate area of the stone. Geometrically, a stone is a circular object, suitable for the application of the watershed method. Based on the derived stone’s candidate area, we checked whether it overlapped with the previously derived ureter area.
We compared the previously calculated Fast R-CNN-based urinary tract region detection results with the watershed-based urinary tract region detection results. This involved comparing the ureter candidate area and the ureter stone candidate area, and the overlapping area was interpreted as the position of the final ureter stone. The ureter area was considered when evaluating the accuracy of the location of the urinary stones. To determine overlap, the area, derived as 1 through the multiply operation of the ureter candidate area and the urinary tract stone candidate area, was judged as the final urinary tract stone.
The criteria for performance evaluation are the determination of (1) the presence or absence of urinary tract stones and (2) their location. In evaluating the presence or absence of urinary tract stones, the sensitivity, specificity, and accuracy are evaluated by calculating a confusion matrix that compares the results of the proposed model to the correct reference values for the presence or absence of urinary tract stones. The evaluation data comprised 150 cases of urinary tract stones and 150 cases of normal data.
Fig. 4 explains the concepts of intersection and cross-validation for combinatorial calculations, and the sensitivity was calculated using the number of true-positive, FP, and false-negative results. The details are shown in Table 1.
We conducted 10-fold cross-validation, splitting the data into 9 sets for learning and 1 for testing a total of 10 different times. As a criterion for efficiency, the sensitivity and specificity of whether urinary tract stones were detected were evaluated based on a threshold 0.8, and the location of urinary tract stones was evaluated based on an accuracy of 0.8. The confusion matrix for whether urinary tract stones are detected is calculated by deriving the presence or absence of stones as values of 1 and 0, respectively. For the location of urinary tract stones, a confusion matrix was derived by calculating the intersection over union (IoU) value (basis: 0.3) of the derived area.
A comparative evaluation with existing methods was also performed using values from existing studies [8].
As a result of deriving the confusion matrix, the sensitivity and specificity of urinary tract stone detection were calculated to be 0.90 and 0.91, and the accuracy for their position was 0.84. This value was higher than 0.8, which is the standard for accuracy, as shown in the following table. For SVM, it is thought that there is a problem with the feature value used as the input value. Mainly, the morphological and strength information of the stone is processed using it as feature values, which seems to be inefficient for matching throughout the entire area. Morphological information considers density and extensibility, and it seems that inaccurate results were derived if the difference between the characteristics of the stone and other object regions was small. Therefore, it was determined that it would be necessary to use additional characteristic information or to separately cut and process the area of interest.
Among the image processing methods, the watershed method, which is effective for detecting circular objects, showed superior performance compared to SVM. A possible explanation for this is that marker-based feature information was used instead of a general gradient image. Similarly, the feature information used in the marker was the morphological information and intensity of the stone, and the presence of the stone was finally determined through the target marker from the segmentation results. Although this was a relatively simple process, it showed superior results to those obtained using the SVM algorithm.
The proposed method was able to detect urinary stones accurately through a method involving fusion, by first detecting the exact ureter area based on the Fast R-CNN AI method and then judging the candidate area of urinary stones using the watershed method.
AI has started to be applied in various medical fields. The urological system is no exception, and AI research is actively conducted for the entire cycle of analysis, diagnosis, treatment, and management of urological diseases. Most AI applications are being studied as an auxiliary means of patient treatment, and increasingly many models using highly advanced technologies are being developed.
In this paper, we propose an optimal ureter stone detection model utilizing multiple AI technologies. Specifically, the proposed model of urinary tract stone detection merges an AI model and an image processing model, resulting in a multimethod approach. The AI model was used to extract the ureter region as the primary region by applying Fast R-CNN, and the image processing model was used to extract the urinary tract stone candidate areas through the watershed method. The urinary tract area and the urinary tract stone area were compared to determine the overlapping area as the urinary stone.
In an evaluation of the performance evaluation of the proposed model, a sensitivity of 0.90 and a specificity of 0.91 were derived for whether urinary tract stones were detected. These were higher than the reference values of 0.8. In addition, the positional accuracy of urinary tract stones was 0.84, which was also higher than the reference value of 0.8. These findings confirmed the model’s validity. It was possible to compare these results with those obtained using the SVM and watershed methods. The SVM method has a problem with the values of the function to be used as an input. Specifically, it encounters inefficiencies in performing detection tasks for stone materials due to difficulties in processing value information and data for the entire area. Differences in information density and height seem to cause inaccuracies in the morphological properties of stone and other objects, requiring additional attribute information to be processed as a separate area of interest. Among the image processing methods, the efficient watershed method showed superior performance for detecting circular objects compared to SVM. A likely explanation for this is that marker-based feature information was used instead of a general gradient image. Despite the simplicity of the process, it yielded superior results. Thus, the proposed multimethod type method was confirmed to be more accurate and efficient.
In addition to diagnosis, future research will be conducted on real-time surgical guide technology and risk prediction technology for patient management, with potential applications of the resulting algorithms in general surgery.
The performance evaluation of the method proposed herein indicated that it can effectively play an auxiliary role in diagnostic decision-making with a clinically acceptable range of safety. In particular, in the case of ambush stones or urinary stones accompanying ureter polyps, the value that could be obtained through combination therapy based on diagnostic assistance could be evaluated. This study verified the safety and effectiveness of this AI model for the management and prevention of medical disorders, and the findings establish a basis for the use of this multimodal technology by doctors, which could be further evaluated in clinical trials.
NOTES
REFERENCES
1. Mytilekas KV, Oeconomou A, Sokolakis I, Kalaitzi M, Mouzakitis G, Nakopoulou E, et al. Defining voiding dysfunction in women: bladder outflow obstruction versus detrusor underactivity. Int Neurourol J 2021;25:244-51. PMID: 33957716




2. Yu J, Jeong BC, Jeon SS, Lee SW, Lee KS. Comparison of efficacy of different surgical techniques for benign prostatic obstruction. Int Neurourol J 2021;25:252-62. PMID: 33957718




3. Kim HW, Lee JZ, Shin DG. Pathophysiology and management of long-term complications after transvaginal urethral diverticulectomy. Int Neurourol J 2021;25:202-9. PMID: 34610713




4. Jang EB, Hong SH, Kim KS, Park SY, Kim YT, Yoon YE, et al. Catheter-related bladder discomfort: how can we manage it? Int Neurourol J 2020;24:324-31. PMID: 33401353




5. Kwon WA, Lee SY, Jeong TY, Moon HS. Lower urinary tract symptoms in prostate cancer patients treated with radiation therapy: past and present. Int Neurourol J 2021;25:119-27. PMID: 33504132




6. Baser A, Zumrutbas AE, Ozlulerden Y, Alkıs O, Oztekın A, Celen S, et al. Is there a correlation between behçet disease and lower urinary tract symptoms? Int Neurourol J 2020;24:150-5. PMID: 32615677




7. Kim SJ, Choo HJ, Yoon H. Diagnostic value of the maximum urethral closing pressure in women with overactive bladder symptoms and functional bladder outlet obstruction. Int Neurourol J 2022;26(Suppl 1):S1-7. PMID: 35236047




8. Eun SJ, Yun MS, Whangbo TK, Kim KH. A study on the optimal artificial intelligence model for determination of urolithiasis. Int Neurourol J 2022;26:210-8. PMID: 36203253




9. Long W, Xinyu L, Liang G. A transfer convolutional neural network for fault diagnosis based on ResNet-50. Neural Comput Appl 2020;32:6111-24. 

10. Kim JW, Kim SJ, Park JM, Na YG, Kim KH. Past, present, and future in the study of neural control of the lower urinary tract. Int Neurourol J 2020;24:191-9. PMID: 33017890




11. Koonce B. Convolutional neural networks with swift for tensorflow. Image recognition and dataset categorization. Berkeley (CA): Apress; 2021. pp. 63-72.
12. Quach LD, Quoc NP, Thi NH, Tran DC, Hassan MF. Using surf to improve resnet-50 model for poultry disease recognition algorithm. 2020 International Conference on Computational Intelligence (ICCI). 2020 Oct 8 9; Bandar Seri Iskandar, Malaysia: IEEE; 2020. 317-21. doi: 10.1109/ICCI51257.2020.9247698. 
13. Novelita Dwi M, Novamizanti L, Rizal S. Convolutional Neural Network pada klasifikasi sidik jari menggunakan RESNET-50. J Tek Inf (Jutif) 2020;1:61-8. 

14. Esteva A, Kuprel B, Novoa RA, Ko J, Swetter SM, Blau HM, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature 2017;542:115-8. PMID: 28117445




15. Uijlings JRR, van de Sande KEA, Gevers T. Selective search for object recognition. Int J Comput Vis 2013;104:154-71. 

16. Girshick R. “Fast R-CNN,”. 2015 IEEE International Conference on Computer Vision (ICCV). 2015 De 7–13; Santiago, Chile: IEEE; 2015. 1440-8. doi: 10.1109/ICCV.2015.169. 
Fig. 4
The intersection over union (IoU) calculation (left) and concept of cross-validation (right) [8].

Table 1
Performance evaluation results of cross-validation