 |
 |


- Search
| Int Neurourol J > Volume 27(1); 2023 > Article |
|
ABSTRACT
Purpose
The relative roles of urinary sphincter damage, aging, and childbirth in stress urinary incontinence (SUI), have not been established. This study was performed to elucidate the roles of these factors.
Methods
The study included: (1) 8 female cynomolgus monkeys (17–19 years of age and 7–8 vaginal births each); (2) six 5-year-old nulliparous monkeys with surgically created chronic urinary sphincter dysfunction; and (3) six 5-year-old, nulliparous, no-surgery controls. Sedated abdominal leak point pressure (ALPP) and maximum urethral sphincter pressures (MUP) were measured. Sphincters, bladders, and pelvic support muscles were quantified for collagen content. Additionally, bladders were analyzed for collagen fiber thickness, length, and angle using CT-FIRE analysis of Picrosirius red-stained tissues.
Results
Resting MUP values were similar in the controls and older multiparous monkeys (P>0.05). However, aging and multiple births reduced pudendal nerve-stimulated increases in MUP (P<0.05 vs. controls). ALPP values were lower in the older multiparous versus younger groups of monkeys (P<0.05). Sphincter collagen content was greater, and muscle content less, in the injury model (P<0.05 vs. controls). However, these measures were not affected by age and childbirth (P>0.05 vs. young groups). Bladder collagen content was greater, and muscle content less, in the old multiparous monkeys (P<0.05 vs. younger groups). Additionally, collagen fibers were thicker and more angular in the bladders of the older multiparous monkeys than in the other nonhuman primate groups (P<0.05). Pelvic support muscles had higher collagen and lower muscle content in the older multiparous monkeys than in the younger groups of monkeys (P<0.05).
- The goal of this study was to define the effects of aging and childbirth on the structure and function of the urinary sphincter, the urinary bladder and pelvic support in a nonhuman primate model.
- When compared to younger nulliparous monkeys, the older multiparous monkeys had more functional and structural defects in their support muscles and bladders than in the urinary sphincter.
- We conclude that the cause of urinary incontinence in older women that had vaginal child births may be more associated with bladder and pelvis support pathology and specific urinary sphincter pathology.
Stress urinary incontinence (SUI) remains a major urological problem and is 2-fold more prevalent in women than in men [1–4]. SUI in women is often caused by decreased outlet resistance during increased abdominal pressure and can be the cause of urethral hypermobility from deficient pelvic support, and/or inadequate coaptation from intrinsic sphincter deficiency. Current treatments provide symptom relief in some patients [5]. However, correcting the underlying etiology could provide relief for a greater number of women. Furthermore, treatments offer only short-term and incomplete relief [6]. This could be due to the multifactorial nature of SUI, including urinary sphincter, bladder, and pelvic support dysfunction. However, the influences of birthing and aging on these pathophysiologic processes are less well characterized.
To advance treatments for SUI, suitable animal models of lower urinary tract disorders mimicking the underlying pathology of SUI in women are desirable. Several animal models have been described, including simulated childbirth, sphincter muscle removal, and direct damage to the pudendal innervation [7,8]. Vaginal distension models are used to mimic childbirth injury and the results of stretch-induced trauma to the urethra, vagina, and pelvic floor [8]. We have developed a nonhuman primate model of selective nerve damage to the peripheral pudendal nerve branches to create reproducible urinary sphincter dysfunction [9]. We now use this model of selective urinary sphincter injury for comparisons with older nonhuman primates with multiple vaginal births. The goal of the present study was to establish the relative contribution of urinary sphincter injuries versus pathologies of the bladder and pelvic support muscles to SUI.
Twenty adult female cynomolgus monkeys (Macaca fascicularis) were used in this study. Older multiparous monkeys (n=8, 17–19 years of age, with 6–7 live vaginal births) were purchased from Alpha Genesis (Yamasee, SC, USA). Younger nulliparous monkeys (n=12, 5 years of age) were purchased from Charles River (Houston, TX, USA). The younger monkeys were divided into a surgically created urinary sphincter-injured group (n=6), or a no-surgery, no-injury control group (n=6). All monkeys were housed in Wake Forest Animal Resources Program–approved and monitored facilities and received environmental enrichment. All studies were performed with the approval of the Internal Animal Care and Use Committee for the protocol. Monkeys were euthanized prior to necropsy using methods approved by the American Veterinary Medical Association (100 mg of sodium pentobarbital given intravenously to sedated monkeys (ketamine hydrochloride 5 mg/kg given intramuscularly).
The monkeys were sedated with 10 to 15 mg/kg ketamine intramuscularly and then with 1% to 5% isoflurane to induce and maintain surgical anesthesia [9]. The monkeys were prepared for aseptic surgery and anesthetized. A 4-cm-long midline abdominal incision was made from the umbilicus to the pelvic symphysis to expose the pelvic area of the abdomen. The distal urinary tract was approached using gentle dissection of connective tissue just ventral to the bladder, extending dorsal to the bladder neck and caudal 2 cm to each side of the rhabdosphincter. The pudendal nerve branches supplying the sphincter were electrocauterized and then transected. Special care was taken not to damage surrounding structures [9]. The abdomen was closed in 2 layers and postoperative support was given. The monkeys were studied 6 months postsphincter injury. In previously published reports, bladder function/contractility was left undisturbed in this model [9].
In the older multiparous monkeys, sphincter and abdominal leak point pressure (ALPP) tests were done 2 months postarrival and then 3 months later just prior to necropsy [10]. In the monkeys with a surgically created urinary sphincter deficiency, the sphincter pressure and ALPP test were done just prior to the injury procedure and 6 months later. In the noninjured controls, function tests were done at baseline and 6 months later just prior to necropsy.
Urodynamic measures were recorded on anesthetized animals using the Tech Urolab Opus System V in combination with a 6-Fr microtip transducer catheter (Millar Instruments, Houston, TX, USA), as previously described by Badra et al. [9]. Briefly, the catheter was inserted transurethrally, and urethral pressure profilometry was performed by automatically withdrawing the sensor catheter at 0.5 mm per second. Rectal pressure was measured with a balloon catheter attached to a transducer (Life-Tech, Stafford, TX, USA). Using the pressure sensors at its tip, a static urethral pressure profile was recorded on the urodynamic machine, and the maximum urethral pressure (MUP) in the external sphincter region was also recorded. This process was repeated 3 times, and the mean values of urethral pressure measurements were calculated for each animal. MUP was then measured during pudendal nerve stimulation. The pudendal nerve stimulation site was proximal to nerve transection and lateral to the rhabdosphincter. A Sierra Wave ball probe was connected to the nerve-stimulating machine with a setting of 1-ms delay and 1-ms pulse width at 15-mA intensity [9]. The probe was used to stimulate each nerve by direct contact. The urethral catheter was withdrawn automatically while monitoring the changes in urethral pressure.
During the same anesthesia to measure sphincter pressure, bladder function studies were performed using a Goby IV – Goby Wireless Urodynamics System from Laborie (Williston, VT, USA), as described previously by Zambon et al. [10]. The intra-vesical pressure was measured with a 6-Fr nasogastric tube connected to the transducer and abdominal pressure was measured with a 7-Fr T-DOC abdominal catheter placed into the distal rectum proximally to the anal sphincter. The bladder was filled at 5 mL/min with warmed 0.9% sodium chloride and methylene blue solution. The first bladder filling was performed to measure bladder compliance and maximum cystometric capacity (MCC). The MCC was estimated when urinary leakage came out through the urethra in the absence of Valsalva events or detrusor overactivity. ALPP was measured at 50% MCC without the intravesical catheter. Each measurement was repeated 3 times.
Older multiparous monkeys were necropsied 2 each week for 4 weeks. Younger sphincter-injured monkeys were necropsied 2 each week for 3 weeks and timed so the necropsies were 6 months postsphincter injury. The 6 uninjured controls were necropsied 2 each week, while the injured monkeys developed sphincter dysfunction for 6 months. At necropsy, the monkeys were euthanized as described. One liter of saline was infused into the left cardiac ventricle exiting the lanced caudal vena cava to remove residual blood. The gross pathology was documented and photographed as described previously [9,10].
The urinary bladders and the urethra from the bladder outlet to the vaginal opening were removed, weighed, photographed, and measured using calipers. The bladder and the urethral sphincter (thickened urethra just distal to the bladder and extending for approximately 3 cm) were incised transversely into 3 sections (proximal, middle, and distal). The sections were immersed in 4% paraformaldehyde for 72 hours and then transferred to 70% ethanol prior to paraffin embedding. Remaining harvested tissues (ovaries, uterus, pelvic support muscles [levator ani and coccygeus]) were removed, immersed in 10% neutral buffered formalin for 48 hours, and then transferred to 70% ethanol prior to paraffin embedding. Paraffinized tissue was sectioned into 5-μm slices using a microtome, and histology slides were stained with Masson’s trichrome stain (#HT15, Sigma-Aldrich Inc., St. Louis, MO, USA) and Picrosirius red (PSR) (#24901, PolySciences, Warrington, PA, USA) for collagen content analyses [10,11].
Images of histology sections were photographed using an Olympus BX63 microscope (Olympus, Center Valley, PA, USA) with an Olympus DP80 camera (Olympus). Ten randomly selected, evenly spaced images were taken at 200× magnification (for Masson’s trichrome stain) and 100× magnification under the polarized light filter (for PSR stain). For tissues stained with Masson’s trichrome stain, quantitative analysis of fibrosis was performed using the Olympus cellSens dimension software (v.1.16), with the Count and Measure application, to calculate the collagen and noncollagen areas (%) of muscle. The results of 10 images were averaged for each monkey (N=30) for further analysis [9,10]. For tissues stained with PSR under polarized light, collagen fiber quantification for length, width, and straightness was performed using the segmentation software CT-FIRE (Laboratory for Optical and Computation Instrumentation, University of Wisconsin). The results of 10 images were averaged for each monkey [11].
The effects of the animal models (older multiparous, young and no injury, young and sphincter injury) on function and histology were evaluated for each group using 1-way analysis of variance for approximately normally-distributed data, and the 1-way Kruskal-Wallis H-test for nonparametric data. Differences between groups were considered statistically significant at P<0.05. Pairwise comparisons of group mean ranks were performed using the Dunn test (1964) with Bonferroni correction for multiple comparisons. Results are shown as mean±standard error of the mean. All statistical analyses were performed using IBM SPSS Statistics ver. 19.0 (IBM Co., Armonk, NY, USA).
The multiparous monkeys were significantly older (18.2 vs. 4.9 and 5.2 years of age, P<0.05). The body weight was also greater in the multiparous group than in the 2 nulliparous groups (7.1 vs. 4.3 and 4.4 kg, P<0.05).
The MUP was lowest in the younger nulliparous monkeys that had received urinary sphincter injury (27.7 cmH2O) compared to 63.7 cmH2O in the young nulliparous monkeys with no sphincter injury and 63.3 cmH2O in the older multiparous monkeys (P<0.05). Pudendal nerve stimulation increased the MUP in the young nulliparous monkeys without sphincter injury, but not in the other 2 groups (P<0.05). The ALPPs were lowest in the older multiparous monkeys (20 cmH2O) (P<0.05 vs. the other 2 groups). The ALPP was lower in the young nulliparous monkeys with sphincter injury (53 cmH2O) than in the no-injury group, but still higher than in the older monkeys (P<0.05). The ALPP was highest in the young nulliparous monkeys without sphincter injury (P<0.05 vs. the other 2 groups) (Table 1).
While the bladder weight was greater in the multiparous monkeys (P<0.05), the ratios of bladder weight to body weight were similar among groups (P>0.05). The bladder capacities were similar among groups (P>0.05). However, bladder wall thickness was less in the multiparous monkeys (2.4 mm) than in the 2 nulliparous monkey groups (4.0 and 3.1 mm, P<0.05). Urinary sphincter weight was least in the nulliparous group (with injury) compared to the other 2 groups (3.1 vs. 4.5 and 4 g, P<0.05). The levator ani and coccygeus muscle weights were similar among groups (P>0.05) (Table 2).
Bladder collagen content was greater in the older multiparous monkeys than in the other groups (37.2% vs. 23.9% and 22.4%, P<0.05). Conversely, bladder muscle content was greater in the 2 younger groups than in the older multiparous group (55% and 45% vs. 38.4%, P<0.05). A similar pattern was observed in urinary sphincters, in that collagen content was greater and muscle content was less in the multiparous monkeys than in the other 2 groups (P<0.05). Again, a similar pattern was observed in the support muscles (levator ani and coccygeus) in that the collagen content was greatest and muscle content least in the older multiparous monkeys compared to the younger nulliparous monkeys (P<0.05) (Table 3).
Collagen fiber width was greater in the old multiparous monkeys (4.3 pixels) than in the young nulliparous, no-injury monkeys (3.0 pixels, P<0.05) (Fig. 1). Collagen fiber straightness was least in the older multiparous monkeys compared to the 2 younger monkey groups (P<0.05, Fig. 2). Collagen fiber length was also the smallest in the older multiparous monkeys compared to the 2 younger groups (P<0.05, Fig. 3).
The major findings of this study on nonhuman primates are that aging and multiple childbirths appeared to be more associated with bladder dysfunction, reduced pelvic muscle support, and compensatory response to neural stimulation than with selective urinary sphincter damage.
The pathogenesis of SUI is considered to include both disruption of the supportive connective tissues of the bladder and urethra and weakening of the muscular structures of the pelvic floor, bladder neck, and urethral sphincters, all leading to reduced urethral closure pressure, which functionally underlies the symptoms of SUI [12–14]. This suggests a multifactorial etiology of SUI. Unfortunately, we do not yet have a complete understanding of the relative importance and interaction of these factors. New studies on continence mechanisms may provide insight into these relationships [15]. Such studies would benefit from an animal model that closely mirrors the underlying pathophysiology of SUI observed in women and could assist in the development of more effective treatments for women with SUI. Nonhuman primates were used in this study because their lower urinary tract has many structural similarities to that of humans, which should increase the translational value of the results [16]. In addition, nonhuman primates are subject to age-related diseases similar to those of human beings [17].
Current data from animal and human studies indicate that aging affects lower urinary tract function through a reduction in ultrastructural (pelvic support structure viability) and physiological (reduced neuronal control) determinants of continence [18–20]. In the present study, both urinary tract structure and function were evaluated, including analysis of the urinary sphincter, the urinary bladder, and pelvic support muscles in an attempt to assign their relative roles in SUI. Two models (one that mirrored age and childbirth vs. one that reflected selective damage to the urinary sphincter) were compared to elucidate these relationships.
While the bladder walls from the older multiparous monkeys were thinner than those of the young animals, there were no significant differences in bladder sizes. Not unexpectedly, older multiparous monkeys had less muscle and more collagen in the bladder walls than the younger groups. Furthermore, this group had greater collagen fiber width and reduced collagen fiber length, making the fibers more angular. An analysis of collagen fiber parameters, such as length, width, angularity, and straightness, in normal and pathologic disease states may help unlock the conditions that favor irreversible fibrosis [21–23]. Relevant to this study, the collagen fiber characteristics found in the older multiparous monkeys have been associated with reduced bladder biomechanics and strength [24]. This underlines the importance of measuring more than just collagen amounts.
While selective urinary injury reduced resting MUP, aging and childbirth had no effect on this measure. However, pudendal nerve-stimulated increases in MUP were reduced in both models. ALPP was also reduced in both models, but to a greater extent in the older/multiparous monkeys. These results suggest that aging and multiple births may be associated with bladder dysfunction and reduced pelvic muscle support, rather than direct urinary sphincter dysfunction at rest. The reduced response to neural stimulation suggests a delayed or reduced response of the pelvic floor to increased Valsalva events. Thus, both types of dysfunction could contribute to the reduced control of continence by somewhat different mechanisms. Progressive age-dependent declines in maximal cystometric bladder capacity, average and maximal urine flow rate, and MUP in women visiting their doctors have been reported [25]. In these women, voiding time and postvoiding residual urine volume also significantly increased with age. An age-dependent decrease in MUP can be found even in the absence of vaginal parity and is associated with the occurrence and severity of SUI.
In the present study, pelvic support muscles had higher collagen and lower muscle content in the older multiparous monkeys than in the younger groups of monkeys (P<0.05). There may be several explanations for this; for instance, the decrease in the pelvic floor response could be neuropathic and result from stretch injury and sarcopenia from aging. Selective neural injury, as in the present sphincter injury model, did not affect the pelvic floor.
A limitation of the present study is the inherent difficulty of mimicking clinical conditions using animal models. The lifespans and risks of age-related diseases in animals do not always correspond to what is found in humans. Nevertheless, we believe that our data have transitional value.
In conclusion, using these 2 nonhuman primate models, it appears that aging and multiple vaginal childbirths do not directly affect sphincter structure and function to any great degree, as opposed to the surgical sphincter model, but does interfere with bladder and pelvic support, muscle structure, and ALPP. These findings support the hypothesis that urinary incontinence in older women with a history of vaginal deliveries is more a result of bladder and pelvic floor dysfunction than urinary sphincter dysfunction. This could explain, in part, why regenerative or bulking treatments focusing on the urinary sphincter alone are not entirely successful.
NOTES
Research Ethics
All studies were done with approval from the Wake Forest Institutional Animal Care and Use Committee, according to all governmental regulations for the use of animals in Biomedical Research and by the American Veterinary Medical Association.
AUTHOR CONTRIBUTION STATEMENT
• Conceptualization: JKW, KEA, GB
• Data curation: JKW, SL
• Formal analysis: JKW
• Funding acquisition: JKW, KEA
• Methodology: JKW, SL, GB
• Project administration: JKW, SL, GB
• Visualization: K Williams, KEA, SL, GB
• Writing - original draft: JKW, KEA, SL, GB
• Writing - review & editing: JKW
REFERENCES
2. Irwin DE, Kopp ZS, Agatep B, Milsom I, Abrams P. Worldwide prevalence estimates of lower urinary tract symptoms, overactive bladder, urinary incontinence and bladder outlet obstruction. BJU Int 2011;108:1132-8. PMID: 21231991


3. Hunskaar S, Lose G, Sykes D, Voss S. The prevalence of urinary incontinence in women in four European countries. BJU Int 2004;93:324-30. PMID: 14764130


4. Minassian VA, Stewart WF, Wood GC. Urinary incontinence in women: variation in prevalence estimates and risk factors. Obstet Gynecol 2008;111:324-31. PMID: 18238969

5. Shah SM, Gaunay GS. Treatment options for intrinsic sphincter deficiency. Nat Rev Urol 2012;9:638-51. PMID: 23027065



6. Morling JR, McAllister DA, Agur W, Fischbacher CM, Glazener CM, Guerrero K, et al. Adverse events after first, single, mesh and non-mesh surgical procedures for stress urinary incontinence and pelvic organ prolapse in Scotland, 1997–2016: a population-based cohort study. Lancet 2017;389:629-40. 
7. Amend B, Harland N, Knoll J, Stenzl A, Aicher WK. Large animal models for investigating cell therapies of stress urinary incontinence. Int J Mol Sci 2021;22:6092. PMID: 34198749



8. Hijaz A, Daneshgari F, Sievert KD, Damaser MS. Animal models of female stress urinary incontinence. J Urol 2008;179:2103-10. 
9. Badra S, Andersson KE, Dean A, Mourad S, Williams JK. A nonhuman primate model of stable urinary sphincter deficiency. J Urol 2013;189:1967-74. PMID: 23022012


10. Zambon JP, Patel M, Hemal A, Badlani G, Andersson KE, Magalhaes RS, et al. Nonhuman primate model of persistent erectile and urinary dysfunction following radical prostatectomy: feasibility of minimally invasive therapy. Neurourol Urodyn 2018;37:2141-50. PMID: 30168617




11. Bennington J, Lankford S, Magalhaes RS, Shankle D, Fanning J, Kartini C, et al. Chemokine therapy in cats with experimental renal fibrosis and in a kidney disease pilot study. Front Vet Sci 2021;8:646087. PMID: 33748219



12. Falah-Hassani K, Reeves J, Shiri R, Hickling D, McLean L. The pathophysiology of stress urinary incontinence: a systematic review and meta-analysis. Int Urogynecol 2021;32:501-52. PMID: 33416968




13. Schmid FA, Williams JK, Kessler TM, Stenzl A, Wilhelm K, Aicher WK, et al. Treatment of stress urinary incontinence with muscle stem cells and stem cell components: chances, challenges and future prospects. Int J Mol Sci 2021;22:3981. PMID: 33921532



14. Wei JT, De Lancey JO. Functional anatomy of the pelvic floor and lower urinary tract. Clin Obstet Gynecol 2004;47:3-17. PMID: 15024268


15. Andersson KE, Birder L, Chermansky C, Chess-Williams R, Fry C. Are there relevant animal models to set research priorities in LUTD? ICI-RS 2019. Neurourol Urodyn 2020;39(Suppl 3):S9-15. PMID: 32662562




16. Gratzke C, Jarajapu YP, Christ GJ, Kaplan JR, Williams JK, Andersson KE, et al. Effects of long-term dietary soy treatment on female urethral morphology and function in ovariectomized nonhuman primates. J Urol 2008;180:2247-53. PMID: 18804802


17. Kaplan JR. Modeling women’s health with nonhuman primates and other animals. ILAR J 2004;45:83-8. PMID: 15111729


18. Lau HH, Su TH, Huang WC. Effect of aging on lower urinary tract symptoms and urodynamic parameters in women. Taiwan J Obstet Gynecol 2021;60:513-6. PMID: 33966738


19. Elbadawi A, Diokno AC, Millard RJ. The aging bladder: morphology and urodynamics. World J Urol 1998;16:Suppl 1. S10-34. PMID: 9775413



20. Birder LA, Kullmann AF, Chapple CR. The aging bladder insights from animal models. Asian J Urol 2018;5:135-40. PMID: 29988876



21. Pointer KB, Clark PA, Schroeder AB, Salamat MS, Eliceiri KW, Kuo JS. Association of collagen architecture with glioblastoma patient survival. J Neurosurg 2017;126:1812-21. 
22. Zhou ZH, Ji CD, Xiao HL, Zhao HB, Cui YH, Bian XW. Reorganized collagen in the tumor microenvironment of gastric cancer and its association with prognosis. J Cancer 2017;8:1466-76. PMID: 28638462



23. Devarasetty M, Dominijanni A, Herberg S, Shelkey E, Skardal A, Soker S. Simulating the human colorectal cancer microenvironment in 3D tumor-stroma co-cultures in vitro and in vivo. Sci Rep 2020;10:9832. 

24. Gilbert TW, Wognum S, Joyce EM, Freytes DO, Sacks MS, Badylak SF. Collagen fiber alignment and biaxial mechanical behavior of porcine urinary bladder derived extracellular matrix. Biomaterials 2008;29:4775-82. PMID: 18801572



25. Al-Mukhtar Othman J, Åkervall S, Milsom I, Gyhagen M. Urinary incontinence in nulliparous women aged 25–64 years: a national survey. Am J Obstet Gynecol 2017;216:149e1-149.e11.
Fig. 1
Collagen fiber width. The 3 study groups were: (1) young adult monkeys, (2) young adult monkeys that had received a surgical urinary sphincter injury 6 months prior to the study, and (3) older adult multiparous monkeys. Analysis of variance was significant (P<0.05). Values are pixel widths. *P<0.05 vs. the other 2 groups with post hoc analysis. Values are presented as mean±standard error of the mean.
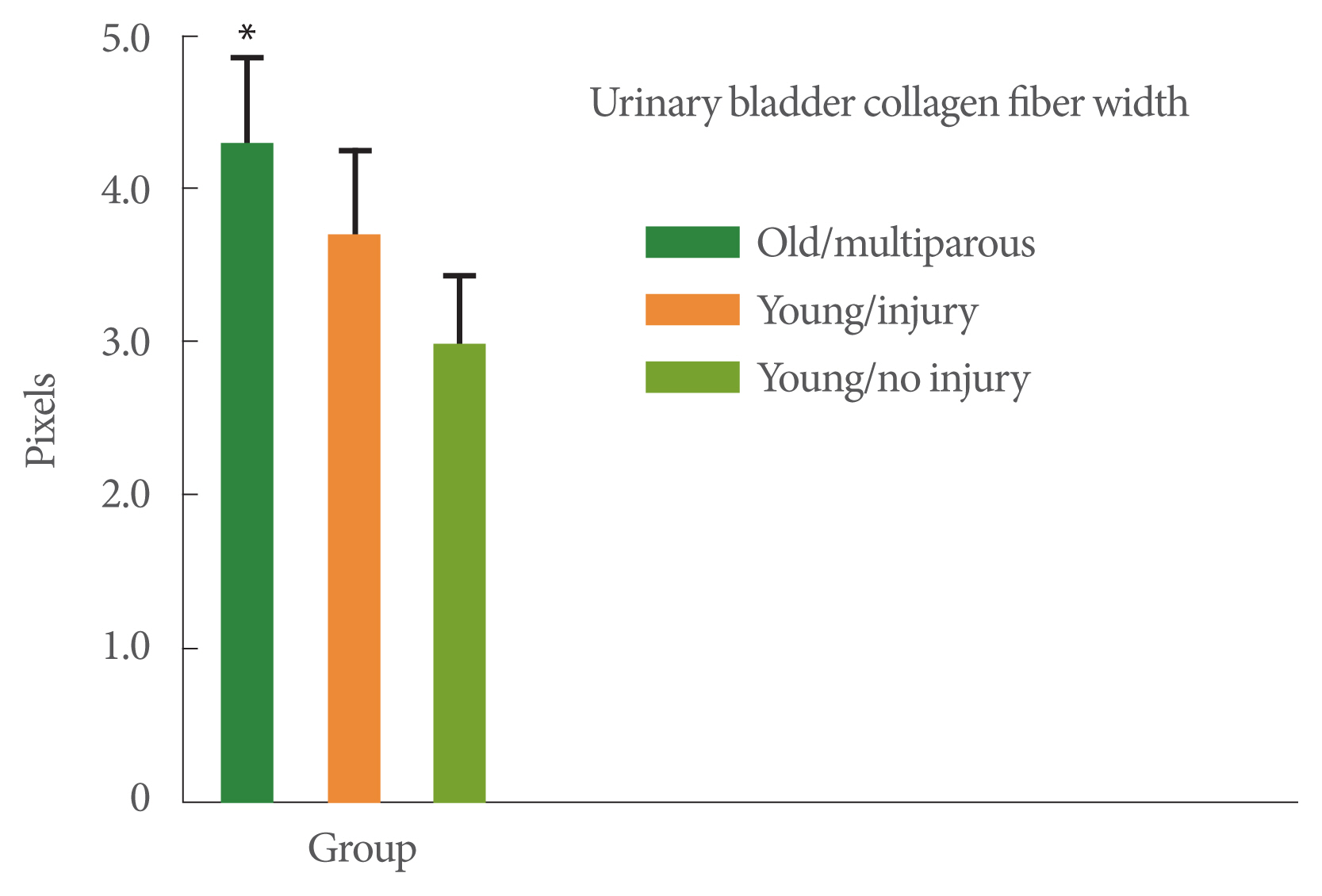
Fig. 2
Collagen fiber straightness. The 3 study groups were: (1) young adult monkeys, (2) young adult monkeys that had received a surgical urinary sphincter injury 6 months prior to the study, and (3) older adult multiparous monkeys. Values are the frequency of straight fibers. Analysis of variance was significant (P<0.05). *P<0.05 vs. the other groups with post hoc analysis. Values are presented as mean±standard error of the mean.

Fig. 3
Fiber length. The 3 study groups were: (1) young adult monkeys, (2) young adult monkeys that had received a surgical urinary sphincter injury 6 months prior to the study, and (3) older adult multiparous monkeys. Values are the frequency of longer fibers. Analysis of variance was significant (P<0.05). *P<0.05 vs. the other groups with post hoc analysis. Values are presented as mean±standard error of the mean.

Table 1
Functional measures
| Measures | Young nulliparous with no sphincter injury | Young nulliparous urethral sphincter injury | Older multiparous | P-value |
|---|---|---|---|---|
| Resting MUP (cmH2O) | 63.7±8 | 27.7±6a) | 63±3 | 0.01a) |
| MUP/nerve stimulation (cmH2O) | 86±4a) | 31±6 | 64±4 | 0.04b) |
| Abdominal leak point pressure (cmH2O) | 73±10 | 53±7a) | 20±4c) | 0.04a)/0.04c) |
The 3 study groups were: (1) young adult monkeys, (2) young adult monkeys that had received a surgical urinary sphincter injury 6 months prior to the study, and (3) older adult multiparous monkeys.
Results presented for resting maximum urethral pressure (MUP), pudendal nerve-stimulated MUP, and abdominal leak point pressure.
Table 2
Tissue weights and sizes
| Measures | Young nulliparous with no sphincter injury | Young nulliparous with urethral sphincter injury | Older multiparous | P-value |
|---|---|---|---|---|
| Bladder weight (g) | 5.5±0.2 | 5.6±0.2 | 8.4±0.5a) | 0.03a) |
| % Bladder weight to body weight | 0.14±0.3 | 0.16±0.5 | 0.15±0.3 | NS |
| % Bladder capacity (mL) to body weight | 50±10 | 45±8 | 52±13 | NS |
| Bladder thickness (mm) | 40±0.05 | 3.1±0.07a) | 2.4±0.07a) | 0.05a) |
| Urinary sphincter weight (g) | 4.5±0.05 | 3.1±0.3a) | 4.0±0.4 | 0.05a) |
| Weight of the levator ani muscle (g) | 3.4±0.4 | 3.6±0.3 | 3.9±0.4 | NS |
| Weight of the coccygeus muscle (g) | 2.9±0.4 | 3.6±0.2 | 3.5±0.2 | NS |
Table 3
Tissue collagen and muscle content
| Measures | Young nulliparous with no sphincter injury | Young nulliparous with urethral sphincter injury | Older multiparous | P-value |
|---|---|---|---|---|
| Bladder collagen content (% of total) | 22.4±3 | 23.9±4 | 37.2±3a) | 0.05a) |
| Bladder muscle content (% of total) | 55±4 | 45±5 | 38±4a) | 0.3a) |
| Sphincter collagen content (% of total) | 31±4 | 63±6a) | 40±5 | 0.05a) |
| Sphincter muscle content (% of total) | 70±5 | 45±4a) | 65±6 | 0.04a) |
| Levator ani collagen (% of total) | 22±5 | 24±7 | 68±10a) | 0.01a) |
| Coccygeus muscle content (% of total) | 68±4 | 65±5 | 32±7a) | 0.03a) |





