Aging-Associated Changes in Oxidative Stress Negatively Impacts the Urinary Bladder Urothelium
Article information
Abstract
Purpose
Lower urinary tract symptoms are known to significantly increase with age, negatively impacting quality of life and self-reliance. The urothelium fulfills crucial tasks, serving as a barrier protecting the underlying bladder tissue from the harsh chemical composition of urine, and exhibits signaling properties via the release of mediators within the bladder wall that affect bladder functioning. Aging is associated with detrimental changes in cellular health, in part by increasing oxidative stress in the bladder mucosa, and more specifically the urothelium. This, in turn, may impact urothelial mitochondrial health and bioenergetics.
Methods
We collected mucosal tissue samples from both young (3–4 months old) and aged (25–30 months old) rats. Tissue was evaluated for p21-Arc, nitrotyrosine, and cytochrome C expression by western immunoblotting. Urothelial cells were cultured for single-cell imaging to analyze basal levels of reactive oxygen species and the mitochondrial membrane potential. Mitochondrial bioenergetics and cellular respiration were investigated by the Seahorse assay, and measurements of adenosine triphosphate release were made using the luciferin-luciferase assay.
Results
Aging was associated with a significant increase in biomarkers of cellular senescence, oxidative stress, and basal levels of reactive oxygen species. The mitochondrial membrane potential was significantly lower in urothelial cell cultures from aged animals, and cultures from aged animals showed a significant decrease in mitochondrial bioenergetics.
Conclusions
Aging-related increases in oxidative stress and excessive reactive oxygen species may be contributing factors underlying lower urinary tract symptoms in older adults. The mechanisms outlined in this study could be utilized to identify novel pharmaceutical targets to improve aging-associated bladder dysfunction.
INTRODUCTION
The prevalence of lower urinary tract symptoms (LUTS), characterized by problems regarding storage and/or voiding of urine, is known to significantly increase with age [1,2]. Multiple bladder components may become dysfunctional with age, including mucosal, muscular, stromal, and neural elements and this dysfunction can also involve surrounding structures (e.g., the prostate) [3,4]. Therefore, in an aging society, LUTS are highly relevant and increasingly important [5].
The inner surface of the bladder wall is covered with a mucosal membrane. This bladder mucosa consists of a multilayered urothelium and underlying lamina propria. The urothelium is highly specialized and has important functions, including acting as a barrier to protect the underlying tissue from the harsh chemical composition of urine [6]. The urothelium furthermore fulfills essential roles in the execution of proper signaling processes in the lower urinary tract (LUT). Stress on the bladder wall induces the release of signaling molecules (e.g., adenosine triphosphate [ATP], acetylcholine, or nitric oxide) from the urothelium, which influences the activity of underlying detrusor muscle layers or afferent nerve fibers located in the lamina propria in close proximity to the urothelium [7-10]. The structural and functional integrity of the urothelium may be diminished in a number of bladder disorders, and damage to the urothelium is a likely contributor to the development or exacerbation of LUTS [11,12].
Healthy mitochondria are essential for the maintenance of proper urothelial function. Mitochondria, which are the primary source of oxidative stress, are particularly susceptible to ageassociated abnormalities [13]. The increased concentration of reactive oxygen species (ROS) and reactive nitrogen species (RNS) is assumed to be one of the underlying causes of the aging process [14]. Oxidative phosphorylation, which is the main mechanism underlying ATP synthesis, is negatively affected by increases in ROS and RNS. Therefore, increased oxidative stress caused by the gradual accumulation of ROS and RNS has been proposed to significantly impact the healthy functioning of the LUT [15,16]. Previous research has indicated that there is a positive relationship between the expression of ROS in urothelial cells (UTCs) and aging in mice [17].
Although aging-related LUT dysfunction in rodents has been previously reported [18], the underlying mechanisms by which aging-related processes influence urothelial health and bladder functioning are not yet fully understood. In the current study, we aimed to further elucidate aging-related changes in urothelial and mucosal biochemistry. We investigated biomarkers of cellular senescence and oxidative stress in mucosal tissue samples collected from young-mature and aged rodents, and assessed cellular respiration, mitochondrial function, and basal ROS in UTC cultures. We hypothesized that aging is associated with augmented oxidative stress, which, over time, may damage multiple components of the LUT system, leading to LUTS in older adults.
MATERIALS AND METHODS
Animals and Tissue Collection
This study utilized female young, though mature (3–4 months old) and aged (25–30 months old) Fischer 344 rats ([F344] Charles River, Wilmington, MA, USA and the National Institute on Aging rodent colony). Animals were kept in standard housing with 12-hour light/dark cycles and ad libitum access to food and water. Mucosal tissue (separated from the detrusor muscle by blunt dissection) was obtained after sacrificing animals by exsanguination during isoflurane anesthesia.
Western Immunoblotting
After surgically separating the urinary mucosa (defined as the urothelium and lamina propria) from the smooth muscle, tissue was homogenized using Lysing Matrix D in a FastPrep 24 instrument (MP Biomedicals, Solon, OH, USA) in HBSS (5mM KCl, 0.3mM KH2PO4, 138mM NaCl, 4mM NaHCO3, 0.3mM Na2HCO3, 0.3mM Na2HPO4, 5.6mM glucose, 10mM HEPES, 1mM DL-dithiothreitol, and 1mM EDTA, pH 7.4) containing a complete protease inhibitor cocktail (1 tablet/10 mL, Roche, Indianapolis, IN, USA) and a phosphatase inhibitor cocktail (Sigma, St. Louis, MO, USA 1:100). After centrifugation (16,200×g; 15 minutes at 4°C), we suspended the membrane pellets in lysis buffer containing 0.3M NaCl, 50mM Tris-HCl (pH 7.6) and 0.5% Triton X-100 and the same concentration of protease inhibitors as above in order to prepare the membrane protein fraction. We then incubated the suspensions on ice, followed by centrifugation (16,200×g; 15 minutes at 4°C). The supernatants were combined and total protein concentrations were assessed using the Pierce BCA protein assay (Thermo Scientific, Waltham, MA, USA). After denaturation (100°C for 5 minutes) with Laemmli sample buffer, lysate from each sample was separated on a 4%–15% TGX Stain-Free sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel (Bio-Rad Laboratories, Hercules, CA, USA). Total protein for each sample was measured using Bio-Rad Stain Free SDS-PAGE gel technology as a loading control. Ultraviolet-activated protein fluorescence was imaged on a ChemiDoc MP (Bio-Rad). Proteins were transferred to polyvinylidene fluoride membranes. We then incubated the membranes in 5% (w/v) dried milk dissolved in TBS-T (20mM Trizma, 137mM NaCl, 0.1% Tween-20, pH 7.6). Membranes were washed with TBS-T, and left to incubate overnight at 4°C with primary antibodies (Synaptic Systems mouse monoclonal anti-p21-Arc antibody: 1:1,000; 303011; Göttingen, Germany [N=14 young, 16 aged]; Abcam mouse anticytochrome C AB13575: 1:1,000; RRID AB_300470; Cambridge, MA, USA [N=6 young, 7 aged]; and Cell Signaling rabbit antinitrotyrosine 9691:1:1,000, RRID AB_331634; Danvers, MA, USA [N=8 young, 11 aged]) diluted in TBS-T containing 5% (w/v) milk (p21-Arc, cytochrome C) or 5% (w/v) bovine serum albumin (BSA) (nitrotyrosine). Membranes were washed in TBS-T and incubated with secondary antibodies (sheep anti-mouse HRP (1:5,000; GE Amersham, Pittsburgh, PA, USA) or donkey anti-rabbit HRP (1:10,000; Advansta, San Jose, CA, USA) for 1 hour in 5% (w/v) milk. The membranes were then washed, incubated in WesternBright Quantum (Advansta, Menlo Park, CA, USA), and imaged on a ChemiDoc MP (Bio-Rad). The volume of each protein species was measured and normalized to total protein using Image Lab software (Bio-Rad).
Primary bladder UTC cultures
Bladders from both young and aged F344 rats were incubated in dispase (2.5 mg/mL; Worthington Biochemical, Lakewood, NJ, USA) overnight at 4°C. UTCs were collected by gentle scraping, placed in trypsin (0.25%; 10–15 minutes, 37°C), dissociated and plated on collagen-coated glass coverslips at a density of 0.3×106 cells/mL. Preparations were used within 48–96 hours. Previous research has indicated that UTC cultures retain their structural and functional properties in short term primary cultures [8,19]. We verified the epithelial nature of cultures using the epithelial cell marker cytokeratin 17, which stained >95% of cultured UTCs, comparable to previous results [20]. In all experiments reported here, each culture contained UTCs obtained from a single animal.
Reagents
We dissolved carbonyl cyanide-4-(trifluoromethoxy) phenylhydrazone (FCCP), oligomycin, and rotenone in dimethyl sulfoxide in a 100-mM stock solution. ATP was dissolved in water in a 10- or 100-mM stock solution. Reagents were diluted to working concentrations using HBSS or cell culture media.
Seahorse assay
UTCs were seeded (0.24×106 cells/mL) into the wells of an SF cell culture microplate and became ~90% confluent following 2–3 days in culture. Cells obtained from all animals (n=4 rats per age group) were assayed simultaneously in 2 separate experiments using the Seahorse XF96 Extracellular Flux analyzer, which simultaneously measures 2 energy-producing pathways in real time: mitochondrial respiration (oxygen consumption rate [OCR]) and glycolysis (extracellular acidification) in living cells. Once basal levels of energy production were established, the cells were stimulated by exposure to 3 compounds that shift cellular bioenergetics: oligomycin, FCCP, and rotenone. Following the addition of 200 μL of protein lysis buffer, the BCA assay was performed to measure protein (results normalized to total protein content).
ATP release
Cultured UTCs (N=3 rats per age group) were placed into a bath perfusion system set at a flow rate of 0.4 mL/min and perfused with HBSS (5mM KCl, 0.3mM KH2PO4, 138mM NaCl, 4mM NaHCO3, 0.3mM Na2HPO4, 5.6mM glucose, 2mM CaCl2, 1mM MgCl2, and 10mM HEPES, pH 7.4, 300 mOsm). A 100 μL sample of perfusate was collected every 60 seconds, and ATP measurements were calculated based on the luciferinluciferase reaction using a standard curve (Adenosine Triphosphate Assay Kit, Millipore Sigma, St. Louis, MO, USA; Glomax 20/20 Luminometer, Promega, Madison, WI, USA). The background release of ATP from the cultures was measured for 15–20 minutes. Cell swelling, used as a surrogate for cell stretching, was tested using a hypotonic stimulus (HBSS containing 69 mM NaCl, 180 mOsm) applied through the perfusion system for another 15–20 minutes. ATP data were analyzed by calculating the area under the curve during each portion of the experiment and normalized by the protein content in the cell culture.
Single-Cell Imaging
Cultured UTCs (N=4 rats per age group) were loaded with tetramethylrhodamine methyl ester (TMRM; 25nM, 10 minutes, 37°C) (Thermo Fisher, Waltham, MA, USA) in order to estimate the mitochondrial membrane potential (Ψm), or dihydrorhodamine 123 (DHR123; 5μM, 30 minutes, 37°C) (Thermo Fisher) to measure ROS. TMRM is a cell-permeant fluorescent dye sequestered by active mitochondria, and the intensity of the fluorescence is proportional to Ψm (i.e., lower fluorescence suggests depolarized Ψm; Ex/Em 544/574 nm). DHR123 is a cell-permeant ROS indicator localizing in the mitochondria, where it is oxidized to cationic rhodamine 123 and exhibits green fluorescence (Ex/Em 488/520 nm). Cells were imaged using an epifluorescence microscope (Olympus IX70; 40× oil objective) and HCImaging software (Hamamatsu Photonics, Bridgewater, NJ, USA) with compounds applied using a gravity-driven application pipette. For analysis, an area of interest was drawn around single cells, and the average fluorescence intensity of pixels included in this area was taken as a single measurement. The fluorescence intensity measured in an area without cells was subtracted to control for the background. For TMRM and DHR123 imaging, pictures of 10–12 random areas per coverslip were taken using the same exposure settings for all coverslips. Data were quantified as fluorescence arbitrary units. For baseline ROS, cells were washed with HBSS and imaged as described above.
Statistics
Data were analyzed in GraphPad Prism 6 (GraphPad, La Jolla, CA, USA). Statistical analysis was performed using the Student t-test or the Welch t-test as applicable to compare young and aged animals. Asterisks in the figures indicate P ≤ 0.05 (*), P≤ 0.01 (**), or P≤0.001 (***).
RESULTS
We first characterized the presence of urothelial cellular senescence using a well-established senescence marker, p21-Arc, in mucosal samples. p21 acts as an inhibitor of the cell cycle and is significantly higher in mucosal tissue from aged rodents (Fig. 1A). The findings illustrated in Fig. 1B-C are consistent with the biomarkers associated with apoptosis and risk for disease. For example, we found significant increases of cytochrome C in aged mucosal tissue (Fig. 1B). Cytochrome C is present in the mitochondrial inner membrane and associated with cell death. We also found increased expression of nitrotyrosine in aged mucosa (Fig. 1C). Nitrotyrosine is considered an indicator of ROS, oxidative damage, and apoptotic cell death. As further support for increased ROS and oxidative stress within the urothelium, we used a cell-permeant reagent, DHR123, which is used to detect ROS, in isolated UTCs. DHR123 itself is nonfluorescent until oxidized to rhodamine 123 in the mitochondria. Our findings revealed that the basal production of ROS was significantly higher in UTC cultures from aged bladders than in cultures from young animals (P≤0.001) (Fig. 1D).
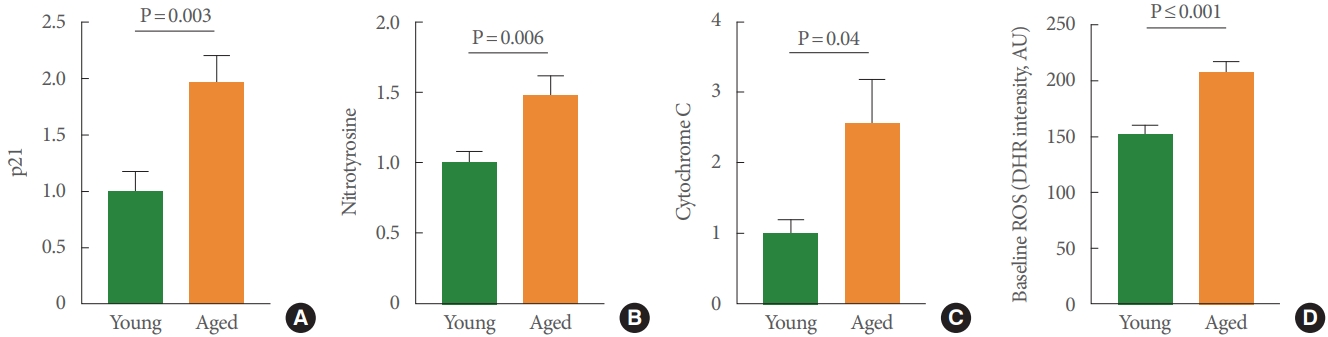
Bar graphs depicting age-associated changes in levels of p21-Arc (P=0.003) (A), nitrotyrosine (P=0.006) (B), cytochrome C (P=0.04) (C), and baseline ROS (P≤0.001) (D). N: numbers, indicating numbers of young versus aged rats per group, are shown in the respective bar graph. ROS, reactive oxygen species.
We then aimed to investigate the detrimental effects of increased oxidative stress and ROS production in UTCs from aged animals on mitochondrial health. Ψm is an essential component of energy storage and a key indicator of mitochondrial activity. We used a cell-permeant dye, TMRM, which accumulates in healthy, active mitochondria with intact membrane potentials. Our findings revealed that TMRM signal intensity was significantly lower in UTCs from aged bladders (indicating loss of membrane potential) than in UTCs isolated from young bladders (Fig. 2).

(A) Bar graphs depicting the TMRM intensity in cultured UTCs from young and aged rat bladders (N=4 each). Ψm was found to differ significantly (P=0.01) between age groups. (B, C) Representative images of TMRM staining in cultured UTCs from young and aged animals. TMRM, tetramethylrhodamine methyl ester; UTC, urothelial cell.
The Seahorse test is the gold standard for measuring mitochondrial function in a number of cell types, and we used this method to assess the bioenergetic profile of young versus aged UTC mitochondria (Fig. 3A). After the basal OCR was determined, oligomycin (2.5μM), a complex V inhibitor, was administered to quantify OCR unrelated to ATP synthesis. The resulting OCR represents the proton leak across the inner mitochondrial membrane. Next, UTCs were exposed to the uncoupler FCCP (0.7μM) to stimulate the maximal respiratory rate by separating oxygen consumption from ATP production. The resulting collapse of the Ψm triggers rapid consumption of energy and oxygen, which was utilized to calculate the spare respiratory capacity of the cell, defined as the difference between maximal and basal respiration. Finally, the complex I inhibitor, rotenone (10μM), was added to establish the non-mitochondrial OCR. UTCs from young-mature and aged rat bladders reacted to all pharmacologic compounds, indicating fully functional mitochondria and generating a measurable bioenergetic profile. The OCR measured at baseline (P=0.001), the maximal respiratory rate (P =0.013), and the spare respiratory capacity (P=0.017) all proved to be significantly lower in aged animals than in young animals (Fig. 3B). These results were further supported by the significant decrease in hypotonic-evoked ATP release that was observed in aged UTCs compared to young-mature UTCs (P=0.006) (Fig. 3C).
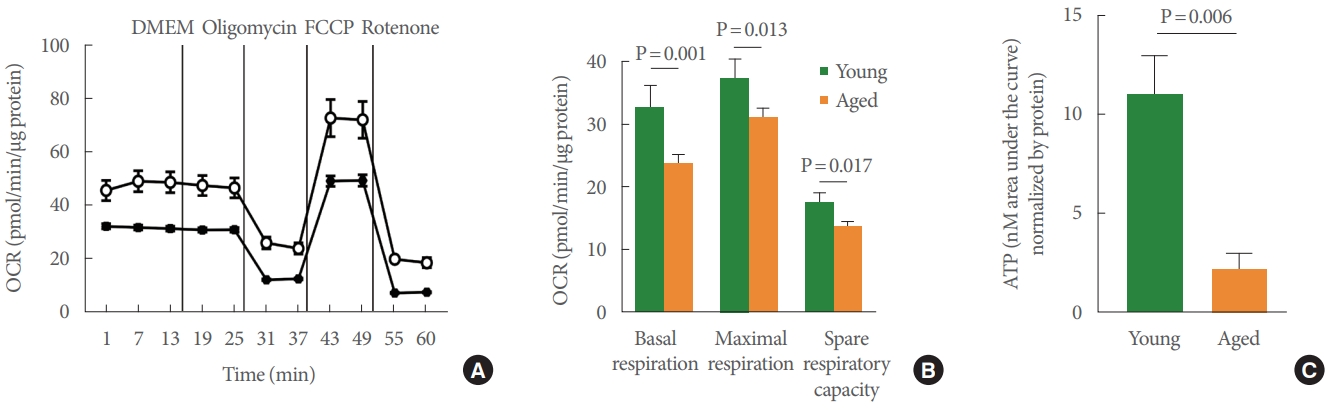
UTCs from aged rats showed altered mitochondria bioenergetics. (A) Representative Seahorse trace measuring oxygen consumption in UTCs from young (open circles) and aged (squares) rats. Trace shows basal respiration, the addition of DMEM as a vehicle control, oligomycin (2.5μM), FCCP (0.7μM), and rotenone (10μM) (as described in the Methods section). (B) Quantitation of data from 2 individual experiments (N=4 rats/group) measuring basal respiration (P=0.001), maximal respiratory rate (P=0.013), and spare respiratory capacity (P=0.017). (C) Hypotonicity-evoked adenosine triphosphate release was significantly lower in UTCs from aged versus young rat bladders by 80%; N=3 each, P=0.006. UTC, urothelial cell; OCR, oxygen consumption rate; DMEM, Dulbecco’s Modified Eagle Medium; FCCP, carbonyl cyanide-4-(trifluoromethoxy) phenylhydrazone.
DISCUSSION
In the present study, we examined how increased age in F344 rats alters urothelial health. Our findings revealed that aging negatively affected a number of biomarkers associated with cellular senescence, increased oxidative stress and mitochondrial bioenergetics. These types of detrimental changes over time could lead to defects in both the barrier and sensory aspects of urothelial functioning, culminating in abnormal bladder functioning and micturition behavior.
Mucosal samples from aged rats showed significantly higher levels of p21-Arc, nitrotyrosine, and cytochrome C than those obtained from younger rats. P21-Arc has been proposed to be a useful biomarker for cellular senescence in rodent urothelial tissue [21,22]. As such, elevated levels of p21-Arc indicate aging-related phenotypical cellular changes, such as decreased responsiveness to growth-promoting stimuli, chromatin reorganization, and the secretion of a number of proinflammatory cytokines. Nitrotyrosine is a product of tyrosine nitration, and its production is highly influenced by the accumulation of RNS. Thus, it is an indicator of oxidative stress, and it is often used as a marker of cell damage. As the terminal enzyme in the respiratory chain in mitochondria, cytochrome C is responsible for the storage of released energy and is therefore essential for the production of ATP. Outside the mitochondria, cytochrome C is one of the main regulators of caspase activated apoptosis. Accumulation of ROS and RNS in the mitochondrion induces the release of cytochrome C into the cytosol where it binds with apoptotic protease activating factor-1 (Apaf-1), activating caspase-9 and thereby initiating the apoptotic pathway. Elevated levels of cytochrome C indicate high oxidative stress in the mitochondria and are linked to a decrease in cell health and ATP production. The increase in both nitrotyrosine and cytochrome C in our study supports the view that the urothelium is significantly negatively impacted by chronic accumulation of ROS and RNS. Future studies are needed to assess the extent of apoptosis in aged animals, in part by investigating levels of caspase-9 or caspase-3 (caspase-9 is related to the initiation of the apoptotic pathway, whereas caspase-3 is involved in the execution phase of the apoptotic pathway). Previous research has reported significantly increased levels of caspase-3 in urothelial samples obtained from interstitial cystitis/ bladder pain syndrome patients [23]. Apoptosis was present in samples from these patients and was found to be mediated by the inflammatory pathway. The elevated levels of p21-Arc and cytochrome C reported here suggest that the secretion of proinflammatory cytokines might be elevated and apoptosis of UTCs might be occurring at an elevated rate. One could hypothesize that levels of caspase-9 and caspase-3 will be elevated in urothelial samples from aged animals. However, an analysis of caspase-3 levels in samples from studies conducted in aged mice reported no increase in apoptotic activity [17]. Thus, it is possible that cytochrome C levels increase in aged UTCs without the execution of apoptosis. This would lead to an accumulation of damaged and unhealthy cells in the urothelium, negatively impacting bladder functioning. A better understanding of the underlying mechanisms could improve bladder health and therapeutic options for the high prevalence of LUTS in older adults.
Our findings in aged bladder UTC cultures indicate a significant accumulation of basal ROS, confirming that, in agreement with our observations in mucosal tissue samples, UTC health is negatively impacted by aging. To assess the effects of oxidative stress on urothelial mitochondrial functioning we investigated the effects of age on the following: (1) Ψm using TMRM intensity measurements, (2) cellular respiration and mitochondrial bioenergetics using the Seahorse assay, and (3) ATP release using the luciferin-luciferase assay. Since TMRM is a cell-permeant dye that accumulates in active mitochondria with intact membrane potentials, it follows that in healthy cells with functioning mitochondria the signal intensity is bright. This is in contrast to aged UTCs, which showed a decreased staining intensity, indicating depolarization or loss of resting mitochondrial membrane potential (Fig. 2C) and suggesting decreased cellular viability.
Ψm drives a number of fundamental processes; thus, the observed age-associated Ψm decrease also implies a deterioration of cellular function, including cellular respiration and, thereby, ATP production. The bioenergetic profile of aged rats (studied using the Seahorse method) indicates that the OCR (monitored in real time) is significantly decreased, suggesting that these cells have decreased their activity. It has been reported that a decrease of Ψm activates the Pink1/parkin system [24]. This system is responsible for initiating signaling pathways related to the removal of damaged mitochondria. Work by our group has observed a significant aging-related increase in the expression of parkin in the bladder wall, indicating activation of this pathway in aged rats [25]. Decreases in Ψm are related to the induction of apoptotic processes [26]. Augmented expression of dynamin-related protein 1 in bladder tissue from aged rats [25] is an indicator of decreased Ψm and increased release of cytochrome C [27], which were both confirmed in the present study.
Based on the findings reported herein, we argue that the aging process has a substantial impact on urothelial health and that the mechanisms outlined may contribute to the deterioration of bladder morphology and bladder functioning, which may play a role in the high prevalence of LUTS in older adults [3-5]. A healthy urothelium is crucial for the maintenance of optimal barrier and signaling properties. We postulate that the observed decline of urothelial health and bioenergetic profile may alter the capacities of the urothelium to optimally execute both barrier and signaling functions. Previous work by our group has indicated that the administration of a purine nucleoside phosphorylase inhibitor, which can increase levels of “uro-protective” precursors (e.g., inosine and guanosine) that reduce oxidative stress while simultaneously decreasing levels of “uro-toxic” products (e.g., hypoxanthine) that are a source of ROS, significantly improves bladder functioning in aged rodents [25,28].
To conclude, in the present work, we showed that aging was associated with a significant increase in cellular senescence and oxidative stress in bladder mucosa samples, and that mitochondrial Ψm, mitochondrial OCR, and ATP release were significantly lower in UTC cultures from aged bladders than in those obtained from younger animals. We propose that these changes might significantly impact the barrier and signaling properties of the urothelium and potentially play an important role in the increased prevalence of LUTS in older adults. Future research should aim to further investigate the nature of the relationship between oxidative stress and bladder functioning, with the potential to identify novel therapeutic targets that might improve the quality of life and self-reliance of many older adults suffering from LUTS.
Notes
Funding Support
This project was funded by NIH/NIA R01 AG056944 grant awarded to Prof. Dr. Birder.
Research Ethics
The Institutional Animal Care and Use Committee (IACUC) approved all procedures reported in this study.
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION STATEMENT
·Conceptualization: SS, LB
·Data curation: MDR, AWJ, AK, ST
·Formal analysis: MDR, AWJ, AK, AK, LB
·Funding acquisition: LB
·Methodology: MDR, AWJ, AK, ST, AK, SS
·Project administration: LB
·Visualization: MDR, AWJ, AK, SS, LB
·Writing-original draft: MDR, LB
·Writing-review & editing: MDR, AWJ, AK, AK, LB
