 |
 |


- Search
| Int Neurourol J > Volume 21(4); 2017 > Article |
|
ABSTRACT
Purpose
To determine an objective cutoff value (COV) for urinary incontinence (UI) using the Expanded Prostate Cancer Composite (EPIC) score after radical prostatectomy (RP).
Methods
From 2004ŌĆō2013, all RP patients at our institution completed the EPIC urinary domain (EPIC-UD) questionnaire preoperatively and 6 weeks; 3, 6, 9, 12, and 18 months postoperatively; and yearly thereafter. The EPIC-UD is composed of several questions, 4 of which address UI qualitatively (EPIC-UI). Furthermore, patients were asked to complete a global quality of life (QoL) questionnaire regarding continence. The EPIC COV was calculated using receiver operating characteristic (ROC) analysis. Correlations between the EPIC-UI and quantitative QoL were evaluated using the Kendall-Tau test.
Results
We analyzed 239 patients with a median age of 63 years (interquartile range [IQR], 59ŌĆō66 years), a median follow-up of 48 months (IQR, 30ŌĆō78 months) and a median preoperative EPIC-UI score of 100 (IQR, 91.75ŌĆō100). The ROC analysis for the distinction between EPIC-UI and the use of Ōēż1 pad/day yielded an EPIC-UI COV of >85, which we termed the UI-85, with an area under the curve of 0.857 (P<0.0001). A stronger correlation was seen between QoL scores and the UI-85 (1 year postoperatively: correlation coefficient [CC], 0.592; P<0.0001) than between QoL and not using a pad (CC, 0.512; P<0.0001).
Conclusions
The calculated COV of the EPIC-UI for continence was 85. UI is a multidimensional condition that cannot be adequately characterized by a single piece of information, such as pad usage only. Hence, the UI-85 represents a nuanced and straightforward tool for monitoring and comparing continence between different time points and cohorts in a multidimensional and objective manner.
Persistent urinary incontinence (UI) after radical prostatectomy (RP) is a significant side effect of a widely performed operation. While UI has a strong and sustained influence on a patientŌĆÖs overall quality of life (QoL), it can have a complex, multidimensional clinical appearance. For example, many continent patients still rely on absorbent security pads, while others do not mind or even notice wet pads. At the other end of the spectrum, others can be devastated by minimal urinary leakage. Many in this latter group change pads multiple times per day though they are nearly dry. Furthermore, there are various types and sizes of pads. With such inconsistencies in mind, it is difficult to report, let alone assesses or compares, UI using existing methods.
In an attempt to make such a comparison, questionnaires have been developed to multidimensionally assess UI and to more accurately report and measure the true value of urinary continence [1]. More exhaustive questionnaires, such as the University of California, Los Angeles Prostate Cancer Index (UCLA-PCI) or the Functional Assessment of Cancer Therapy ŌĆō Prostate, are promising, but they are subjective and lack exact quantitative cutoff values (COVs) [2].
One widely used questionnaire is known as the Expanded Prostate Cancer Composite (EPIC) [3], an expansion of the original UCLA-PCI [2] that was developed to assess the presence of bothersome symptomatology after prostate cancer treatment. A section of this scoring system is solely dedicated to the assessment of urinary problems and is composed of 12 questions that address continence, as well as hematuria, dysuria, and other voiding problems. In addition to requiring objective answers (such as the number of pads used daily), the EPIC also allows for subjective answers, such as ŌĆ£occasionalŌĆØ and ŌĆ£frequent.ŌĆØ In developing an objective method to better assess UI post-RP, it is also important to recognize that many existing assessments such as the EPIC also lack a clear, uniform definition that differentiates continence from incontinence. This, in turn, can also restrict the objectivity of these assessments.
Again, having uniform definitions and criteria would greatly facilitate the comparison of various patient populations and experimental trials. As quantitative assessments typically facilitate objectivity, we sought to establish a standardized definition of continence based on a quantitative COV. Between 2004 and 2013, our institution prospectively collected functional and oncological data (including the EPIC score) from patients undergoing RP. Therefore, the aim of our investigation was to define a COV for the EPIC score by assessing the functional data of our long-term RP cohort.
The EPIC score is a self-assessment questionnaire that is widely used to evaluate a multitude of urological problems. Between 2004 and 2013, all patients undergoing open retropubic RP at our institution were asked to complete the EPIC urinary domain (EPIC-UD), a subset of questions within the EPIC that focuses exclusively on urinary symptomatology in the context of prostate cancer treatment. Questions were answered in a semiquantitative manner, with scores ranging from 0 to 100 points and higher scores signifying a better quality of urination [3]. Patients were also asked to globally quantify their QoL with regards to continence using a 7-point Likert scale (0, excellent; 6, very bad). These questionnaires were completed preoperatively as well as at the following postoperative time points: 6 weeks; 3, 6, 9, 12, and 18 months; and yearly thereafter.
All patients who underwent RP, with or without a nervesparing approach, were included in our analysis.
Our analysis focused on the EPIC-UI, which consists of the following 4 questions extracted from the EPIC-UD that are specific for UI [3]. Of note, when the EPIC was first used, a factor analysis consistent with this grouping was performed:
- Question 1: Over the past 4 weeks, how often have you leaked urine?
- Question 4: Which of the following best describes your urinary control over the last 4 weeks?
- Question 5: How many pads or adult diapers per day did you usually use to control leakage during the last 4 weeks?
- Question 6a: How big of a problem has (dripping or leaking urine) been for you during the last 4 weeks?
To measure significant changes in continence over time, the Wilcoxon signed-rank test and the McNemar test for dependent values were used. Receiver operating characteristic (ROC) curves were used to generate a COV from the EPIC-UI results, with the goal of identifying maximal specificity and sensitivity for not using a pad.
The Kendall-Tau test (with a 2-sided P-value) was applied to the cohort functional data to verify the applicability of the calculated total EPIC-UI COV. Here, we compared the correlation between QoL and both definitions of continence at all-time points. Bootstrapping was used to calculate the 95% CI.
By applying the Kaplan-Meier method for regaining continence, the cumulative proportion of continent men and the median time to continence (including the 95% confidence interval [CI]) were calculated for both definitions of continence.
Microsoft Access 2010 (Microsoft Corp., Redmond, WA, USA) was used for data acquisition, while IBM SPSS Statistics ver. 23.0 (IBM Co., Armonk, NY, USA) was utilized for calculations.
A total of 239 patients with a median age of 63 years old (interquartile range [IQR], 59ŌĆō66 years) participated in the study (Table 1). The median follow-up time was 48 months (IQR, 30ŌĆō78 months). The median preoperative EPIC-UI score was 100 points (IQR, 91.75ŌĆō100 points). One year postoperatively, the EPIC-UI score decreased to 85.5 points (IQR, 58.5ŌĆō100 points).
An overview of continence development in our cohort (from the preoperative period to 10 years postoperatively) can be obtained by looking at the mean EPIC-UI (Fig. 1). A significant increase in the mean EPIC-UI was observed postoperatively between 6 weeks and 12 months. After 12 months, this mean score remained stable and did not significantly increase or decrease. This, in conjunction with the fact that a sizable number of subjects remained in the study at 1 year (n=177), led us to use the 1-year time-point as the cutoff time-point for our COV calculations.
As stated previously, the EPIC-UI is composed of 4 questions regarding urine leakage (item 1), control (item 4), number of pads per day (item 5), and urine loss (item 6a). With no use of a pad utilized as a hard criterion for satisfactory continence, scores for the 3 remaining questions (items 1, 4, and 6a) were summed and used to develop the COV. A significant correlation was seen between overall pad usage and the sum of the 3 remaining EPIC-UI questions, with a correlation coefficient of 0.528 (95% CI, 0.439ŌĆō0.609; P<0.0001). A ROC distribution comparing pad usage (0 vs. 1+ per day) against the sum of the 3 remaining EPIC-UI questions yielded a COV for maximum sensitivity (0.767) and specificity (0.811) of 240, with an area under the curve of 0.857 (95% CI, 0.8ŌĆō0.914; P<0.0001) (Fig. 2).
To complete our calculations with EPIC-UI, the value for not using a pad (=100) was added to our previous COV of 240, yielding a new value of 340. Thus, the COV was defined as an average score of 85 (=340/4) (UI-85). Scores greater than 85 predicted no usage of pads.
Based on the assumption that QoL is the major concern of patients suffering from UI, we then decided to compare quantitative QoL scores against the UI-85 as well as against the old standard of no pad use. This comparison was designed to assess the accuracy of competing definitions of continence (UI-85 vs. pad/no-pad). Starting at 9 months postoperatively, the data revealed a stronger correlation between QoL and the UI-85 than between QoL and the binary definition of continence (Fig. 3).
The percentage of continent men defined according to the UI-85 at each time point was also calculated and graphically presented (Fig. 4). A significant increase in the number of continent patients took place between 6 weeks and 6 months postoperatively (UI-85; P<0.0001), after which no significant changes occurred. For patients with an EPIC-UI score of 100 (signifying no issues with urinary continence), the data demonstrate a significant (P<0.0001) increase in continence up to 3 years postoperatively, followed by a plateau thereafter. In the same manner, QoL answers regarding continence can be shown over time (Fig. 4). For instance, the percentages of patients with EPIC-UI scores >85 and =100 compared against QoL continence answers (such as ŌĆ£satisfiedŌĆØ or ŌĆ£excellentŌĆØ) are similar (Fig. 4).
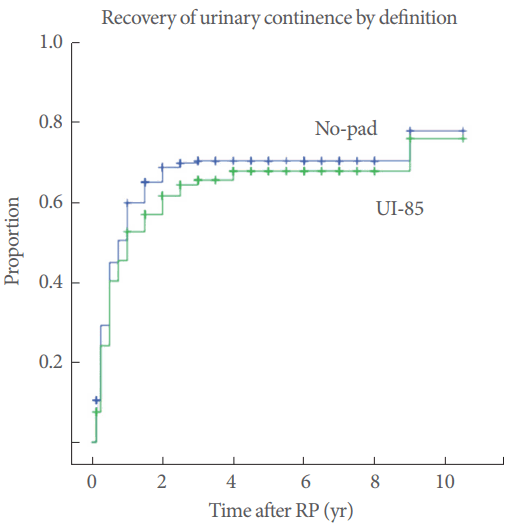
One year postoperatively, the Kaplan-Meier curve (Fig. 5) demonstrated a cumulative continence rate of 52.5% (95% CI, 46%ŌĆō59%) using the UI-85 and 59.7% (95% CI, 53.4%ŌĆō66%) using the binary continence definition. By the same analyses, the median time to regaining continence was 12 months (IQR, 7.4ŌĆō16.6 months) using the UI-85 and 9 months (IQR, 6.4ŌĆō11.6 months) when UI was defined as not using a pad.
Several methods have been developed for determining and documenting UI, and they range from simple to highly complex. However, no existing method accurately characterizes the multidimensional nature of UI. We surmised that an ideal method would be (1) objective, (2) easy to understand for the patient, (3) easy to collect as a healthcare provider, and (4) easy to interpret accurately. We consider these criteria to be equally important and, to our knowledge, such an ideal test does not yet exist. Therefore, our aim with this paper was to demonstrate our attempt at developing a quantitative COV based on an existing exam: the EPIC-UD.
Thus far, the gold standard for assessing UI has been simply to ask a patient regarding his or her use of absorbent pads for urine leakage. The use of such a pad after RP would imply some degree of persistent UI. While this method fulfills 3 of the 4 criteria listed above, it disregards the fact that this assessment could be strictly or liberally interpreted. For instance, many physicians consider a patient to be continent even if that patient occasionally relies on a security pad [4]. Additionally, the need for a pad is not well-defined, which negatively affects the accuracy of answers to this question. For example, an incontinent patient who loses urine weekly and refuses to wear pads could hardly be accurately assessed using this criterion. Conversely, a careful man who hardly ever loses urine, but changes pads multiple times daily to maintain his hygiene, may be misinterpreted to be reliant on pads when he is actually not. Therefore, there is a need for standardized criteria that accurately classify incontinence as well as its management.
To instill a degree of objectivity, some authors have weighed patientsŌĆÖ pads before and after having patients perform several standardized exercises [5,6]. However, such an approach is likely to be too cumbersome for daily clinical practice. This is further exacerbated by the lack of a routine assessment at all follow-up visit. Despite such drawbacks, such questionnaires have the potential to provide accurate clinical data even if they are not the easiest or most practical to implement. In this study, we present an EPIC score-based COV for urinary continence. These 4 questions solely addressing urinary continence were utilized to generate to a sub-score known as the EPIC-UI.
In this paper, we sought to determine an objective COV based on the EPIC scoring system. Our calculations identified a COV of 85 (UI-85) for continence. To assess the clinical relevance of this COV, we compared it against the binary definition of continence (i.e., pad or no-pad) as well as to QoL. In these calculations, the UI-85 was strongly correlated with QoL; this correlation was stronger than that between QoL and the binary definition of continence.
Furthermore, we applied the UI-85 to our cohort of RP patients. While the cumulative continence rates were similar for both the UI-85 and the binary definition (52.5% and 59.7%, respectively), the median time to regaining continence was longer using the UI-85 (12 months vs. 9 months) for the same cohort. Hence, it appears that the UI-85 is a more sensitive and nuanced tool for describing postoperative continence rehabilitation, as it could detect slight increases in continence between months 9 and 12, whereas the binary definition plateaued earlier.
We also analyzed and compared the functional outcomes of our cohort. The percentage of men with EPIC-UI scores >85 at different time points demonstrated a high degree of similarity to the percentage of men who reported that they were ŌĆ£satisfiedŌĆØ with their QoL. This further corroborates the clinical relevance of the UI-85. We recognize that there are many different ways of measuring continence. For instance, weighing pads after predefined exercises is a relatively simple way of doing so. However, it is resource-intensive for both patients and their physicians. In contrast, the EPIC-UI can easily be extracted from the widely used EPIC score, making it an uncomplicated tool for monitoring continence over time or comparing the urinary continence between different cohorts.
Advances in operative techniques have done little to alleviate the doubt surrounding the impact of these approaches and technologies on UI [7]. Some trials have demonstrated better recovery of early incontinence (3ŌĆō6 months postoperatively) following robotic RP, whereas other trials have suggested the opposite [8-10]. While the achievement and quality of early continence are important to understand, the majority of RP patients typically have a long life expectancy, making the analysis of long-term continence (more than 6 months postoperatively) outcomes dramatically more important and more clinically relevant.
While the results in our cohort only differed by 1.9% at the 10-year time-point, our Kaplan-Meier curves revealed a faster rate of increase of continence using the no-pad definition than when the UI-85 was used. At 2 years postoperatively, the plateau for the no-pad definition was reached, whereas continence levels per UI-85 plateaued after 3 years postoperatively. Thus, the EPIC-UI COV of 85 (i.e., the UI-85) appears to represent a more accurate and nuanced tool for monitoring postoperative continence.
When applying the UI-85, it is important to recognize that some men will eventually demonstrate slight UI. Hence, the UI-85 is not completely equivalent to no-pad. Nonetheless, our calculations showed that patients with an EPIC-UI > 85 were more likely to be satisfied with their degree of urinary continence. Over a several-year timeframe, it is possible that patients may accept a slight degree of incontinence, potentially leading to a bias (for e.g., reporting better outcomes despite unchanged continence or incontinence). We attempted to account for this problem by basing our calculations on data no older than the 1-year postoperative period, as practically, RP patients are less likely to enter the mental state of resignation at 1 year than at a later follow-up time.
Another consideration in the broader milieu of urinary continence is that the impact of nerve-sparing prostatectomy on urinary continence is still unclear and remains a matter of ongoing discussion [11-17]. In our cohort, only 39 patients who completed the EPIC questionnaire at 1 year postoperatively did not undergo nerve-sparing surgery. Furthermore, these patients were statistically significantly older and had a higher tumor burden, both of which could have contributed to a substantial amount of bias if the ROC calculations for COVs had been performed for nerve-sparing and nonŌĆōnerve-sparing groups separately. Hence, to generate a universally applicable definition, patients with and without nerve-sparing RP were collectively included in the study.
In conclusion, the COV of the EPIC-UI for continence was found to be 85 (the UI-85). The UI-85 represents a nuanced and straightforward tool to monitor and compare urinary continence between different time points and cohorts in a multidimensional and objective manner.
NOTES
REFERENCES
1. Abrams P, Avery K, Gardener N, Donovan J; ICIQ Advisory Board. The International Consultation on Incontinence Modular Questionnaire: www.iciq.net. J Urol 2006;175(3 Pt 1):1063-6. PMID: 16469618


2. Litwin MS, Hays RD, Fink A, Ganz PA, Leake B, Brook RH. The UCLA Prostate Cancer Index: development, reliability, and validity of a health-related quality of life measure. Med Care 1998;36:1002-12. PMID: 9674618


3. Wei JT, Dunn RL, Litwin MS, Sandler HM, Sanda MG. Development and validation of the expanded prostate cancer index composite (EPIC) for comprehensive assessment of health-related quality of life in men with prostate cancer. Urology 2000;56:899-905. PMID: 11113727


4. Sandhu JS. Urinary dysfunction in prostate cancer: a mangement guide. London: Springer International Publishing; 2016.
5. Tsui JF, Shah MB, Weinberger JM, Ghanaat M, Weiss JP, Purohit RS, et al. Pad count is a poor measure of the severity of urinary incontinence. J Urol 2013;190:1787-90. PMID: 23727311


6. Nitti VW, Mourtzinos A, Brucker BM; SUFU Pad Test Study Group. Correlation of patient perception of pad use with objective degree of incontinence measured by pad test in men with postprostatectomy incontinence: the SUFU Pad Test Study. J Urol 2014;192:836-42. PMID: 24650425


7. Yaxley JW, Coughlin GD, Chambers SK, Occhipinti S, Samaratunga H, Zajdlewicz L, et al. Robot-assisted laparoscopic prostatectomy versus open radical retropubic prostatectomy: early outcomes from a randomised controlled phase 3 study. Lancet 2016;388:1057-66. PMID: 27474375


8. Herlemann A, Cowan JE, Carroll PR, Cooperberg MR. Community-based outcomes of open versus robot-assisted radical prostatectomy. Eur Urol 2017 May 9;[Epub]. pii: S0302-2838(17)30335-4. https://doi.org/10.1016/j.eururo.2017.04.027. 
9. Ong WL, Evans SM, Spelman T, Kearns PA, Murphy DG, Millar JL. Comparison of oncological and health-related quality of life outcomes between open and robot-assisted radical prostatectomy for localised prostate cancer - findings from the population-based Victorian Prostate Cancer Registry. BJU Int 2016;118:563-9. PMID: 26573954


10. Wallerstedt A, Tyritzis SI, Thorsteinsdottir T, Carlsson S, Stranne J, Gustafsson O, et al. Short-term results after robot-assisted laparoscopic radical prostatectomy compared to open radical prostatectomy. Eur Urol 2015;67:660-70. PMID: 25308968


11. Burkhard FC, Kessler TM, Fleischmann A, Thalmann GN, Schumacher M, Studer UE. Nerve sparing open radical retropubic prostatectomy--does it have an impact on urinary continence? J Urol 2006;176:189-95. PMID: 16753399


12. Nandipati KC, Raina R, Agarwal A, Zippe CD. Nerve-sparing surgery significantly affects long-term continence after radical prostatectomy. Urology 2007;70:1127-30. PMID: 18158032


13. Reeves F, Preece P, Kapoor J, Everaerts W, Murphy DG, Corcoran NM, et al. Preservation of the neurovascular bundles is associated with improved time to continence after radical prostatectomy but not long-term continence rates: results of a systematic review and meta-analysis. Eur Urol 2015;68:692-704. PMID: 25454614


14. Park YH, Kwon OS, Hong SH, Kim SW, Hwang TK, Lee JY. Effect of nerve-sparing radical prostatectomy on urinary continence in patients with preoperative erectile dysfunction. Int Neurourol J 2016;20:69-74. PMID: 27032560




15. Kundu SD, Roehl KA, Eggener SE, Antenor JA, Han M, Catalona WJ. Potency, continence and complications in 3,477 consecutive radical retropubic prostatectomies. J Urol 2004;172(6 Pt 1):2227-31. PMID: 15538237


16. Tzou DT, Dalkin BL, Christopher BA, Cui H. The failure of a nerve sparing template to improve urinary continence after radical prostatectomy: attention to study design. Urol Oncol 2009;27:358-62. PMID: 18439849


Fig.┬Ā1.
Mean EPIC-UI scores (95% confidence interval) over a time period of 10 years. The table shows the mean EPIC-UI scores with standard deviations (SDs) and significant P-values according to the Wilcoxon signed-rank test for dependent values. EPIC-UI, Expanded Prostate Cancer Composite urinary incontinence. RP, radical prostatectomy. Arrow indicates a significant increase.
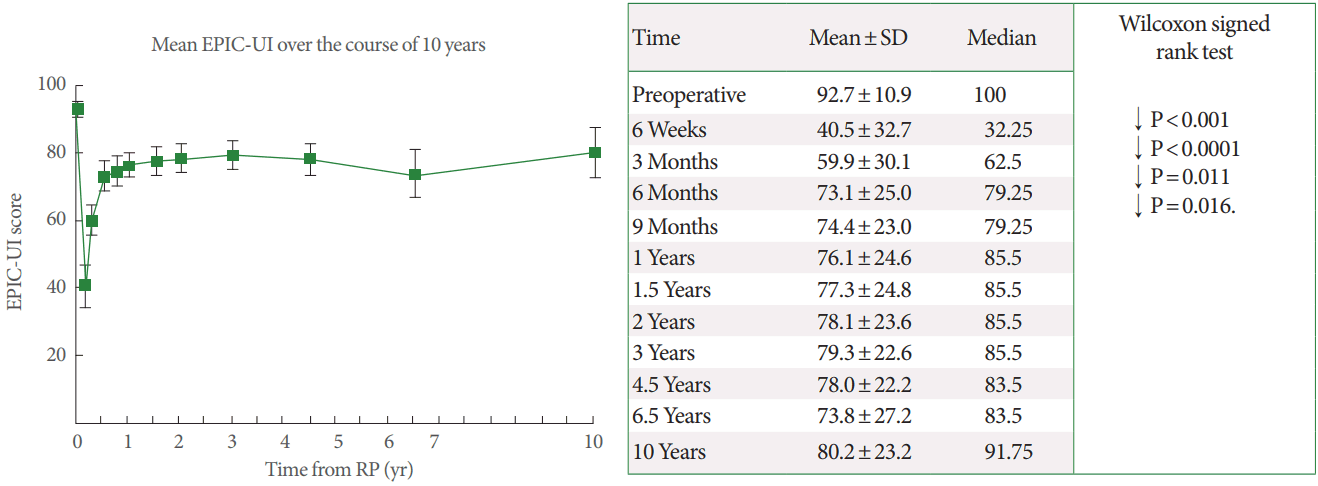
Fig.┬Ā2.
ROC curve for pad usage with remaining EPIC-UI questions, including visualization of the COV at maximum sensitivity and specificity. ROC, receiver operating characteristic; EPIC-UI, Expanded Prostate Cancer Composite urinary incontinence; COV, cutoff value; AUC, area under the curve.
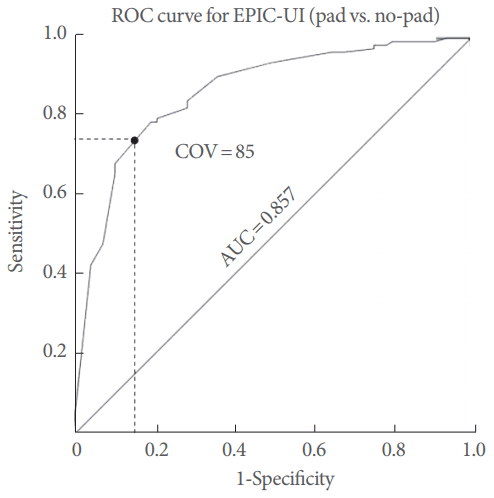
Fig.┬Ā3.
Graphical overview of the correlation coefficient (CC) and 95% confidence intervals measured by Kendal-Tau test and bootstrapping over time (blue: the no-pad definition of urinary incontinence; red: the UI-85 definition of urinary incontinence). QoL, quality of life; UI, urinary incontinence; RP, radical prostatectomy.
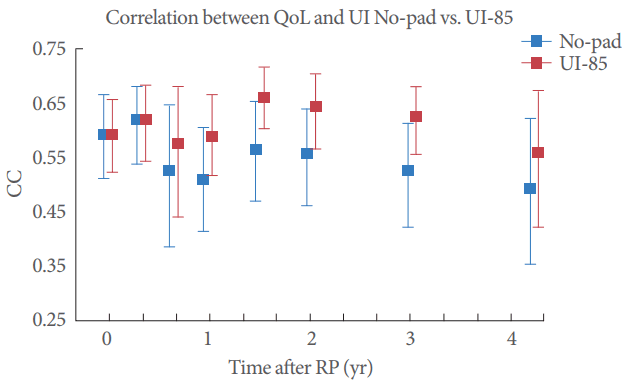
Fig.┬Ā4.
QoL (continence) and proportion of continent patients 0 2 4 6 8 10 over time. Blue (EPIC): percentage of continent men by UI-85 (light blue) and patients without problems concerning incontinence (dark blue; EPIC-UI =100) over a period of 10 years. Green (QoL): percentage of patients who were satisfied (light green) and very satisfied (dark green). EPIC-UI, Expanded Prostate Cancer Composite urinary incontinence; QoL, quality of life.
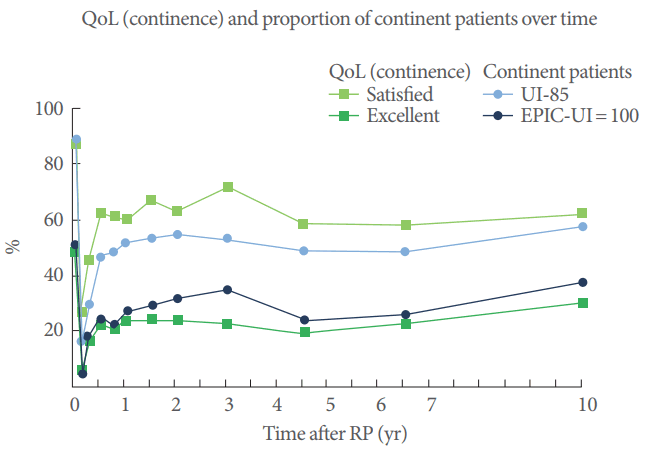
Fig.┬Ā5.
Kaplan-Meier curves for returning to continence using both definitions (UI-85 and no-pad) to show the development over time for the whole cohort. UI, urinary incontinence; RP, radical prostatectomy.
Table┬Ā1.
Baseline data of cohort (n=239)





