INTRODUCTION
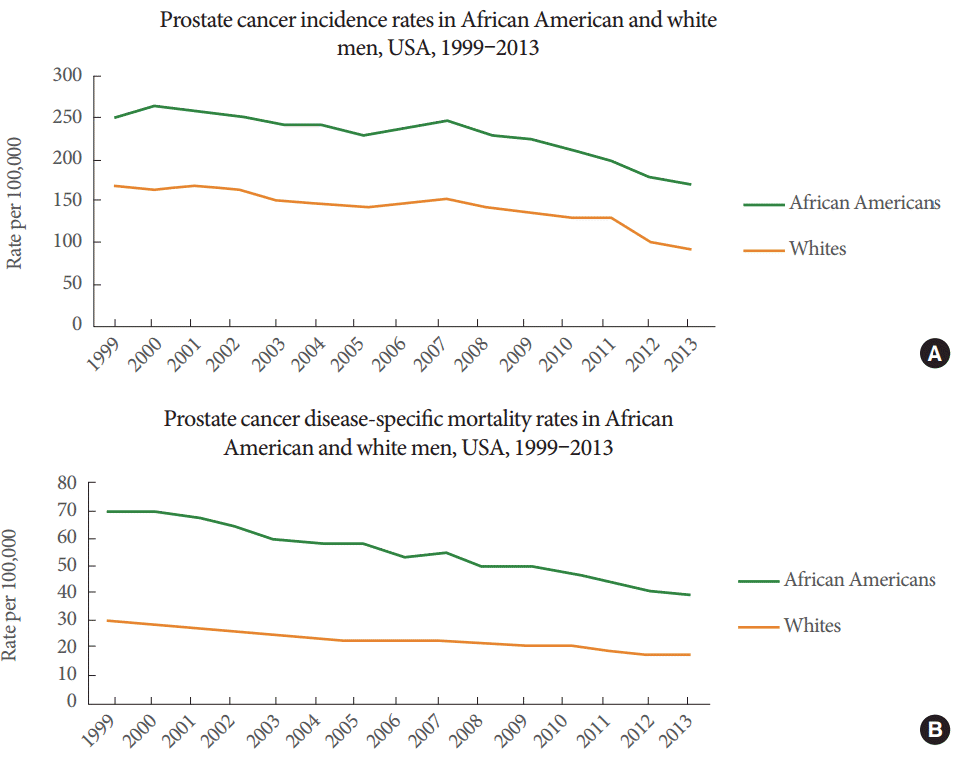
Prostate cancer (PCa), the most common malignancy in adult men, is the second most common cancer and the fifth-highest cause of cancer mortality in the United States (US). The American Cancer Society estimates that 180,890 new cases and 26,120 deaths due to PCa will occur in 2016. The incidence rates between 2008 and 2012 were 131.5 and 21.4 deaths per 100,000 persons, respectively [1]. The recent decrease in the incidence of PCa was driven, in part, by the release of the US Preventive Services Task Force draft recommendation against prostate-specific antigen (PSA) screening in 2011 [2]. The incidence rates in different ethnic groups in the US are significantly different: 123.0 and 208.7 per 100,000 persons in non-Hispanic whites and African Americans, respectively. The mortality rates were 19.1 per 100,000 persons in non-Hispanic whites and 44.2 per 100,000 persons in African Americans [1]. Thus, African Americans suffer a disproportionate burden from PCa, having the highest incidence and mortality rates of any racial or ethnic group (Fig. 1). The causes of these disparities are complicated and they may occur in part due to environmental factors, such as socioeconomic status, and biological differences, such as genetic susceptibility. Currently, the only well-established risk factors for PCa are age, race, and family history of the disease. Men who have a first-degree relative with a history of PCa are 2 to 3 times more likely to develop PCa [3].
The National Institutes of Health Surveillance Epidemiology and End Results (SEER) data between 1975 and 2012 showed that 309,289 men were diagnosed with PCa during this period, and that fatal PCa accounted for 17% of the cases. During the 10-year period following diagnosis, PCa was the most common cause of death (34%). In addition, African American men had an earlier mean age at diagnosis (69.1 years vs. 71.1 years) and shorter mean survival period (44.4 months vs. 48.4 months). The ratio of the mortality rates of PCa in African American and white men has not changed over the past few decades, with African American men showing a consistent 2.2‚Äď3.0 fold higher risk. In addition, this ratio was found to be 4.2 fold among young adults (45‚Äď49 years old) and 2.0 fold higher among older African American men (>60 years old) [4]. The lifetime probabilities of diagnosis or disease-specific death from PCa in US between 2010 and 2012 were 18.2% and 4.4% in African Americans, respectively, and 13.3% and 2.4% in non-Hispanic whites, respectively [3].
The Health and Retirement Study measured changes in PCa screening prevalence in US men aged over 50 years between 1996 and 2008, including 1,359 African Americans and 8,226 whites. The results showed an improvement from 62.0% to 71.2% in African Americans and from 68.6% to 71.3% in whites. These data indicated a significant time and race interaction and an increased prevalence of prostate screening in African American men [5].
In general, socioeconomic status is inversely correlated with incidence and mortality rates. African American men have lower levels of income, and only 15% of African American men have a bachelor’s degree, in contrast to 30.7% of white men. A higher proportion of African American men than white men reported medical mistrust [6], which may explain why African American men receive screening tests less frequently.
DIAGNOSIS
The disparities between African Americans and whites in PCa diagnosis have been well established in the US [7]. However, the contribution of PSA screening tests to the incidence rate is more controversial. The results of a US-based randomized trial indicated no reduction in PCa mortality as a result of PSA screening tests, while several European trials showed a modest benefit [3]. However, other important variables should be considered for early detection or screening.
The Selenium and Vitamin E Cancer Prevention Trial investigated whether supplementation with selenium, vitamin E, or both would reduce PCa incidence. This trial was performed between 2001 and 2004, and included 4,674 African Americans and 27,566 non-Hispanic white men (>55 years old) with normal PSA levels (<4.0 ng/mL) and normal results of a digital rectal examination. Over 5.6 years of follow-up, 1,723 men were diagnosed with PCa. The researchers observed that African Americans were diagnosed younger (by a mean of 4.2 years), were less likely to have a college degree (33.2% vs. 55.4% completed college), and had a higher prevalence of diabetes, smoking, and obesity (38.9% vs. 30.1%) than white men. The incidence of PCa was significantly higher among African Americans in the trial [8]. Goovaerts et al. [8] observed that household median income and advanced-stage diagnosis were inversely correlated and that advanced-stage diagnosis was more common in African American (21%‚Äď24%) than in white men (17%‚Äď18%) based on the Florida Cancer Data System (n = 256,365).
In the SEER data, socioeconomic status was positively correlated with 5-year cause-specific survival in both racial groups. DeSantis et al. [3] similarly found that the risk of death from PCa in African Americans was consistently higher than in whites in all socioeconomic status strata. Among patients with the same high socioeconomic status, cancer screening was more common in whites than African Americans, and cancer detection was earlier in whites. Furthermore, nonaggressive tumors were found more often in whites than African Americans [9].
Trantham et al. [10] reported that nearly 25% of African Americans did not have a PSA test for at least 12 months in the first 5 years after initial PCa. Non-Hispanic white men were more likely to receive at least one annual test during the first 5 years after PCa treatment than men of other ethnic groups based on SEER data. African Americans men waited longer between diagnosis and treatment than white men after adjusting for age, annual household income, level of education, and health insurance status [6]. In addition, among 62,644 high-risk and nonmetastatic cases (PSA >20 ng/mL, Gleason score of 8‚Äď10, or stage‚Č•cT3a) in the SEER data, the African American patients were more likely to exhibit elevated PSA levels (17.6% vs. 9.9%, P<0.001) [11].
Moses et al. [12] observed that African Americans had higher percentages of T1c disease and higher PSA levels than white men (6.1 ng/mL vs. 4.7 ng/mL, P <0.005) among surgically treated young PCa patients (<50 years old) at Memorial Sloan-Kettering Cancer Center. However, significant differences were not found in clinical or pathological characteristics between the 2 racial groups in the Gleason score, extracapsular extension, lymph node involvement, or seminal vesicle invasion. However, they found a significant difference in survival between African Americans and whites in locally confined cases, but not in patients with advanced disease. African American patients with locally advanced disease were at a greater risk of early biochemical recurrence than white patients.
TREATMENT
Based on to the current Clinical Practice Guidelines in Oncology of the National Comprehensive Cancer Network, the available options for PCa treatment are radical prostatectomy, external beam radiation therapy, brachytherapy, cryotherapy, androgen deprivation therapy, and combination therapy [13]. Many studies have been performed on disparities in health services and treatments offered to members of different ethnic and racial groups, especially in the US.
Powell et al. [14] performed survival analyses for both races in 2 distinct periods, the pre-PSA era (1973‚Äď1994) and the PSA era (1998‚Äď2005), using the SEER data [14]. They found that African American patients had a significantly lower survival rate than whites in both eras among patients who received surgical or radiation therapy. Furthermore, the proportion of African American patients with metastasis was significantly higher than the proportion of white patients with metastasis, and the mortality rate of African American patients was 2‚Äď3 times higher than that of white patients in both eras. Mahal et al. [15] reported that elderly African American patients (>70 years of age) with high-risk localized PCa (PSA>20 ng/mL, Gleason score of 8‚Äď10, or stage‚Č•cT3a) received less definitive treatment, such as radical prostatectomy, radiation therapy, or combination therapy, than white men (52.0% vs. 64.8%, P<0.001). Among patients with high-risk localized PCa, African Americans had higher preoperative PSA levels (median, 8.0 ng/mL vs. 6.3 ng/mL; P=0.001), while white men were more likely to have T3‚ÄďT4 stage disease (26.1% vs. 21.3%, P =0.001). In both racial groups, insured patients were more likely to undergo definitive therapy than uninsured patients, although the difference was more pronounced in African American men. Therefore, medical insurance may reduce the health disparity between whites and African Americans regarding treatment [15].
Moses et al. [16], using the SEER database with 327,641 men diagnosed with clinically located PCa between 2004 and 2011, showed a significant difference in the treatment type received according to racial group. While 31.1% of African American men were treated with radical prostatectomy, 38.2% of white men received this form of treatment. It was also found that 26.8% of African Americans received external beam radiation therapy, compared to 22.7% of white men. However, no significant difference in cryotherapy was found. This study also showed that the African Americans were more likely to experience treatment delays and postoperative complications and less likely to undergo lymph node dissection than white men. An independent study observed a similar treatment pattern. Data between 2007 and 2008 from the Diagnosis and Decisions in Prostate Cancer Treatment Outcomes trial showed that prostatectomy was the main treatment in 79.5% of whites and 69.4% of African Americans, and that African Americans were more likely to receive radiation therapy than white men (22.6% vs. 16.1%) [6].
In a study conducted at the Durham Veterans Affairs Medical Center, African American patients had a lower clinical stage than white patients (P=0.001), and were less likely to receive preradiation androgen deprivation therapy (P=0.013) [17]. Allott et al. [17] found a significant association between visceral fat area and aggressive PCa in African Americans (P=0.035), suggesting a potential association between obesity and the risk of aggressive PCa. However, this association was not found in white patients. This association between obesity and PCa was confirmed by us [18].
Wang et al. [19] reported that the treatments received between 2004 and 2011 varied significantly according to race. While a radical prostatectomy was performed in 39.8% of white men, this occurred in only 27.5% of African Americans. Moreover, this disparity persisted over time. Between 2004 and 2005, 27.7% of white patients underwent radical prostatectomy, in contrast to 19.9% of African American patients (P<0.001), and in the period between 2010 and 2011, this disparity was even greater (44.6% vs. 34.1%, P <0.001). Although patients had similar clinical characteristics, the treatment plan was different in different groups. A greater percentage of African Americans (37.2%) received treatment with external beam radiation therapy, in contrast to 33.1% of white men (P<0.001). Androgen deprivation therapy was received by 9.5% of African Americans and 5.7% of white men (P<0.001). Additionally, 12.5% of African Americans were in watchful waiting, compared to only 7.2% of whites (P<0.001) [19].
GENETIC SUSCEPTIBILITY
As described above, African American men suffer from higher rates of aggressive PCa, including lethal cancers, than whites in the US [20]. However, the determinants of this high rate of incidence and aggressiveness of PCa in African Americans remain unresolved. This pattern may be attributed to socioeconomic status, detection at a late stage, biological aggressiveness [21], and differences in genetic susceptibility [18,22-28]. Selected characteristics and results of PCa genetic studies of African American populations are presented in Table 1.
Recently, a large analysis of the genetic contribution to PCa risk was performed at the Prostate Cancer Association Group to Investigate Cancer Associated Alterations in the Genome consortium, with DNA samples of over 40,000 Caucasians and 2,000 individuals of African descent from the US and Europe, identifying over 100 risk loci [29,30]. These genetic biomarkers were identified based upon analyses of a mostly Caucasian population. Few PCa genome-wide association studies have been performed on African American populations [31]. We and others have attempted to validate the risk loci identified in association studies or among whites [24,32-34]. However, as expected, most loci identified in whites were not confirmed in African-ancestry population association studies [18,22-27,31-37]. However, some loci were confirmed, and some may explain the significant health disparities in PCa (Table 1). For example, we investigated the association between genetic variations and risk in both racial groups, and further explored whether these associations varied according to obesity. We found that two single-nucleotide polymorphisms (SNPs) in HNF1B were associated with PCa risk only in African American men. As expected, obesity was a significant risk factor in African American men but not whites. Interestingly, HNF1B rs7501939 was associated with PCa risk exclusively in obese African American men (odds ratio, 2.14; P=0.01) [18].
Chang et al. [24] tested risk loci identified in white populations using 4,040 cases and 3,748 controls of African ancestry from 19 institutions in the US and United Kingdom under the Men of African Descent and Carcinoma of the Prostate consortium. Among 47 SNPs, including 21 from the 8q24 region, they validated 13, including 7 from the 8q24 region. Six SNPs were at JAZF1 (rs10486567), MSMB (rs10993994), 11q13 (rs12418451 and rs7931342), and NUDT10/11 (rs5945572 and rs5945619). The direction of association and odds ratios of the validated SNPs were similar to those observed in whites.
Recently we conducted a large genetic association study of an African American population with 9,531 subjects (4,853 cases and 4,678 controls) to validate previously identified risk loci for PCa and to assess their implications for the etiology of PCa. Among the 82 SNPs tested, 30 (37%) were validated to have a significant association with PCa risk. We concluded that the presence of the same directionality of influence and similar effects in the African American population indicated that loci have the same biological functions across populations, although most risk loci were not confirmed [27]. Therefore, larger association studies of African American populations are needed to identify African American-specific risk loci for PCa.
CONCLUSION
Although this review has shown inequalities in the diagnosis and treatment of PCa among different racial, economic, and social groups, further research must be conducted to better understand the causes of these disparities. The therapeutic options recommended by the physician and the final decisions of patients undergoing definitive treatment should also be evaluated. Additionally, the quality of the healthcare environment, the resources available to patients and health service providers, as well as the knowledge and skills of the professionals involved play a role in these disparities.
Patients’ understanding of the disease, acceptance of treatment, and possible complications may also differ substantially among different racial, cultural, and socioeconomic groups, leading patients to make different choices regarding treatment. A more even distribution of resources among health centers can help align the decisions made by providers about treatment, as well as the behavioral differences of patients regarding their decisions.
As discussed in this review, African Americans are more likely to be diagnosed with advanced stages of cancer when treatment options are more limited and often less effective. This may shape some of the disparities that we presented. Higher rates of comorbidities, such as obesity, diabetes and hypertension, among African American patients may also affect the treatment and survival rates in this population.
Improving the geographical distribution of federally qualified health centers that serve a large proportion of poor patients may be an effective mechanism for the prevention and control of PCa. These federally qualified health centers serve approximately 20 million patients annually in the US, and nearly two-thirds of patients seen are ethnic minorities, low-income, and uninsured [38], indicating that these facilities have the potential to reduce health disparities in ethnic minorities and other underserved populations.
It is not known whether increased access to tertiary medical centers will provide an opportunity to reduce the disparities in the treatment of clinically aggressive PCa, especially considering the need for multimodal treatment and greater access to technologically advanced treatment. Although no evidence indicates that different responses to therapy to PCa contribute to racial disparities and patient survival, African Americans are underrepresented in clinical trials, making it difficult to assess the effectiveness of cancer therapies in African American patients.
Obesity has a significant positive association with PCa risk. For low-grade PCa, obesity was positively associated with risk among African American men, as presented in this review. This effect on low-grade cancer increased significantly with obesity in African Americans. For high-grade cancer, the risk associated with obesity is even higher among African Americans. Public policies that aim to reduce obesity among African Americans may be important in the prevention of PCa and other diseases in this population.