Intravesical OnabotulinumtoxinA Injection for Overactive Orthotopic Ileal Neobladder: Feasibility and Efficacy
Article information
Abstract
The efficacy of intravesical onabotulinumtoxinA (BTXA) in the treatment of overactive bladder (OAB) has been well documented. The use of BTXA injection in orthotopic neobladders is yet to be studied. We present 4 cases of patients injected with intravesical BTXA for overactive orthotopic ileal neobladder. We recorded patient demographics, presenting and follow-up symptoms, urodynamic profiles, and Patient Global Impression of Improvement (PGI-I) scores. The 4 patients reported varying degrees of subjective improvements in the symptoms, including urgency, urge incontinence, and pad usage. Mean follow-up duration was 8.3 months (range, 5–14 months). Average PGI-I score was 3 (“a little better”) (range, 2–4). To our knowledge, the current study is the first case series examining BTXA injection for orthotopic neobladder overactivity. BTXA injection yielded varying degrees of objective and subjective improvements, without significant complications. Intravesical BTXA injection is feasible and may be considered as a potential treatment alternative for OAB in orthotopic neobladders, although further study is warranted.
Although excellent continence rates have been described following radical cystectomy and orthotopic ileal neobladder, voiding dysfunction remains prevalent [1]. Urinary frequency, nocturia, as well as day and night-time incontinence are potential issues [1]. Treatment options remain limited for this challenging condition. To our knowledge, the use of OnabotulinumtoxinA (BTXA; Allergan, Irvine, CA, USA) for neobladder overactivity is yet to be studied.
CASE REPORTS
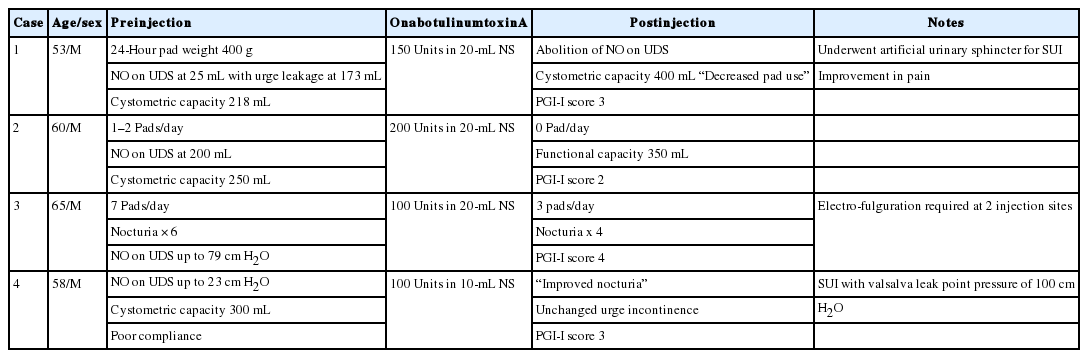
Four patients in the case series are described below. A modified Patient Global Index of Improvement (PGI-I) scale was used to quantify patient perceived outcomes according to Appendix 1 [2]. Patient demographics, clinical parameters, procedure information, and outcomes are summarized in Table 1.
Case 1
A 53-year-old man underwent radical cystoprostatectomy and ileal neobladder in 2011. The patient reported urgency, urge incontinence, and stress urinary incontinence (SUI) with an average 24-hour pad weight of 400 g. For a trial, the patient was administered oral anticholinergic therapy that ameliorated the suprapubic pain, although urgency and incontinence remained unchanged.
Urodynamic studies (UDS) demonstrated phasic neobladder overactivity at 25 mL, 95 mL (up to 40 cm H2O), and 173 mL (up to 83 cm H2O with leakage) (Fig. 1).
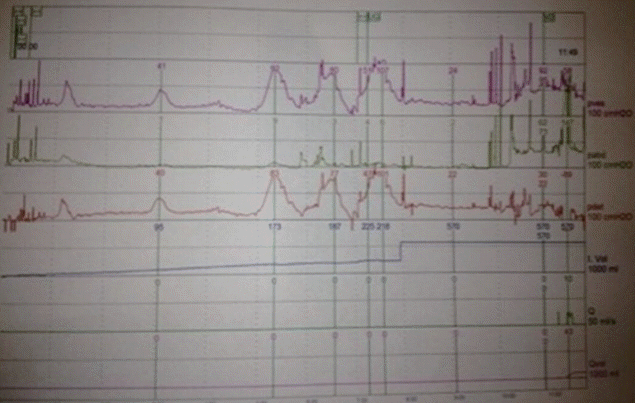
“Case 1” preinjection urodynamic study demonstrating phasic neobladder overactivity and maximum cystometric capacity of 218 mL.
Persistent filling resulted in further phasic neobladder overactivity to 101 cm H2O with leakage. Cystometric neobladder capacity was 218 mL. Valsalva leak point pressure (VLPP) was 62 cm H2O. The patient voided by Valsalva maneuver with a maximum flow rate (Qmax) of 10 mL/sec and postvoid residual of 50 mL.
The patient was injected 150 units of BTXA in 20-mL normal saline into the neobladder mucosa without any complication. He reported subjective improvement in urinary urgency, urge incontinence, and pad use after the injection.
The patient’s PGI-I score was 3. UDS demonstrated elimination of the phasic neobladder overactivity, a cystometric bladder capacity of 400 mL, but decreasing compliance (Fig. 2).
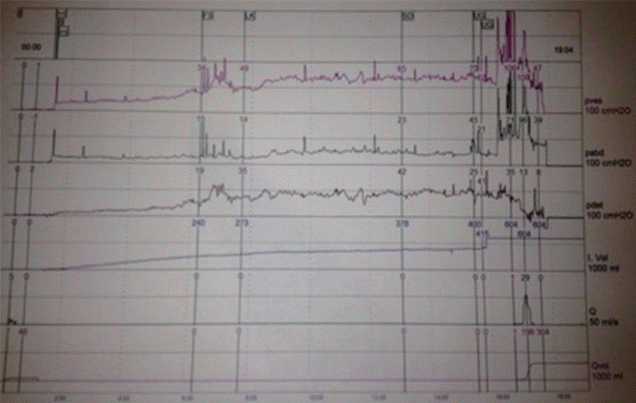
“Case 1” postinjection urodynamic study demonstrating abolition of phasic neobladder overactivity and maximum cystometric capacity of 400 mL.
Repeat injections of 200 units were continued up to 4 months after the initial injection. The patient subsequently underwent artificial urinary sphincter insertion for persistent SUI. At 14-month follow-up, he is currently pad free and is monitored with upper tract ultrasonography.
Case 2
A 60-year-old man underwent radical cystoprostatectomy and ileal neobladder in 2006. At 18 months postoperatively, he developed increasing urinary urgency and urge incontinence, requiring 1–2 pads per day. Voiding was achieved by increasing the intra-abdominal pressure. UDS demonstrated urgency with neobladder overactivity at 200 mL. The cystometric bladder capacity was 250 mL and Qmax was 10 mL/sec.
The patient was injected 200 units of BTXA into the neobladder mucosa without any complication. Postoperatively, up to 8 months of follow-up, he reported improved urgency and no incontinence. Functional bladder capacity was 350 mL. The patient’s PGI-I score was 2.
Case 3
A 65-year-old man underwent radical cystoprostatectomy and orthotopic ileal neobladder in 1999. The patient developed renal failure and underwent renal transplantation. He was referred for urinary incontinence postoperatively. The patient reported intermittent sterile catheterization (ISC) hourly with volumes up to 500 mL. He reported troublesome urge urinary incontinence between ISC, requiring 7 pads per day and nocturia 6 times per night.
UDS demonstrated neobladder overactivity during filling (up to 79 cm H2O). The cystometric capacity was 430 mL, with no leakage demonstrated.
The patient was injected 100 units of BTXA in 0.5-mL aliquots in the neobladder. Electro-fulguration was required to obtain hemostasis at 2 injection sites. At 2-month follow-up, the patient reported minimal subjective improvement in urgency and urge urinary incontinence. He continued hourly ISC, requiring 3 pads per day, with nocturia 4 times per night. The patient’s PGI-I score was 4. The patient was injected 200 units of BTXA repeatedly for up to 6 months from date of first injection, with no additional improvement. Repeat UDS post-injection demonstrated reduced-amplitude overactivity, with SUI and impaired compliance.
Case 4
A 58-year-old man underwent radical cystoprostatectomy and ileal neobladder in 2011. The patient complained of sudden leakage at rest, as well as SUI. Functional bladder capacity was 200 mL. UDS demonstrated impaired compliance (Pdet 30 cm H2O at 300 mL) and neobladder overactivity up to 23 cm H2O during filling. SUI was demonstrated, with VLPP of 100 cm H2O.
Solifenacin and buscopan had been trialed without benefit. The patient was injected 100 units of BTXA, without any complications. Upon follow-up at 5 months, the patient reported some improvement in nocturnal enuresis, though minimal improvement overall. Postinjection PGI-I score was 3.
Of note, no significant adverse effects such as infection, hematuria, urinary retention, or systemic side effects occurred in any of the four patients.
DISCUSSION
Currently, treatment options for neobladder overactivity are limited. Oxybutynin and verapamil both have demonstrated improvements in the urodynamic parameters in the treatment of nocturnal enuresis after ileal neobladder [3]. It was noted that volume at first desire, frequency of uninhibited contractions, and maximum amplitude of uninhibited contractions improved significantly with both oxybutynin and verapamil. Oxybutynin had a better subjective response rate than verapamil, although the adverse effects were more common [3]. Injecting BTXA after failed augmentation cystoplasty has also been reported. BTXA showed improved symptoms of varying degrees in 55.5% of patients, although the technical aspects of injection techniques were not described [4].
The mechanism of action of intravesical BTXA injections is attributed to inhibited SNARE (soluble NSF attachment protein receptor)-complex proteins, leading to a decreased release of acetylcholine, adenosine triphosphate, and substance P [5]. In addition, BTXA injections have also demonstrated peripheral anti-nociceptive properties distinct from their function in neuromuscular blockade [6].
It is also worth noting that case 1 reported significant improvement in subjective “crampy” pain, in addition to the decreasing sensation of neobladder fullness and urgency. This may support the mechanism of action proposed by Apostolidis et al. [5] in inhibited afferent and efferent pathways, and a central desensitization via a decrease in substance P and neurotropic factors.
Because neobladder BTXA injections have not been described, the optimal injection technique remains to be elucidated. Technical considerations are the relative thinness of the bowel mucosa compared to the detrusor and the associated potential for bowel injury or perforation of the neobladder. Ways to circumvent this issue would be to ensure a minimal depth of needle penetration into the neobladder mucosa, or using a shorter injection needle. In the cases presented, it was found that the technical component of injection was relatively straightforward (Fig. 3).

Intraoperative endoscopic view demonstrating onabotulinumtoxinA injection into the neobladder mucosa.
In the 4 described cases, the needle was inserted approximately 2–3 mm into the neobladder mucosa and 100–200 units of BTXA was delivered in 0.5- to 1-mL injection volumes with a concentration varying between 5–10 units per mL. An approach similar to standard bladder BTXA injections was adopted according to the surgeon’s preference. Amount of BTXA delivered and choice of sites of injection were at the discretion of the surgeon, as no protocols for neobladder injection exist. General anesthesia was administered in all cases. No adverse events were noted. The only intraoperative complication was minor bleeding from 2 injection sites in case 3. Satisfactory hemostasis was achieved using endoscopic diathermy.
The optimal BTXA dosing for neobladder injection has yet to be investigated. For idiopathic OAB, 100 units is the preferred starting dose, as it has been thought to best balance BTXA efficacy and safety [7]. Two large phase III studies on BTXA in the setting of neurogenic detrusor overactivity compared placebo to both 200 and 300 units. These showed no significant benefit of 300 over 200 units, with adverse effects being more prevalent in the 300 unit group [8,9].
Though BTXA is generally well tolerated, systemic side effects, such as weakness, can occur. It is thought to be related to the migration of toxin away from the detrusor [10]. In the idiopathic OAB population undergoing BTXA injection, the rate of urinary retention is approximately 5% [10]. The pharmacodynamic changes that may develop during absorption when injecting into the small bowel mucosa as compared to bladder injections remain unknown. Patients with orthotopic ileal neobladder typically void with abdominal straining, which combined with the tendency towards SUI after radical cystectomy, would suggest the risk of urinary retention post injection to be minimal. We would therefore propose a BTXA dose of 200 units as most appropriate for overactive neobladder.
Managing voiding dysfunction in orthotopic neobladders can be challenging. This series of 4 patients injected with neobladder BTXA provides a potential therapeutic alternative. The patients in this series demonstrated varying degrees of improvement. BTXA can be helpful in the long-term management of neobladder overactivity. It also has the potential to improve symptoms while the neobladder gains functionality or may potentially obviate the need for neobladder augmentation. We believe that the feasibility of neobladder BTXA injections has been demonstrated in this series, as they were administered without any difficulty or complication. Efficacy of the therapy will be more difficult to substantiate because of the complexity of these patients and the inherent element of SUI. However, it does present an intriguing area for further study, and could prove to be useful tool for the urologists in managing a difficult clinical entity in the future.
Notes
Conflict of Interest
Vincent Tse: Allergan (Advisory board), Americal Medical Systems (Speaking honorarium); Astellas (Speaking honorarium); Audrey Wang: Allergan (Advisory board); Johan Gani: Medtronic (Speaking honorarium), Astellas (Principal investigator). Except for that, no potential conflict of interest relevant to this article was reported.