Association of a Missense ALDH2 Single Nucleotide Polymorphism (Glu504Lys) With Benign Prostate Hyperplasia in a Korean Population
Article information
Abstract
Purpose
Aldehyde dehydrogenase 2 (ALDH2) is a well-known gene involved in alcohol and aldehyde metabolism. Moreover, recent studies have reported associations between ALDH2 and age-related disorders. Benign prostate hyperplasia (BPH) is an age-related disorder and genetic factors may contribute to its onset. In this study, we investigated the association of a well-studied ALDH2 single nucleotide polymorphism (SNP), rs671, with the onset and clinical features of BPH.
Methods
A total of 222 BPH patients and 214 control subjects were genotyped. The clinical features of the BPH patients (prostate volume, prostate-specific antigen level, and International Prostatic Symptom Score) were analyzed.
Results
The results show that rs671 was only associated with the volume of BPH in genotype and allele frequencies (P<0.05).
Conclusion
We propose that rs671 is an Asian-specific SNP in ALDH2 that may affect the disease progression of BPH in the Korean population.
INTRODUCTION
Aldehyde dehydrogenase 2 (ALDH2) has a role in the metabolism of aldehydes [1], and its function in alcohol metabolism pathways has been well studied. However, many recent studies have reported that ALDH2 may also be associated with chronic and age-related disorders.
A genome-wide association study (GWAS) performed in the Japanese population indicated that the chromosomal loci of 12q24, which is close to BRAP and ALDH2, is associated with coronary artery disease [2]. Similar results were reported in another GWAS in East Asians suggesting that 12q24.13, which is near to ALDH2, was strongly associated with blood pressure [3]. ALDH2 polymorphism was linked to essential hypertension in Mongolians [4]. Hypertension is a component of metabolic syndrome, and interestingly, certain ALDH2 genotypes have also been proposed as risk factors for metabolic syndrome with liver dysfunction [5].
Although benign prostate hyperplasia (BPH) and cardiovascular disease, which is associated with hypertension, arise due to separate pathological processes, both share similarities in age-related elevations in sympathetic tone that may contribute to the pathophysiology of disease [6]. Additionally, the International Prostatic Symptom Score (IPSS), which is usually used to assess BPH, was found to correlate with blood pressure [7]. In many cases, BPH coexists with diabetes [8] or metabolic syndrome [9]. Since such diseases are prevalent in aged men, there may be a genetic factor that affects the aging process and in turn, contributes to the underlying pathophysiology.
ALDH2 is an isoform of aldehyde dehydrogenase present in mitochondria; a specific ALDH2 polymorphism, known as ALDH2*2, is present in North-East Asians [10]. Research suggests that ALDH2 polymorphisms may exhibit deficient enzyme activity. ALDH2 polymorphism is associated with late-onset Alzheimer disease in apolipoprotein E4 carriers, and mutant ALDH2 is linked to cellular vulnerability to peroxidized lipids, which may lead to geriatric diseases [10].
Family history is a risk factor for BPH [11,12], and disease features in BPH patients are linked to various genetic polymorphisms [13-16]. The aforementioned studies suggest that polymorphisms in ALDH2 may increase the susceptibility to BPH, with no difference in the age of individuals and exposure to environmental factors and stressful conditions being observed in these studies [17]. However, there is a lack of studies examining whether ALDH2 polymorphisms contribute to BPH. Therefore, in this study, we investigated the association of a single nucleotide polymorphism (SNP) in ALDH2 with the onset and clinical features of BPH.
MATERIALS AND METHODS
Study subjects
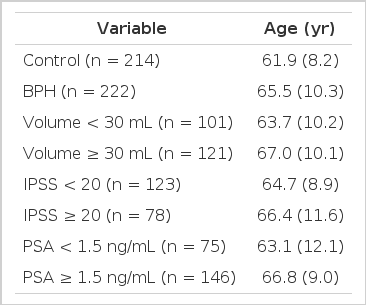
BPH patients were recruited from Kyung Hee Medical Center between January 2002 and December 2008. Table 1 displays the demographic and clinical features of the control and BPH groups. A total of 222 BPH patients and 214 control subjects were included in the study. The control group was composed of middle-aged to elderly males, who were free of a severe disease. The lower urinary tract symptoms (LUTS) of BPH patients were scaled using IPSS. Serum prostate-specific antigen (PSA) levels were tested in all BPH patients. Prostate sizes of the patients were estimated using transrectal ultrasound, and those patients who exhibited serum PSA levels above 4 ng/mL underwent prostate biopsies. Any patients with urinary tract infections, prostate cancer, a neurogenic bladder, uncontrolled diabetes mellitus, and/or cardiovascular diseases were not included in this study. In the analysis of clinical features, BPH patients were divided into groups based on the following features: IPSS (<20 vs. ≥20), serum PSA level (<1.5 ng/mL vs. ≥1.5 ng/mL), and prostate volume (<30 mL vs. ≥30 mL) (Table 1). Ethical approval of this study was obtained from the Ethics Review Committee of Medical Research Institute, Kyung Hee University School of Medicine, Seoul, Korea.
SNP selection and genotyping
We selected one missense SNP, rs671 (Glu504Lys), of the ALDH2 gene (http://www.ncbi.nlm.nih.gov/SNP) for analysis. Peripheral blood samples from each patient were stored in ethylenediaminetetraacetic acid in blood sampling tubes at -20℃. Genomic DNA was extracted using the QIAamp DNA mini kit (QIAGEN, Valencia, CA, USA). Polymerase chain reaction experiments were performed to amplify target sequences including rs671 (sense 5'-CTCAGGAAGCTGAGGCAGGA-3', antisense 5'-GGCTGGGTCTTTACCCTCTC-3', 652 base pairs). Direct sequencing and analysis were performed with an ABI PRISM 3730XL analyzer (PE Applied Biosystems, Foster City, CA, USA) and SeqManII software (DNASTAR, Madison, WI, USA).
Statistical analysis
IBM SPSS ver. 20.0 (IBM Co., Armonk, NY, USA) was used to obtain odds ratios (ORs), 95% confidence intervals (CIs), and P-values adjusted for age as a covariable. Multiple logistic regression models (codominant1, codominant2, dominant, and recessive models) were applied in the analysis of genotypes. P<0.05 was considered significant.
RESULTS
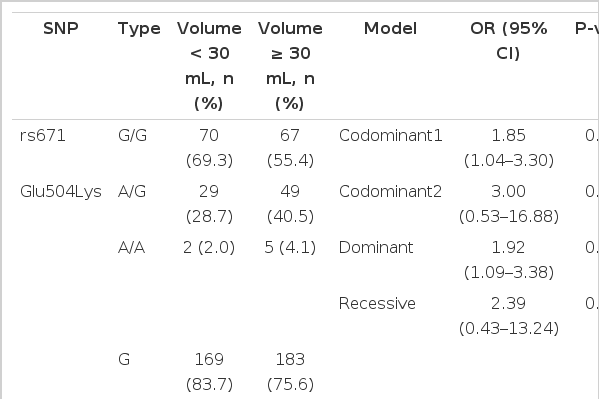
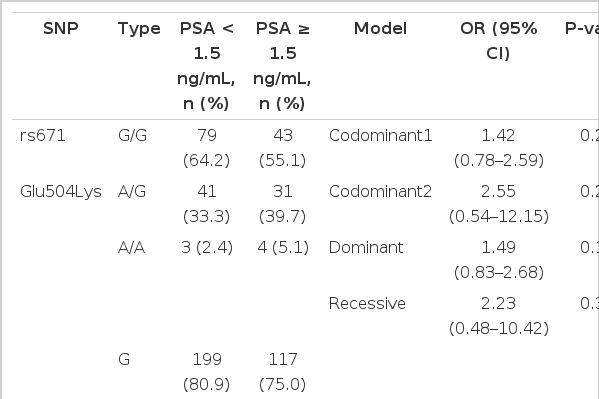
The genotypic distribution of rs671 in the study subjects was in Hardy-Weinberg equilibrium (data not shown). The distribution of the tested SNP was not significantly different between control and BPH groups (Table 2). However, in the analysis of clinical features in BPH patients, the genotypes (codominant1: OR, 1.85; 95% CI, 1.04-3.30, P=0.037; dominant: OR, 1.92; 95% CI, 1.09-3.38, P=0.024) and allele distribution (OR, 1.65; 95% CI, 1.03-2.65; P=0.038) were associated with larger prostates in the BPH group (Table 3). IPSS (Table 4) and PSA (Table 5) were not significantly associated with rs671 in the tested population.
DISCUSSION
ALDH2 is located on 12q24.2 (http://www.ncbi.nlm.nih.gov/gene/217) and this locus has been linked to drinking behavior in the Chinese [18], alcohol dependence [19], squamous carcinoma [20], and obesity [21]. Moreover, a microdeletion in the locus was associated with insulin-dependent diabetes [22]. Such diseases, including obesity [23], are related to increased alcohol ingestion and ALDH2 activity in individuals. ALDH2*2 type, which is prevalent in North-East Asians, shows decreased enzyme activity [10]. Accordingly, rs11067228, which is a SNP on 12q24, was not associated with PSA levels in Caucasian men [24].
Aldehyde products in adipose tissue may contribute to obesity [23], and the detoxification of aldehydes may help protect against the oxidative stress of lipid oxidation [25]. ALDH2 belongs to the aldehyde dehydrogenase family, which functions to metabolize alcohols and other aldehyde derivatives produced in lipid metabolism [10]. This suggests that lipid peroxidation in prostate inflammation may be ameliorated by high ALDH2 activity, since reductions in aldehydes are beneficial in inflamed tissues [23,26], and aldehyde production is significantly elevated in BPH [27].
Although many SNPs exist in the exon region of ALDH2 (http://www.ncbi.nlm.nih.gov/snp), the majority are within the range of minor allele frequency (MAF) <0.05, and there is an absence of genotype-phenotype correlations. One missense SNP, rs671, which was investigated in this study, is monotype (G allele) in the European population, MAF 0.156 (major allele G and minor allele A) in the Chinese population, and MAF 0.233 (also, major allele G and minor allele A) in the Japanese population. The MAF of rs671 in all subjects in this study was most similar to that of the Japanese population; however, the MAF in the control group was similar to the Chinese population. In line with previous studies, our results show that the missense ALDH2 polymorphism, rs671 (Glu504Lys), may affect certain diseases in Asians.
The wild-type variant of rs671 is known as ALDH2*1 and the East Asian variant type, which is an inactive version of ALDH2, is called ALDH2*2 [28]. ALDH2*2 is associated with significantly lower activity in the metabolism of short chain aliphatic aldehydes, such as formaldehyde, propionaldehyde, n-butyraldehyde, capronaldehyde, and heptaldehyde [29]. Malondialdehyde is a short chain aliphatic aldehyde that has been considered as a marker for oxidative stress in many studies; it is also associated with prostate hypertrophy [27]. Previous research indicates a relationship between BPH and the biological function of ALDH2 types.
In our study, rs671 was not associated with BPH development. However, prostate volume was associated with ALDH2 type. Interestingly, a recent report suggested that chronic inflammation may lead to the progression of BPH, and such progression was characterized by prostate calcifications, volume, LUTS severity, and prostatitis-like symptoms [30]. This partly correlates with our observation that chronically elevated aldehyde levels may increase inflammatory responses in BPH, resulting in larger prostate volumes. The minor allele in this study was A, and this corresponds with the previously reported ALDH2*2 type, an inactive form that may lead to the decreased oxidation of aldehydes. The minor allele in this study was associated with elevated ORs (1.85 in codominant1, 1.92 in dominant, and 1.65 in alleles). Moreover, the dominant model correlated with a more progressive form of BPH (significant in age-adjusted males), which corresponds to previous findings that ALDH2*1/*2 type exhibited reduced aldehyde metabolism [29].
In conclusion, we suggest that the missense ALDH2 polymorphism, rs671, may contribute to BPH disease progression. There were limitations in our study: only Korean populations were included and hypertension was not analyzed due to inappropriate clinical data. Therefore, further study using a larger pool of BPH patients and more appropriate controls is required to confirm our results.
ACKNOWLEDGEMENTS
This work was carried out with the support of "Cooperative Research Program for Agriculture Science & Technology Development (Project No. PJ009558)" Rural Development Administration, Korea.
Notes
No potential conflict of interest relevant to this article was reported.