|
 |


- Search
| Int Neurourol J > Volume 19(3); 2015 > Article |
|
ABSTRACT
Purpose:
Methods:
Results:
NOTES
REFERENCES
Fig. 1.
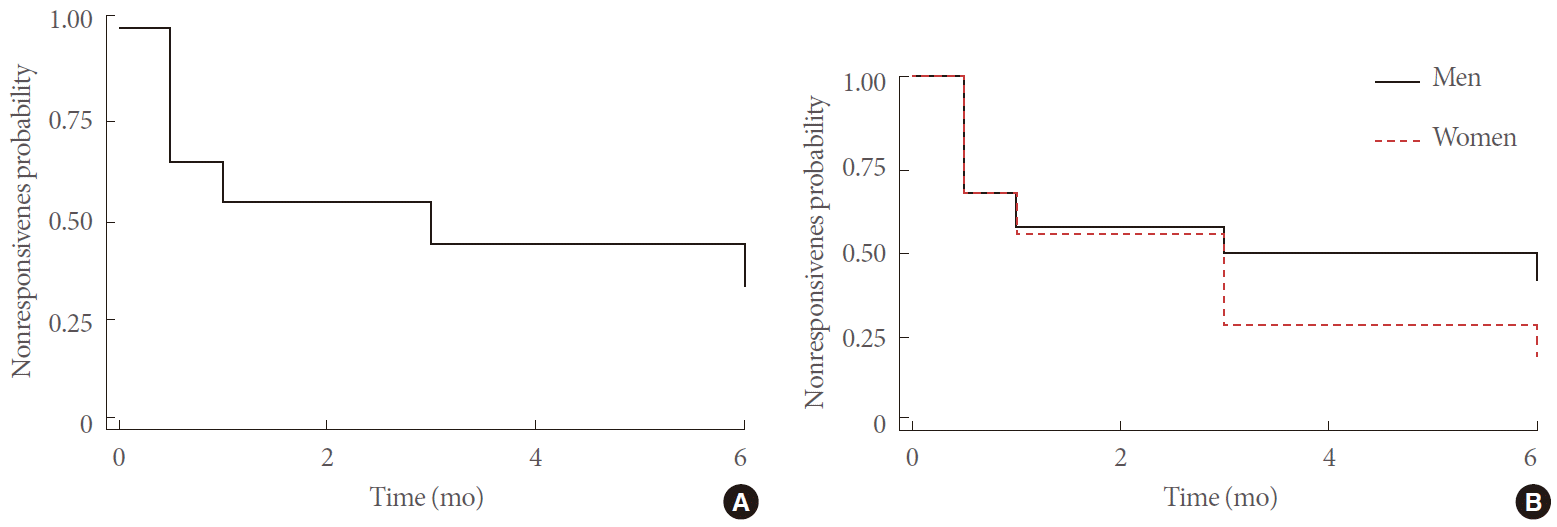
Fig. 2.
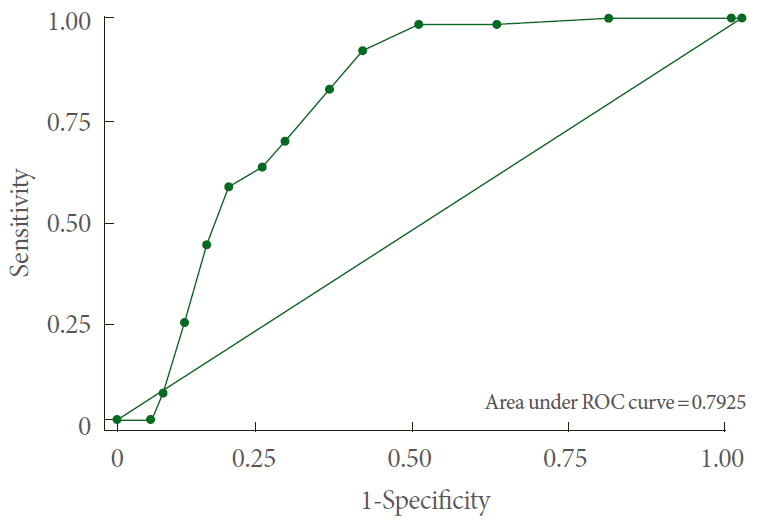
Table 1.
| Variable | Total (n = 125) | Women (n = 44) | Men (n = 81) | P-valuea) |
|---|---|---|---|---|
| Age (yr) | 71.1 ± 13.0 | 66.5 ± 15.2 | 73.5 ± 11.0 | 0.007 |
| OAB-wet | 98 (78) | 32 (73) | 66 (81) | 0.358 |
| Treatment | ||||
| Solifenacin | 106 (85) | 39 (89) | 67 (83) | 0.444 |
| Tolterodine | 19 (15) | 5 (11) | 14 (17) | - |
| Diabetes mellitus | 21 (17) | 8 (18) | 13 (16) | 0.761 |
| Hypertension | 30 (24) | 8 (18) | 22 (27) | 0.262 |
| CRF | 7 (6) | 2 (5) | 5 (6) | 1.000 |
| Stroke | 10 (8) | 3 (7) | 7 (9) | 0.740 |
| Parkinsonism | 6 (5) | 1 (2) | 5 (6) | 0.424 |
| BPH in men (n = 81) | 69 | - | 69 | - |
| Concomitant treatment for BPH | ||||
| Alpha blocker | 59 | - | 59 | - |
| 5ARI | 12 | - | 12 | - |
| PPBC | 3.7 ± 1.7 | 3.9 ± 1.7 | 3.6 ± 1.7 | 0.332 |
| OABSS | 8.6 ± 3.6 | 8.5 ± 3.8 | 8.7 ± 3.6 | 0.729 |
| IUSS | 3.4 ± 1.3 | 3.2 ± 1.5 | 3.4 ± 1.2 | 0.452 |
| IPSS-V | 5.9 ± 5.5 | 5.0 ± 5.7 | 6.4 ± 5.4 | 0.137 |
| IPSS-S | 7.7 ± 3.5 | 8.4 ± 3.4 | 7.4 ± 3.5 | 0.116 |
| TPV (mL) | 40.7 ± 18.2 | - | 40.7 ± 18.2 | - |
| TZI (%) | 32.7 ± 13.5 | - | 32.7 ± 13.5 | - |
| Qmax (mL/sec) | 13.3 ± 7.9 | 16.5 ± 9.9 | 11.5 ± 5.8 | 0.007 |
| VV (mL) | 171 ± 102 | 176 ± 106 | 168 ± 99 | 0.831 |
| PVR (mL) | 46.9 ± 57.6 | 43.8 ± 54.0 | 48.6 ± 59.8 | 0.416 |
| Response | 62 (50) | 23 (52) | 39 (48) | 0.660 |
| Dry mouth | 24 (19) | 9 (20) | 15 (19) | 0.793 |
| Constipation | 4 (3) | 1 (2) | 3 (4) | 1.000 |
| Dizziness | 4 (3) | 2 (5) | 2 (2) | 0.613 |
Values were presented as mean±standard deviation or number (%) unless otherwise indicated.
OAB, overactive bladder; CRF, chronic renal failure; BPH, benign prostate hyperplasia; 5ARI, 5-alpha reductase inhibitor; PPBC, patient perception of bladder condition; OABSS, overactive bladder symptom score; IUSS, indevus urgency severity score; IPSS, International Prostate Symptom Score; IPSS-V, IPSS voiding subscore; IPSS-S, IPSS storage subscore; TPV, total prostate volume; TZI, transition zone index; Qmax, maximum flow rate; VV, voided volume; PVR, postvoidal residual.
Table 2.
HR, hazard ratio; CI, confidence interval; OAB, overactive bladder; CRF, chronic renal failure; BPH, benign prostate hyperplasia; 5ARI, 5-alpha reductase inhibitor; PPBC, patient perception of bladder condition; OABSS, overactive bladder symptom score; IUSS, indevus urgency severity score; IPSS, International Prostate Symptom Score; IPSS-V, IPSS voiding subscore; IPSS-S, IPSS storage subscore; TPV, total prostate volume; TZI, transition zone index; Qmax, maximum flow rate; VV, voided volume; PVR, postvoidal residual.
Table 3.
| Variable |
Baseline |
Change from baseline |
ρ for responsivenessb) | ||||
|---|---|---|---|---|---|---|---|
| Response | Nonresponse | P-valuea) | Response | Nonresponse | P-valuea) | ||
| Age (yr) | 69.8 ± 13.2 | 72.3 ± 12.8 | 0.237 | ||||
| Male sex | 39 (63) | 42 (67) | 0.660 | ||||
| OAB wet | 59 (60) | 3 (12) | <0.001 | ||||
| PPBC | 4.2 ± 1.5 | 3.2 ± 1.7 | 0.002 | –1.9 ± 2.1 | –0.2 ± 1.8 | <0.001 | –0.44*** |
| OABSS | 10.4 ± 2.6 | 6.6 ± 3.7 | <0.001 | –5.2 ± 3.6 | 1.0 ± 2.9 | <0.001 | –0.75*** |
| IUSS | 3.9 ± 0.3 | 2.8 ± 1.7 | <0.001 | –2.1 ± –2.5 | 0.0 ± 1.7 | <0.001 | –0.48*** |
| IPSS-V | 5.7 ± 5.6 | 6.1 ± 5.5 | 0.641 | –1.4 ± 5.1 | –0.9 ± 4.6 | 0.520 | –0.06 |
| IPSS-S | 8.4 ± 3.4 | 7.0 ± 3.5 | 0.017 | –3.2 ± 4.4 | –0.1 ± 3.9 | <0.001 | –0.38*** |
| Qmax (mL/sec) | 13.5 ± 8.5 | 13.1 ± 8.5 | 0.913 | 4.5 ± 10.8 | 2.8 ± 6.9 | 0.553 | 0.05 |
| VV (mL) | 170.5 ± 98.2 | 171.3 ± 106.6 | 0.811 | 80.3 ± 141.3 | 45.6 ± 123.6 | 0.217 | 0.11 |
| PVR (mL) | 49.8 ± 63.5 | 43.5 ± 50.1 | 0.713 | 8.1 ± 80.0 | 35.8 ± 70.0 | 0.082 | –0.16 |
Values are presented as mean±standard deviation or number (%).
OAB, overactive bladder; PPBC, patient perception of bladder condition; OABSS, overactive bladder symptom score; IUSS, indevus urgency severity score; IPSS, International Prostate Symptom Score; IPSS-V, IPSS voiding subscore; IPSS-S, IPSS storage subscore; Qmax, maximum flow rate; VV, voided volume; PVR, postvoidal residual.