|
 |


- Search
| Int Neurourol J > Volume 20(3); 2016 > Article |
|
ABSTRACT
Purpose
Methods
Results
Conclusions
NOTES
REFERENCES
Fig.┬Ā1.
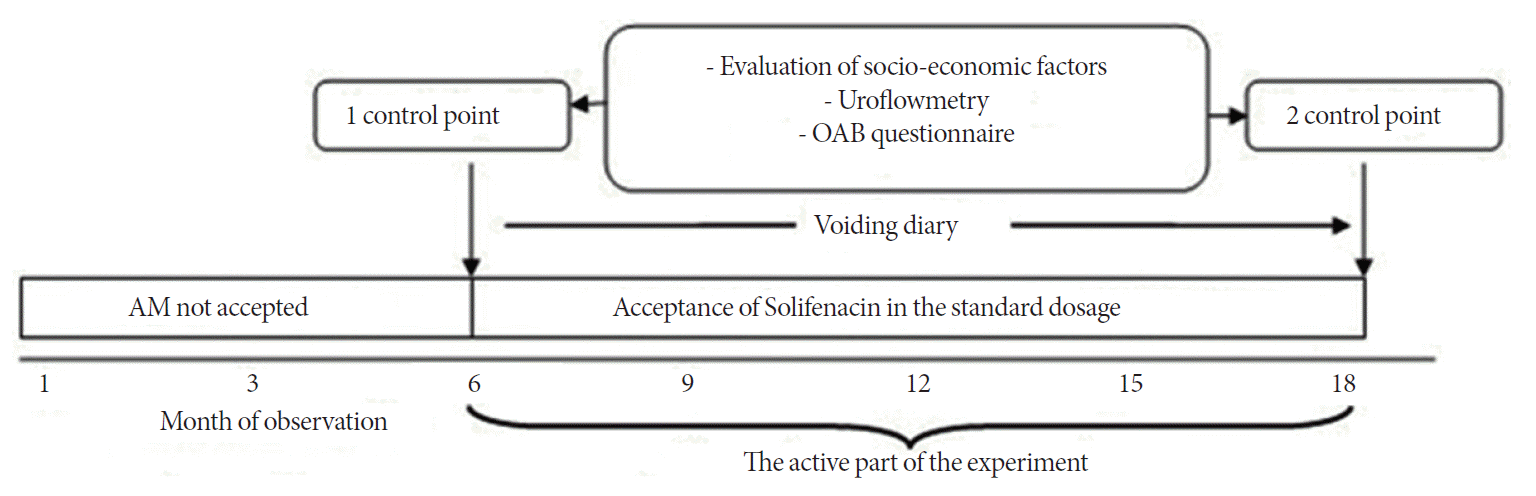
Fig.┬Ā2.
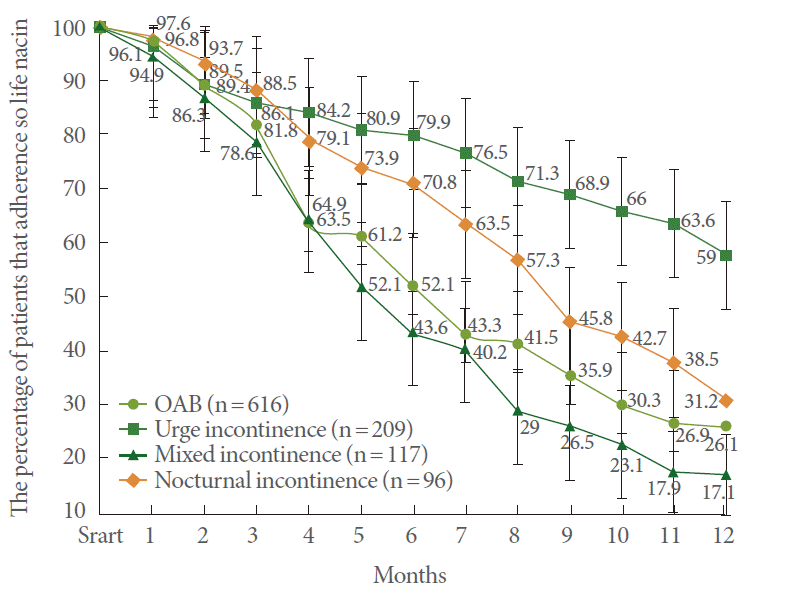
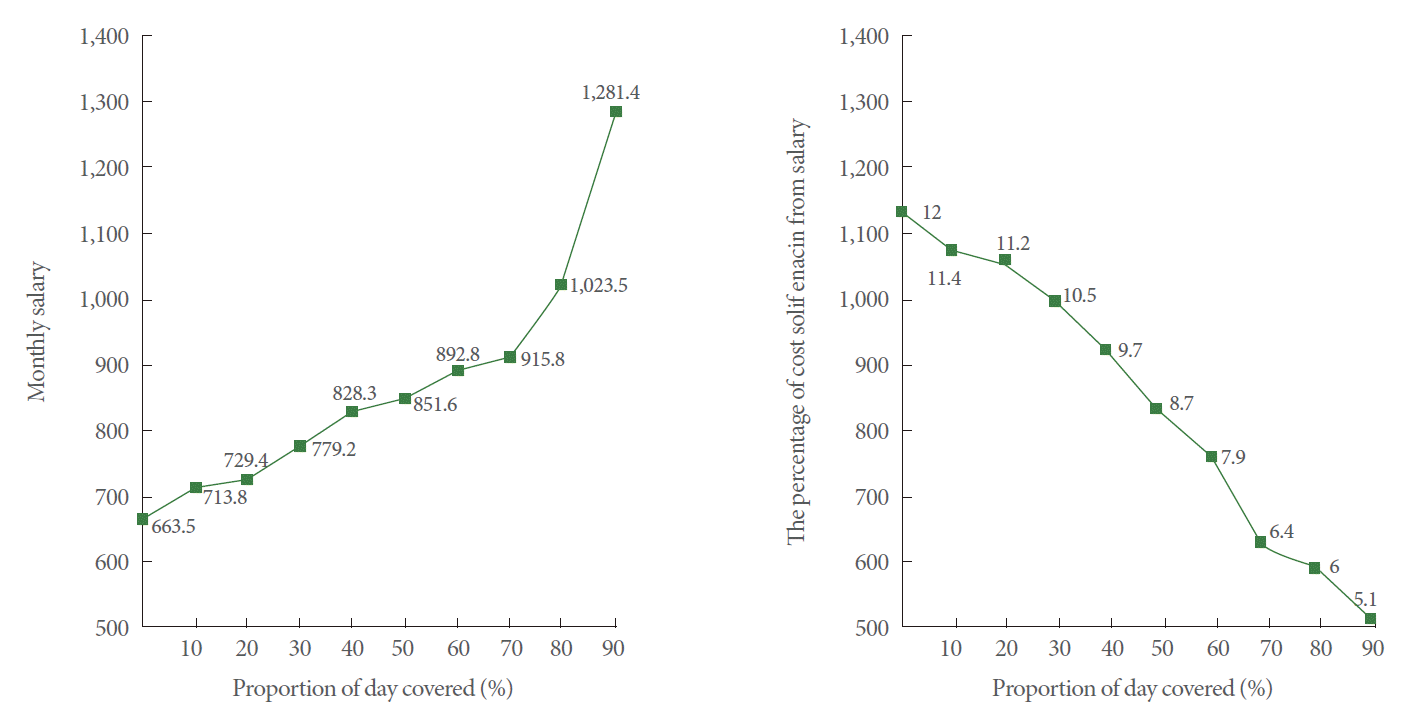
Fig.┬Ā3.

Fig.┬Ā5.
Table┬Ā1.
| Parameter | Group 1 (n=191) | Group 2 (n=481) | Group 3 (n=366) |
P-value |
||
|---|---|---|---|---|---|---|
| P1/2 | P2/3 | P1/3 | ||||
| Age (yr) | 32.7┬▒7.8 | 39.2┬▒11.2 | 53.8┬▒7.9 | Ōēź0.05 | Ōēź0.05 | Ōēż0.01 |
| Female sex | 112 (58.6) | 295 (61.3) | 214 (58.5) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Married | 125 (65.4) | 318 (66.1) | 281 (76.8) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Not married | 66 (34.5) | 163 (33.9) | 85 (23.2) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Children | 1.2┬▒0.4 | 0.9┬▒0.4 | 1.4┬▒0.7 | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Field of activity | ||||||
| ŌĆāIndustry | 38 (19.9) | 107 (22.2) | 63 (17.2) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| ŌĆāTransportation | 20 (10.5) | 21 (4.3) | 9 (2.4) | Ōēź0.05 | Ōēź0.05 | Ōēż0.05 |
| ŌĆāRetail | 21 (11.0) | 37 (7.7) | 36 (9.8) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| ŌĆāAgriculture | 31 (16.2) | 61 (12.7) | 29 (7.9) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| ŌĆāEducation | 18 (9.4) | 129 (26.8) | 107 (29.2) | Ōēż0.05 | Ōēź0.05 | Ōēż0.01 |
| ŌĆāMedicine | 27 (14.1) | 84 (17.4) | 96 (26.2) | Ōēź0.05 | Ōēż0.05 | Ōēż0.01 |
| ŌĆāOther | 36 (18.8) | 42 (8.7) | 26 (7.1) | Ōēż0.05 | Ōēź0.05 | Ōēż0.05 |
| Annual salary $ USA | 7,933.20┬▒2,455.8 | 11,494.80┬▒2,712.8 | 19,947.60┬▒3,829.3 | Ōēź0.05 | Ōēż0.05 | Ōēż0.01 |
| The average salary monthly $ USA | 661.1┬▒208.5 | 957.9┬▒318.9 | 1662.3┬▒405.7 | Ōēź0.05 | Ōēź0.05 | Ōēż0.05 |
| Constant work (yr) | 6.7┬▒2.3 | 11.4┬▒7.2 | 12.1┬▒6.9 | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Full working day | 177┬▒92.7 | 451┬▒93.8 | 302┬▒82.5 | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Combining work | 56┬▒29.3 | 115┬▒23.9 | 119┬▒32.5 | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Monthly expenses on Solifenacin $ USA | 40.7┬▒6.1 | 40.7┬▒4.2 | 40.7┬▒2.4 | Ōēź0.05 | Ōēź0.05 | Ōēż0.05 |
| Monthly expenditure on other medications $ USA | 59.8┬▒13.8 | 71.3┬▒22.5 | 73.9┬▒11.5 | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Employee turnover | 27 (14.1) | 61 (12.3) | 58 (15.8) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Urge UI | 21 (10.0) | 69 (33.0) | 119 (56.9) | Ōēż0.05 | Ōēż0.01 | Ōēż0.01 |
| Mixed UI | 36 (30.8) | 31 (26.5) | 50 (42.7) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Nocturnal UI | 29 (30.2) | 37 (38.5) | 30 (31.2) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| OAB | 167 (27.1) | 245 (39.8) | 202 (32.8) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Severe symptoms (EUI Ōēź3/day) | 34 (7.0) | 182 (37.7) | 267 (55.3) | Ōēż0.01 | Ōēż0.05 | Ōēż0.01 |
| Satisfaction with treatmenta) | 43 (22.5) | 267 (55.5) | 294 (80.3) | Ōēż0.05 | Ōēż0.05 | Ōēż0.05 |
| Efficacy of treatmentb) | 68 (35.6) | 196 (40.7) | 311 (84.9) | Ōēź0.05 | Ōēż0.01 | Ōēż0.01 |
| Unsatisfactory experience in treating | 117 (61.2) | 216 (44.9) | 127 (34.7) | Ōēż0.05 | Ōēź0.05 | Ōēż0.01 |
| Awareness of treatment options | 155 (81.1) | 373 (77.5) | 306 (83.6) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Taking medications to treat comorbidities | 81 (42.4) | 157 (32.6) | 113 (30.9) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Bad habits | ||||||
| ŌĆāAlcohol abuse | 36 (18.9) | 51 (10.6) | 53 (14.5) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| ŌĆāSmoking | 87 (45.5) | 187 (39.3) | 159 (43.4) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| ŌĆāCaffeine abuse | 73 (38.2) | 146 (30.3) | 41 (11.2) | Ōēź0.05 | Ōēż0.05 | Ōēż0.01 |
| Observance of sleep-wake cycle | 145 (75.9) | 288 (59.8) | 231 (63.1) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Charlson Comorbidity Index | 7.7┬▒2.7 | 11.9┬▒5.8 | 11.0┬▒2.1 | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| Kaplan Comorbidity Index | 15.7┬▒4.9 | 14.1┬▒6.9 | 11.3┬▒7.9 | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
Values are presented as median (range), mean┬▒standard deviation, or number (%).
Total percentage of urge, mixed, nocturnal incontinence and OAB in total exceeds 100% due to the fact that the combination of these diseases may occur in one person.
Group 1, compliance<50%; Group 2, 50%Ōēżcompliance<80%; Group 3, complianceŌēź80%; UI, urge incontinence; OAB, overactive bladder; EUI, episodes of urge incontinence; P1/2, significant differences between groups 1 and 2, P2/3, significant differences between groups 2 and 3, P1/3, significant differences between groups 1 and 3.
Table┬Ā2.
| Parameter |
Group 1 (n=191) |
Group 2 (n=481) |
Group 3 (n=366) |
P-value (after treatment) |
|||||
|---|---|---|---|---|---|---|---|---|---|
| Start | Finish | Start | Finish | Start | Finish | P1/2 | P2/3 | P1/3 | |
| OAB-SF questionnaire (in scores) | |||||||||
| ŌĆāDaytime frequency | 4.3 (1.7) | 3.9 (1.8) | 3.8 (1.1) | 3.1 (2.1) | 4.0 (1.1) | 1.2 (1.1)* | Ōēź0.05 | Ōēż0.05 | Ōēż0.05 |
| ŌĆāNight frequency | 3.9 (1.8) | 2.9 (2.2) | 3.8 (2.1) | 3.0 (0.9) | 3.5 (1.2) | 0.9 (1.1)* | Ōēź0.05 | Ōēż0.05 | Ōēż0.05 |
| ŌĆāUrgency | 4.9 (1.1) | 4.0 (0.8) | 3.9 (2.1) | 2.5 (1.7) | 4.2 (2.7) | 2.0 (0.9) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| ŌĆāUrge incontinence | 4.5 (2.1) | 4.1 (1.4) | 4.5 (2.0) | 3.9 (1.2) | 4.7 (0.9) | 1.6 (0.7)* | Ōēź0.05 | Ōēż0.05 | Ōēż0.05 |
| Diaries of urination (No. of episodes/day) | |||||||||
| ŌĆāDaytime frequency | 8.8 (2.0) | 6.7 (1.6) | 9.5 (1.8) | 6.1 (1.4) | 9.6 (2.7) | 5.6 (1.8) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| ŌĆāNight frequency | 2.2 (0.9) | 1.5 (1.1) | 2.0 (0.6) | 1.1 (0.5) | 1.9 (0.6) | 0.8 (0.3) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| ŌĆāUrgency | 5.0 (1.1) | 3.0 (0.8) | 5.3 (2.1) | 3.2 (0.7) | 5.2 (1.3) | 1.3 (0.6) | Ōēź0.05 | Ōēż0.05 | Ōēż0.05 |
| ŌĆāIncontinence | 4.1 (1.5) | 3.8 (0.9) | 5.4 (1.7) | 2.0 (0.9) | 4.3 (0.9) | 1.5 (0.8) | Ōēź0.05 | Ōēź0.05 | Ōēż0.05 |
| Urodynamic parameters (uroflowmetry) | |||||||||
| ŌĆāQaver, mL/sec | 13.7 (4.8) | 17 (2.9) | 11.9 (3.3) | 18.0 (3.7) | 11.1 (3.5) | 20.7 (4.2) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
| ŌĆāQmax, mL/sec | 18.6 (3.9) | 21.4 (3.9) | 17.7 (5.1) | 20.8 (3.9) | 16.9 (5.2) | 26.8 (3.5) | Ōēź0.05 | Ōēź0.05 | Ōēź0.05 |
Values are presented as median (range).
Group 1, compliance<50%; Group 2, 50%Ōēżcompliance<80%; Group 3, complianceŌēź80%; OAB, overactive bladder; OAB-SF, overactive bladder short form; Qaver, average flow rate; Qmax, maximum flow rate; P1/2, significant differences between groups 1 and 2; P2/3, significant differences between groups 2 and 3; P1/3, significant differences between groups 1 and 3.